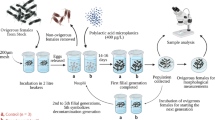
Abstract
Tributyltin (TBT) is widely used in various commercial applications due to its biocidal properties. Toxicological and genotoxicological data on TBT exposure to amphibians is insufficient. Our study aimed to determine the acute toxicity and genotoxic potential of TBT in Fejervarya limnocharis tadpoles. Furthermore, oxidative stress was also investigated in TBT-treated tadpoles. Tadpoles of Gosner stage (26–30) were screened and subjected to increasing concentrations of TBT (0, 3, 7, 11, 15, 19, 23 µg/L) for determining the LC50 values for 24 h, 48 h, 72 h, and 96 h. LC50 values of TBT for 24 h, 48 h, 72 h, and 96 h were found to be 19.45, 15.07, 13.12, and 11.84 μg/L respectively. Based on the 96 h LC50 value (11.84 µg/L), tadpoles were exposed to different sub-lethal concentrations of TBT for the evaluation of its genotoxic potential and effects on oxidative balance. The role of TBT on survivability, growth, and time to metamorphosis was also assessed. TBT exposure significantly altered the life history traits measured, increased mortality, and delayed the time taken to metamorphosis. Results indicated significant induction of micronucleus (MN, p < 0.001) and other erythrocytic nuclear aberrations (ENA, p < 0.01) in the TBT-treated groups. Significant alterations in comet parameters and oxidative balance were also observed in the treated groups. The present study findings might add to the cause of the gradual population decline seen in the amphibians. This study also demonstrates the alteration of the life-history traits, oxidative balance, and DNA damage upon TBT exposure which can have long-term consequences for the anuran amphibian F. limnocharis.





Similar content being viewed by others
Data Availability
Data will be made available on reasonable request.
References
Annamalai SK, Arunachalam KD (2017) Uranium (238U)-induced ROS and cell cycle perturbations, antioxidant responses and erythrocyte nuclear abnormalities in the freshwater iridescent shark fish Pangasius sutchi. Aquat Toxicol 186:145–158. https://doi.org/10.1016/j.aquatox.2017.03.002
Antizar-Ladislao B (2008) Environmental levels, toxicity and human exposure to tributyltin (TBT)-contaminated marine environment. A review. Environ Int 34:292–308. https://doi.org/10.1016/j.envint.2007.09.005
Bhuyan K, Patar A, Singha U, Giri S, Giri A (2020) Phenanthrene alters oxidative stress parameters in tadpoles of Euphlyctis cyanophlyctis (Anura, Dicroglossidae) and induces genotoxicity assessed by micronucleus and comet assay. Environ Sci Pollut Res 27:20962–20971. https://doi.org/10.1007/s11356-020-08609-3
Braham RP, Blazer VS, Shaw CH, Mazik PM (2017) Micronuclei and other erythrocyte nuclear abnormalities in fishes from the Great Lakes Basin, USA. Environ Mol Mutagen 58:570–581. https://doi.org/10.1002/em.22123
Brodeur J, Candioti JV (2017) Impacts of agriculture and pesticides on amphibian terrestrial life stages: Potential biomonitor/bioindicator species for the Pampa region of Argentina, Ecotoxicology and Genotoxicology, pp 163–194. https://doi.org/10.1039/9781788010573-00163
Bryan GW, Gibbs PE (2020) Impact of low concentrations of tributyltin (TBT) on marine organisms: a review. In: Metal Ecotoxicology Concepts and Applications. CRC Press, pp 323–361
Carvalho PN, Basto M, Silva MF, Machado A, Bordalo A, Vasconcelos M (2010) Ability of salt marsh plants for TBT remediation in sediments. Environ Sci Pollut Res 17:1279–1286. https://doi.org/10.1007/s11356-010-0307-1
Chen M, Guo J, Ruan J, Yang Z, He C, Zuo Z (2020) Neonatal exposure to environment-relevant levels of tributyltin leads to uterine dysplasia in rats. Sci Total Environ 720:137615. https://doi.org/10.1016/j.scitotenv.2020.137615
Collins A, Møller P, Gajski G, Vodenková S, Abdulwahed A, Anderson D, Bankoglu EE, Bonassi S, Boutet-Robinet E, Brunborg G (2023) Measuring DNA modifications with the comet assay: A compendium of protocols. Nat Protoc 18:929–989. https://doi.org/10.1038/s41596-022-00754-y
do Amaral DF, Guerra V, Motta AGC, e Silva DdM, Rocha TL (2019) Ecotoxicity of nanomaterials in amphibians: a critical review. Sci Total Environ 686:332–344. https://doi.org/10.1016/j.scitotenv.2019.05.487
Ferraro MVM, Fenocchio AS, Mantovani MS, Ribeiro CdO, Cestari MM (2004) Mutagenic effects of tributyltin and inorganic lead (Pb II) on the fish H. malabaricus as evaluated using the comet assay and the piscine micronucleus and chromosome aberration tests. Genet Mol Biol 27:103–107. https://doi.org/10.1590/S1415-47572004000100017
Ferreira M, Blanco L, Garrido A, Vieites JM, Cabado AG (2013) In vitro approaches to evaluate toxicity induced by organotin compounds tributyltin (TBT), dibutyltin (DBT), and monobutyltin (MBT) in neuroblastoma cells. J Agric Food Chem 61:4195–4203. https://doi.org/10.1021/jf3050186
Fort DJ, Degitz S, Tietge J, Touart LW (2007) The hypothalamic-pituitary-thyroid (HPT) axis in frogs and its role in frog development and reproduction. Crit Rev Toxicol 37:117–161. https://doi.org/10.1080/10408440601123545
Gabbianelli R, Moretti M, Carpenè E, Falcioni G (2006) Effect of different organotins on DNA of mollusk (Scapharca inaequivalvis) erythrocytes assessed by the comet assay. Sci Total Environ 367:163–169. https://doi.org/10.1016/j.scitotenv.2006.02.021
Gaweł S, Wardas M, Niedworok E, Wardas P (2004) Malondialdehyde (MDA) as a lipid peroxidation marker. Wiadomosci lekarskie (Warsaw, Poland: 1960) 57:453–455
Giri A, Giri S (2019) Micronucleus assays in amphibians, The Micronucleus Assay in Toxicology, pp 259–272. https://doi.org/10.1039/9781788013604-00259
Gosner KL (1960) A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16:183–190
Guo S, Qian L, Shi H, Barry T, Cao Q, Liu J (2010) Effects of tributyltin (TBT) on Xenopus tropicalis embryos at environmentally relevant concentrations. Chemosphere 79:529–533. https://doi.org/10.1016/j.chemosphere.2010.02.021
Hagger JA, Depledge MH, Galloway TS (2005) Toxicity of tributyltin in the marine mollusc Mytilus edulis. Mar Pollut Bull 51:811–816. https://doi.org/10.1016/j.marpolbul.2005.06.044
Herek JS, Vargas L, Trindade SAR, Rutkoski CF, Macagnan N, Hartmann PA, Hartmann MT (2021) Genotoxic effects of glyphosate on Physalaemus tadpoles. Environ Toxicol Pharmacol 81:103516. https://doi.org/10.1016/j.etap.2020.103516
Hopkins WA (2007) Amphibians as models for studying environmental change. ILAR J 48:270–277. https://doi.org/10.1093/ilar.48.3.270
Janesick A, Blumberg B (2012) Obesogens, stem cells and the developmental programming of obesity. Int J Andrology 35:437–448. https://doi.org/10.1111/j.1365-2605.2012.01247.x
Jha AN (2008) Ecotoxicological applications and significance of the comet assay. Mutagenesis 23:207–221. https://doi.org/10.1093/mutage/gen014
Jha AN, Hagger JA, Hill SJ (2000) Tributyltin induces cytogenetic damage in the early life stages of the marine mussel, Mytilus edulis. Environ Mol Mutagen 35:343–350. https://doi.org/10.1002/1098-2280(2000)35:4%3c343::AID-EM9%3e3.0.CO;2-5
Khondee P, Srisomsap C, Chokchaichamnankit D, Svasti J, Simpson RJ, Kingtong S (2016) Histopathological effect and stress response of mantle proteome following TBT exposure in the Hooded oyster Saccostrea cucullata. Environ Pollut 218:855–862. https://doi.org/10.1016/j.envpol.2016.08.011
Lam NH, Jeong H-h, Kang S-d, Kim D-J, Ju M-J, Horiguchi T, Cho H-S (2017) Organotins and new antifouling biocides in water and sediments from three Korean Special Management Sea Areas following ten years of tributyltin regulation: Contamination profiles and risk assessment. Mar Pollut Bull 121:302–312. https://doi.org/10.1016/j.marpolbul.2017.06.026
Laughlin R, Lindén O (1982) Sublethal responses of the tadpoles of the european frogRana temporaria to two tributyltin compounds. Bull Environ Contam Toxicol 28:494–499. https://doi.org/10.1007/BF01607718
Li B, Sun L, Cai J, Wang C, Wang M, Qiu H, Zuo Z (2015) Modulation of the DNA repair system and ATR-p53 mediated apoptosis is relevant for tributyltin-induced genotoxic effects in human hepatoma G2 cells. J Environ Sci 27:108–114. https://doi.org/10.1016/j.jes.2014.05.032
Liang X, Souders CL II, Zhang J, Martyniuk CJ (2017) Tributyltin induces premature hatching and reduces locomotor activity in zebrafish (Danio rerio) embryos/larvae at environmentally relevant levels. Chemosphere 189:498–506. https://doi.org/10.1016/j.chemosphere.2017.09.093
Liu HG, Wang Y, Lian L, Xu LH (2006) Tributyltin induces DNA damage as well as oxidative damage in rats. Environ Toxicol: An Int J 21:166–171. https://doi.org/10.1002/tox.20170
Liu W-Y, Wang C-Y, Wang T-S, Fellers GM, Lai B-C, Kam Y-C (2011) Impacts of the herbicide butachlor on the larvae of a paddy field breeding frog (Fejervarya limnocharis) in subtropical Taiwan. Ecotoxicology 20:377–384. https://doi.org/10.1007/s10646-010-0589-6
Lushchak VI (2011) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101:13–30. https://doi.org/10.1016/j.aquatox.2010.10.006
Magara G, Elia AC, Dörr AJM, Abete MC, Brizio P, Caldaroni B, Righetti M, Pastorino P, Scoparo M, Prearo M (2021) Metal load and oxidative stress driven by organotin compounds on rainbow trout. Environ Sci Pollut Res 28:35012–35022. https://doi.org/10.1007/s11356-021-12984-w
Mandal A, Ghosh M, Talukdar D, Dey P, Das A, Giri S (2023) Cytotoxicity and genotoxicity of tributyltin in the early embryonic chick, Gallus gallus domesticus. Mut Res/Genet Toxicol Environ Mutagenesis 503656. https://doi.org/10.1016/j.mrgentox.2023.503656
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474
Martínez-Paz P, Morales M, Martínez-Guitarte JL, Morcillo G (2013) Genotoxic effects of environmental endocrine disruptors on the aquatic insect Chironomus riparius evaluated using the comet assay. Mutat Res/Genet Toxicol Environ Mutagenesis 758:41–47. https://doi.org/10.1016/j.mrgentox.2013.09.005
Meador JP (1997) Comparative toxicokinetics of tributyltin in five marine species and its utility in predicting bioaccumulation and acute toxicity. Aquat Toxicol 37:307–326. https://doi.org/10.1016/S0166-445X(96)00827-2
Meador JP, Sommers FC, Cooper KA, Yanagida G (2011) Tributyltin and the obesogen metabolic syndrome in a salmonid. Environ Res 111:50–56. https://doi.org/10.1016/j.envres.2010.11.012
Mengeling BJ, Murk AJ, Furlow JD (2016) Trialkyltin rexinoid-X receptor agonists selectively potentiate thyroid hormone induced programs of Xenopus laevis metamorphosis. Endocrinology 157:2712–2723. https://doi.org/10.1210/en.2016-1062
Micael J, Reis-Henriques M, Carvalho A, Santos M (2007) Genotoxic effects of binary mixtures of xenoandrogens (tributyltin, triphenyltin) and a xenoestrogen (ethinylestradiol) in a partial life-cycle test with Zebrafish (Danio rerio). Environ Int 33:1035–1039. https://doi.org/10.1016/j.envint.2007.06.004
Morales M, Martínez-Paz P, Ozáez I, Martínez-Guitarte JL, Morcillo G (2013) DNA damage and transcriptional changes induced by tributyltin (TBT) after short in vivo exposures of Chironomus riparius (Diptera) larvae. Comp Biochem Physiol c: Toxicol Pharmacol 158:57–63. https://doi.org/10.1016/j.cbpc.2013.05.005
Niki E (2008) Lipid peroxidation products as oxidative stress biomarkers. BioFactors 34:171–180
Patar A, Giri A, Boro F, Bhuyan K, Singha U, Giri S (2016) Cadmium pollution and amphibians–Studies in tadpoles of Rana limnocharis. Chemosphere 144:1043–1049. https://doi.org/10.1016/j.chemosphere.2015.09.088
Rani KU, Musthafa MS, War M, Al-Sadoon MK, Paray BA, Shareef T, Nawas P (2015) Impact of tributyltin on antioxidant and DNA damage response in spermatozoa of freshwater prawn Macrobrachium rosenbergii. Environ Sci Pollut Res 22:20000–20006. https://doi.org/10.1007/s11356-015-5202-3
Rehman S-U (1984) Lead induced regional lipid peroxidation in brain. Toxicol Lett 21:333–337
Rouse JD, Bishop CA, Struger J (1999) Nitrogen pollution: an assessment of its threat to amphibian survival. Environ Health Perspect 107:799–803. https://doi.org/10.1289/ehp.99107799
Samojeden CG, Pavan FA, Rutkoski CF, Folador A, da Fré SP, Müller C, Hartmann PA, Hartmann M (2022) Toxicity and genotoxicity of imidacloprid in the tadpoles of Leptodactylus luctator and Physalaemus cuvieri (Anura: Leptodactylidae). Sci Rep 12:11926. https://doi.org/10.1038/s41598-022-16039-z
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205
Sharan S, Nikhil K, Roy P (2014) Disruption of thyroid hormone functions by low dose exposure of tributyltin: an in vitro and in vivo approach. Gen Comp Endocrinol 206:155–165. https://doi.org/10.1016/j.ygcen.2014.07.027
Shi H, Zhu P, Guo S (2014) Effects of tributyltin on metamorphosis and gonadal differentiation of Xenopus laevis at environmentally relevant concentrations. Toxicol Ind Health 30:297–303. https://doi.org/10.1177/0748233712457440
Shi Y, Chen C, Li M, Liu L, Dong K, Chen K, Qiu X (2021) Oral exposure to tributyltin induced behavioral abnormality and oxidative stress in the eyes and brains of juvenile Japanese medaka (Oryzias latipes). Antioxidants 10:1647. https://doi.org/10.3390/antiox10111647
Singha U, Pandey N, Boro F, Giri S, Giri A, Biswas S (2014) Sodium arsenite induced changes in survival, growth, metamorphosis and genotoxicity in the Indian cricket frog (Rana limnocharis). Chemosphere 112:333–339. https://doi.org/10.1016/j.chemosphere.2014.04.076
Tang L, Zhang Y-H, Wang X, Zhang C-C, Qin G, Lin Q (2021) Effects of chronic exposure to environmental levels of tributyltin on the lined seahorse (Hippocampus erectus) liver: Analysis of bioaccumulation, antioxidant defense, and immune gene expression. Sci Total Environ 801:149646. https://doi.org/10.1016/j.scitotenv.2021.149646
Tiano L, Fedeli D, Moretti M, Falcioni G (2001) DNA damage induced by organotins on trout-nucleated erythrocytes. Appl Organomet Chem 15:575–580. https://doi.org/10.1002/aoc.207
Turkez H, Arslan ME, Ozdemir O (2017) Genotoxicity testing: progress and prospects for the next decade. Expert Opin Drug Metab Toxicol 13:1089–1098. https://doi.org/10.1080/17425255.2017.1375097
US EPA (2003) Ambient aquatic life water quality criteria for Tributyltin (TBT)—Final. United States Environmental Protection Agency, Office of Water, EPA 822-R-03–031, Springfield, VA. http://www.epa.gov. Accessed 26 Nov 2023
Wallis DJ, Truong L, La Du J, Tanguay RL, Reif DM (2021) Uncovering evidence for endocrine-disrupting chemicals that elicit differential susceptibility through gene-environment interactions. Toxics 9:77. https://doi.org/10.3390/toxics9040077
Zhang J, Sun P, Yang F, Kong T, Zhang R (2016) Tributyltin disrupts feeding and energy metabolism in the goldfish (Carassius auratus). Chemosphere 152:221–228. https://doi.org/10.1016/j.chemosphere.2016.02.127
Zoll-Moreux C, Ferrier V (1999) The Jaylet test (Newt micronucleus test) and the micronucleus test in Xenopus: two in vivo tests on Amphibia evaluation of the genotoxicity of five environmental pollutants and of five effluents. Water Res 33:2301–2314. https://doi.org/10.1016/S0043-1354(98)00464-3
Acknowledgements
Abhijit Mandal sincerely acknowledges the Council of Scientific and Industrial Research (CSIR), Govt. of India for Senior Research Fellowship (Sanction letter no. 09/747(0014)/2018-EMR-I).
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Abhijit Mandal: Conceptualization, Methodology, Investigation, Data analysis, Original draft preparation; Sarbani Giri: Conceptualization, Methodology, Writing and reviewing the manuscript, Overall supervision and Anirudha Giri: Methodology, Review and editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Larval rearing and subsequent experiments were done following the institutional ethical guidelines for laboratory animal care and getting approval of the Assam University institutional ethical committee bearing approval number AUS/IAEC/AM-R(e)/2023-1/001.
Consent to participate
Not applicable.
Consent to publish
All authors have approved the final version of the manuscript to be published.
Competing interests
The authors declare that there is no conflict of interest.
Additional information
Responsible Editor: Cinta Porte
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mandal, A., Giri, S. & Giri, A. Assessment of toxicity, genotoxicity and oxidative stress in Fejervarya limnocharis exposed to tributyltin. Environ Sci Pollut Res 31, 14938–14948 (2024). https://doi.org/10.1007/s11356-024-32220-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-32220-5