Abstract
The capacity of zinc-based 2-amino-4-(1H-1,2,4-triazole-4-yl)benzoic acid coordination complex (Zn(NH2-TBA)2) and modified Zn(NH-TBA)2COMe complex for removal of 2,4-dichlorophenoxyacetic acid (2,4-D) from aqueous solutions was investigated through adsorption modeling and artificial intelligence tools. Analyzing the adsorption characteristics of pesticides helps in studying the groundwater pollution by pesticides in agriculture area.
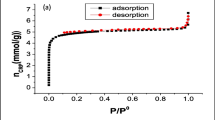
In this study, Zn(NH2-TBA)2 was synthesized using Schiff base and its surface was modified using acetic anhydride group and their physical characteristics were identified using proton NMR, FTIR, and XRD. NMR results showed maximum modification yield obtained was 65% after 5 days. The porous structure and surface area monitored using nitrogen isotherm and BET surface area analysis presented relatively less surface area and porosity after modification. Adsorption modelling indicated that Toth model with a maximum adsorption capacity of 150.8 mg/g and 100.7 mg/g represents the homogenous adsorption systems which satisfy both low- and high-end boundary of adsorbate concentration in all settings according to the optimum point, while the kinetics and rate of 2,4-D adsorption follow the pseudo-first-order kinetic model in all situations. Artificial neural network (ANN), support vector regression, and particle swarm optimized least squares-support vector regression (PSO-LSSVR) were used for the optimization and modelling of adsorbent mass, adsorbate concentration, contact time, and temperature to develop predictive equations for the simulation of the adsorption efficiency of 2,4-D pesticide. The obtained results exhibited the better performance of ANN and PSO-LSSVR for prediction of adsorption results. The mean square error values of ANN (0.001, 0.012) and PSO-LSSVR (0.121, 0.105) were obtained for Zn(NH2-TBA)2 and Zn(NH-TBA)2COMe, respectively, while their respective coefficient of determination (R2) obtained were 0.999 and 0.988 for ANN and 0.980 and 0.825 for PSO-LSSVR. The study specified that machine learning predictive behavior performed better for Zn(NH2-TBA)2 compared to Zn(NH-TBA)2COMe that is also supported by theoretical kinetics and isotherm models. The research concludes that artificial intelligence models are the most efficient tools for studying the predictive behavior of adsorption data.
Graphical Abstract






















Similar content being viewed by others

References
Ahnfeldt T, Gunzelmann D, Wack J, Senker J, Stock N (2012) Controlled modification of the inorganic and organic bricks in an Al-based MOF by direct and post-synthetic synthesis routes. CrystEngComm 14:4126–4136
Amrutha JG, Girish C, Prabhu B, Mayer K (2023) Multi-component adsorption isotherms: review and modeling studies. Environ Process 10:38
Bemani A, Baghban A, Mohammadi AH, Andersen PØ (2020) Estimation of adsorption capacity of CO2, CH4, and their binary mixtures in Quidam shale using LSSVM: Application in CO2 enhanced shale gas recovery and CO2 storage. J Nat Gas Sci Eng 76:103204
Bouchelkia N, Tahraoui H, Amrane A, Belkacemi H, Bollinger J-C, Bouzaza A, Zoukel A, Zhang J, Mouni L (2023) Jujube stones based highly efficient activated carbon for methylene blue adsorption: kinetics and isotherms modeling, thermodynamics and mechanism study, optimization via response surface methodology and machine learning approaches. Process Saf Environ Prot 170:513–535
Chen T, Da T, Ma Y (2021) Reasonable calculation of the thermodynamic parameters from adsorption equilibrium constant. J Mol Liq 322:114980
Chhajer P, Shah M, Kshirsagar A (2022) The applications of artificial neural networks, support vector machines, and long–short term memory for stock market prediction. Decis Anal J 2:100015
Chowdhury I, Doran GS, Stodart BJ, Chen C, Wu H (2023) Fate of herbicide residues in soil-Australian context: insights towards mechanism, aspects, and recent advancements. PREPRINT (Version 1) available at Research Square. https://doi.org/10.21203/rs.3.rs-2685000/v1
Culpepper AS, Sosnoskie LM, Shugart J, Leifheit N, Curry M, Gray T (2018) Effects of low-dose applications of 2, 4-D and dicamba on watermelon. Weed Technol 32:267–272
Dietl C, Hintz H, Rühle B, Schmedt auf der Günne J, Langhals H, Wuttke S (2015) Switch‐on fluorescence of a perylene‐dye‐functionalized metal–organic framework through postsynthetic modification. Chem-A Eur J 21:10714-10720. https://doi.org/10.1002/chem.201406157
Garabrant DH, Philbert MA (2002) Review of 2, 4-dichlorophenoxyacetic acid (2, 4-D) epidemiology and toxicology. Crit Rev Toxicol 32:233–257
Giannakoudakis DA, Kyzas GZ, Avranas A, Lazaridis NK (2016) Multi-parametric adsorption effects of the reactive dye removal with commercial activated carbons. J Mol Liq 213:381–389
Grossmann K (2010) Auxin herbicides: current status of mechanism and mode of action. Pest Manag Sci: Formerly Pest Sci 66:113–120
Grover R, Franklin C, Muir N, Cessna A, Riedel D (1986) Dermal exposure and urinary metabolite excretion in farmers repeatedly exposed to 2, 4-D amine. Toxicol Lett 33:73–83
Hand LC, Vance JC, Randell TM, Shugart J, Gray T, Luo X, Culpepper AS (2021) Effects of low-dose applications of 2, 4-D and dicamba on cucumber and cantaloupe. Weed Technol 35:357–362
Hussami L (2010) Synthesis, characterization and application of multiscale porous materials, KTH. Retrieved from https://urn.kb.se/resolve?urn=urn:nbn:se:kth:diva-27158
Igberase E, Osifo P (2019) Mathematical modelling and simulation of packed bed column for the efficient adsorption of Cu (II) ions using modified bio-polymeric material. J Environ Chem Eng 7:103129
Joseph C, Elilarasi L (2017) Removal of methylene blue dye from aqueous solution using a newly synthesized TiO2-SiO2 photocatalyst in the presence of active chlorine species. IOP Conf Ser: Mater Sci Eng. IOP Publishing 206:012090. https://doi.org/10.1088/1757-899X/206/1/012090
Knopp D, Glass S (1991) Biological monitoring of 2, 4-dichlorophenoxyacetic acid-exposed workers in agriculture and forestry. Int Arch Occup Environ Health 63:329–333
Kreno LE, Leong K, Farha OK, Allendorf M, Van Duyne RP, Hupp JT (2012) Metal–organic framework materials as chemical sensors. Chem Rev 112:1105–1125
Lee J-SM, Otake K-i, Kitagawa S (2020) Transport properties in porous coordination polymers. Coord Chem Rev 421:213447
Li J, Li X, Wang L, Li Y, Wang K (2019) Prediction of PM2. 5 concentration based on PSO-LSSVR. Published In 2019 IEEE 8th Data Driven Control and Learning Systems Conference (DDCLS). IEEE, pp 723–727. https://doi.org/10.1109/DDCLS.2019.8909007
Lu P, Wu Y, Kang H, Wei H, Liu H, Fang M (2014) What can p K a and NBO charges of the ligands tell us about the water and thermal stability of metal organic frameworks? J Mater Chem A 2:16250–16267
Mansab S, Rafique U (2021) In situ remediation of 2, 4-dicholrophenoxyacetic acid herbicide using amine-functionalized imidazole coordination complexes. Environ Sci Pollut Res 28:15099–15113
Mazloom MS, Rezaei F, Hemmati-Sarapardeh A, Husein MM, Zendehboudi S, Bemani A (2020) Artificial intelligence based methods for asphaltenes adsorption by nanocomposites: application of group method of data handling, least squares support vector machine, and artificial neural networks. Nanomaterials 10:890
Michałowicz J, Duda W (2007) Phenols--Sources and Toxicity. Pol J Environ Stud 16(3):347–362
Mohan B, Jana A, Das N, Bharti S, Choudhary M, Muhammad S, Kumar S, Al-Sehemi AG, Algarni H (2019) A dual approach to study the key features of nickel (II) and copper (II) coordination complexes: synthesis, crystal structure, optical and nonlinear properties. Inorg Chim Acta 484:148–159
Pan B, Pan B, Zhang W, Lv L, Zhang Q, Zheng S (2009) Development of polymeric and polymer-based hybrid adsorbents for pollutants removal from waters. Chem Eng J 151:19–29
Qurratu A, Reehan A (2016) A review of 2, 4-Dichlorophenoxyacetic acid (2, 4-D) derivatives: 2, 4-D dimethylamine salt and 2, 4-D butyl ester. Int J Appl Eng Res 11:9946–9955
Rasheed T, Rizwan K, Bilal M, Iqbal HM (2020) Metal-organic framework-based engineered materials—fundamentals and applications. Molecules 25:1598
Rowsell JL, Yaghi OM (2004) Metal–organic frameworks: a new class of porous materials. Microporous Mesoporous Mater 73:3–14
Roy DK, Lal A, Sarker KK, Saha KK, Datta B (2021) Optimization algorithms as training approaches for prediction of reference evapotranspiration using adaptive neuro fuzzy inference system. Agric Water Manag 255:107003
Salehi MB, Moghadam AM, Panahi R (2021) Chapter 11 - Sorbent hydrogels to control heavy metal pollution in water. In: Núñez-Delgado A (ed) Sorbents materials for controlling environmental pollution. Elsevier, pp 247–283
Sarma P, Gomila RM, Frontera A, Barcelo-Oliver M, Bhattacharyya MK (2023) Cooperative ternary assemblies involving anion–π/π–π/anion–π assemblies and unconventional Cl⋯ Cl interactions in Cu (II) coordination compounds: experimental and theoretical studies. Crystals 13:517
Shikuku VO, Jemutai-Kimosop S (2020) Efficient removal of sulfamethoxazole onto sugarcane bagasse-derived biochar: two and three-parameter isotherms, kinetics and thermodynamics. South Afr J Chem 73:111–119. https://doi.org/10.17159/0379-4350/2020/v73a16
Song JF, Zhou RS, Zhang J, Xu CY, Li YB, Wang BB (2011) Three new cpt‐metal complexes displaying 0D, 1D, and 3D topology structures. Zeitschrift für anorganische und allgemeine Chemie 637:589–595
Steinbacher JL, Landry CC (2014) Adsorption and release of siRNA from porous silica. Langmuir 30:4396–4405
Storck S, Bretinger H, Maier WF (1998) Characterization of micro-and mesoporous solids by physisorption methods and pore-size analysis. Appl Catal A 174:137–146
Subramanyam B, Ashutosh D (2012) Adsorption isotherm modeling of phenol onto natural soils–applicability of various isotherm models. Int J Environ Res Public Health 6:265–276
Vinayagam R, Pai S, Murugesan G, Varadavenkatesan T, Narayanasamy S, Selvaraj R (2022) Magnetic activated charcoal/Fe2O3 nanocomposite for the adsorptive removal of 2, 4-dichlorophenoxyacetic acid (2, 4-D) from aqueous solutions: synthesis, characterization, optimization, kinetic and isotherm studies. Chemosphere 286:131938
Yettou A, Laidi M, El Bey A, Hanini S, Hentabli M, Khaldi O, Abderrahim M (2021) Ternary multicomponent adsorption modelling using ANN, LS-SVR, and SVR approach–case study. Kemija u Industriji: Časopis Kemičara i Kemijskih Inženjera Hrvatske 70:509–518
Zango ZU, Sambudi NS, Jumbri K, Ramli A, Abu Bakar NHH, Saad B, Rozaini MNH, Isiyaka HA, Osman AM, Sulieman A (2020) An overview and evaluation of highly porous adsorbent materials for polycyclic aromatic hydrocarbons and phenols removal from wastewater. Water 12:2921
Zhou L (2019) Prediction of CO2 adsorption on different activated carbons by hybrid group method of data-handling networks and LSSVM. Energy Sources A: Recovery Util Environ Eff 41:1960–1971
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Saira Mansab and Uzaira Rafique The first draft of the manuscript was written by Saira Mansab and Uzaira Rafique commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mansab, S., Rafique, U. Adsorption simulation of 2,4-D pesticide on novel zinc–based 2-amino-4-(1H-1,2,4-triazole-4-yl)benzoic acid coordination complexes using machine learning approach. Environ Sci Pollut Res (2024). https://doi.org/10.1007/s11356-024-33668-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-024-33668-1