Abstract
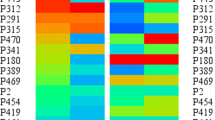
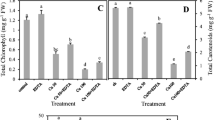
Phytoremediation is a widely used and cost-effective technique for in situ remediation of heavy metals. Brassica napus L. genotype with high Cd accumulation and strong Cd tolerance is an ideal candidate for phytoremediation. In this study, a hydroponic experiment was conducted to select a Brassica napus genotype with either high or low Cd accumulation from a panel of 55 genotypes. The physiological mechanisms governing Cd accumulation and Cd tolerance were then explored. BN400 and BN147 were identified as the high and low Cd accumulating genotypes, respectively. Additionally, BN400 exhibited greater tolerance to Cd stress compared to BN147. Root morphology analysis revealed that BN400 exhibited longer root length, smaller root surface area and root volume, and less root tips but bigger root diameter than BN147. Subcellular Cd distribution showed that the Cd concentrations in the cell wall and vacuole in shoot were significantly higher in BN400 than in BN147, whereas the opposite trend was observed in the roots.. Pectate/protein-integrated Cd was found to be the predominant form of Cd in both shoots and roots, with significantly higher levels in BN400 compared to BN147 in the shoot, but the opposite trend was observed in the roots. These results suggest that the long fine roots play a role in Cd accumulation. The high Cd accumulating genotype was able to retain Cd in leaf cell walls and vacuoles, and Cd was mainly present in the form of pectate/protein-integrated Cd, which contributes to its strong Cd tolerance. These findings have important implications for the screening and breeding of Brassica napus genotypes with high Cd accumulation for phytoremediation purposes.







Similar content being viewed by others
References
Arao T, Ae N (2003) Genotypic variations in Cd levels of rice grain. Soil Sci Plant Nutri 49:473–479. https://doi.org/10.1080/00380768.2003.10410035
Bian JL, Guo JM, Wang XD, Yang JX, Yang J, Chen TB, Cao L, Cheng YX, Ren ZH, Wang J, Zhou XY (2020) Tolerance mechanism and Cd enrichment abilities in two B. napus L. cultivars. Huan Jing Ke Xue 41(2):970–978. https://doi.org/10.13227/j.hjkx.201906175
Chen Y, Xie T, Liang Q, Liu M, Zhao M, Wang M, Wang G (2016) Effectiveness of lime and peat applications on Cd availability in a paddy soil under various moisture regimes. Environ Sci Pollut Res Int 23(8):7757–7766. https://doi.org/10.1007/s11356-015-5930-4
Gao MY, Chen XW, Huang WX, Wu L, Yu ZS, Xiang L, Mo CH, Li YW, Cai QY, Wong MH, Li H (2021) Cell wall modification induced by an arbuscular mycorrhizal fungus enhanced cadmium fixation in rice root. J Hazard Mater 416:125894. https://doi.org/10.1016/j.jhazmat.2021.125894
Gill SS, Khan NA, Tuteja N (2011) Differential Cd stress tolerance in five indian mustard (Brassica juncea L.) cultivars: an evaluation of the role of antioxidant machinery. Plant Signal Behav 6(2):293–300. https://doi.org/10.4161/psb.6.2.15049
Gong Y, Zhao D, Wang Q (2018) An overview of field-scale studies on remediation of soil contaminated with heavy metals and metalloids: Technical progress over the last decade. Water Res 147:440–460. https://doi.org/10.1016/j.watres.2018.10.024
Grant CA, Clarke JM, Duguid S, Chaney RL (2008) Selection and breeding of plant cultivars to minimize Cd accumulation. Sci Total Environ 390(2–3):301–310. https://doi.org/10.1016/j.scitotenv.2007.10.038
Gu JF, Zhou H, Tang HL, Yang WT, Zeng M, Liu ZM, Peng PQ, Liao BH (2019) Cd and arsenic accumulation during the rice growth period under in situ remediation. Ecotoxicol Environ Saf 171:451–459. https://doi.org/10.1016/j.ecoenv.2019.01.003
Hu Y, Cheng H, Tao S (2016) The Challenges and Solutions for Cadmium-contaminated Rice in China: A Critical Review. Environ Int 92–93:515–532. https://doi.org/10.1016/j.envint.2016.04.042
Huang B, Xin J, Dai H, Liu A, Zhou W, Yi Y, Liao K (2015) Root morphological responses of three hot pepper cultivars to Cd exposure and their correlations with Cd accumulation. Environ Sci Pollut Res Int 22(2):1151–1159. https://doi.org/10.1007/s11356-014-3405-7
Huang B, Dai H, Zhou W, Peng L, Li M, Wan R, He W (2019) Characteristics of Cd accumulation and distribution in two sweet potato cultivars. Int J Phytoremediation 21(4):391–398. https://doi.org/10.1080/15226514.2018.1524846
Ishimaru Y, Takahashi R, Bashir K, Shimo H, Senoura T, Sugimoto K, Ono K, Yano M, Ishikawa S, Arao T, Nakanishi H, Nishizawa NK (2012) Characterizing the role of rice NRAMP5 in Manganese. Iron Cadmium Transp Sci Rep 2:286. https://doi.org/10.1038/srep00286
Kaur R, Yadav P, Thukral AK, Walia A, Bhardwaj R (2017) Co-application of 6-ketone type brassinosteroid and metal chelator alleviates Cd toxicity in B. juncea L. Environ Sci Pollut Res Int 24(1):685–700. https://doi.org/10.1007/s11356-016-7864-x
Khaokaew S, Chaney RL, Landrot G, Ginder-Vogel M, Sparks DL (2011) Speciation and release kinetics of Cd in an alkaline paddy soil under various flooding periods and draining conditions. Environ Sci Technol 45(10):4249–4255. https://doi.org/10.1021/es103971y
Kniuipytė I, Dikšaitytė A, Praspaliauskas M, Pedišius N, Žaltauskaitė J (2023) Oilseed rape (Brassica napus L.) potential to remediate Cd contaminated soil under different soil water content. J Environ Manag 325(Pt A):116627. https://doi.org/10.1016/j.jenvman.2022.116627
Kubo K, Watanabe Y, Matsunaka H, Seki M, Fujita M, Kawada N, Hatta K, Nakajima T (2011) Differences in Cd accumulation and root morphology in seedlings of Japanese wheat varieties with distinctive grain Cd concentration. Plant Prod Sci 14:148–155. https://doi.org/10.1626/pps.14.148
Lan JX, Chen XS (2022) Effect of “VIP + n” technology on cadmium reduction of paddy soil and rice. Hunan Agric Sci 2022(06):40–46. https://doi.org/10.16498/j.cnki.hnnykx.2022.006.010
Li T, Yang X, Lu L, Islam E, He Z (2009) Effects of zinc and Cd interactions on root morphology and metal translocation in a hyperaccumulating species under hydroponic conditions. J Hazard Mater 169(1–3):734–741. https://doi.org/10.1016/j.jhazmat.2009.04.004
Li M, Xi X, Xiao G, Cheng H, Yang Z, Zhou G, Ye J, Li Z (2014a) National multi-purpose regional geochemical survey in China. J Geochem Explor 139:21–30. https://doi.org/10.1016/j.gexplo.2013.06.002
Li W, Xu B, Song Q, Liu X, Xu J, Brookes PC (2014b) The identification of “hotspots” of heavy metal pollution in soil-rice systems at a regional scale in eastern China. Sci Total Environ 472:407–420. https://doi.org/10.1016/j.scitotenv.2013.11.046
Li S, Wang F, Ru M, Ni W (2014c) Cd tolerance and accumulation of Elsholtzia argyi origining from a zinc/lead mining site - a hydroponics experiment. Int J Phytoremediation 16(7–12):1257–1267. https://doi.org/10.1080/15226514.2013.828010
Li K, Yu H, Li T, Chen G, Huang F (2017) Cadmium accumulation characteristics of low-cadmium rice (Oryza sativa L.) line and F1 hybrids grown in cadmium-contaminated soils. Environ Sci Pollut Res Int 24(21):17566–17576. https://doi.org/10.1007/s11356-017-9350-5
Li X, Ma H, Li L, Gao Y, Li Y, Xu H (2019) Subcellular distribution, chemical forms and physiological responses involved in cadmium tolerance and detoxification in Agrocybe Aegerita. Ecotoxicol Environ Saf 171:66–74. https://doi.org/10.1016/j.ecoenv.2018.12.063
Liao Q, Jian SF, Song HX, Guan CY, Lepo JE, Ismail AM, Zhang ZH (2019) Balance between nitrogen use efficiency and Cd tolerance in B. napus and Arabidopsis thaliana. Plant Sci 284:57–66. https://doi.org/10.1016/j.plantsci.2019.04.003
Liu Y, Hu C, Li B, Ding D, Zhao Z, Fan T, Li Z (2021) Subsurface drip irrigation reduces Cd accumulation of pepper (Capsicum annuum L.) plants in upland soil. Sci Total Environ 755(Pt 2):142650. https://doi.org/10.1016/j.scitotenv.2020.142650
Loix C, Huybrechts M, Vangronsveld J, Gielen M, Keunen E, Cuypers A (2017) Reciprocal Interactions between Cadmium-Induced Cell Wall Responses and Oxidative Stress in Plants. Front Plant Sci 8:1867. https://doi.org/10.3389/fpls.2017.01867
Lu Z, Zhang Z, Su Y, Liu C, Shi G (2013) Cultivar variation in morphological response of peanut roots to Cd stress and its relation to Cd accumulation. Ecotoxicol Environ Saf 91:147–155. https://doi.org/10.1016/j.ecoenv.2013.01.017
Ma Q, Cao X, Tan X, Si L, Wu L (2017) Effects of Cd stress on pakchoi (Brassica chinensis L.) growth and uptake of inorganic and organic nitrogenous compounds. Environ Exp Bot 137:49–57. https://doi.org/10.1016/j.envexpbot.2017.02.001
Martinoia E (2018) Vacuolar Transporters - Companions on a Longtime Journey. Plant Physiol 176(2):1384–1407. https://doi.org/10.1104/pp.17.01481
Meng JG, Zhang XD, Tan SK, Zhao KX, Yang ZM (2017) Genome-wide identification of Cd-responsive NRAMP transporter genes and analyzing expression of NRAMP 1 mediated by miR167 in Brassica napus. Biometals 30(6):917–931. https://doi.org/10.1007/s10534-017-0057-3. (Epub 2017 Oct 9)
Mwamba TM, Li L, Gill RA, Islam F, Nawaz A, Ali B, Farooq MA, Lwalaba JL, Zhou W (2016) Differential subcellular distribution and chemical forms of Cd and copper in B. napus. Ecotoxicol Environ Saf 134:239–249. https://doi.org/10.1016/j.ecoenv.2016.08.021
Pittman JK (2005) Managing the manganese: molecular mechanisms of manganese transport and homeostasis. New Phytol 167(3):733–742. https://doi.org/10.1111/j.1469-8137.2005.01453.x
Qiu Q, Wang Y, Yang Z, Yuan J (2011) Effects of phosphorus supplied in soil on subcellular distribution and chemical forms of cadmium in two Chinese flowering cabbage (Brassica parachinensis L.) cultivars differing in cadmium accumulation. Food Chem Toxicol 49(9):2260–2267. https://doi.org/10.1016/j.fct.2011.06.024
Rai PK (2018) Phytoremediation of Emerging Contaminants in Wetlands. CRC Press, Taylor & Francis, Boca Raton, p 248
Sauvé S, Hendershot W, Allen HE (2000) Solid-solution partitioning of metals in contaminated soils:dependence on pH, total metal burden, and organic matter. Environ Sci Technol 34:1125–1131. https://doi.org/10.1021/es9907764
Seregin IV, Kozhevnikova AD, Zhukovskaya NV, Schat H (2015) Cd tolerance and accumulation in Excluder Thlaspi arvense and various accessions of hyperaccumulator Noccaea caerulescens. Russ J Plant Physiol 62(6):837–846. https://doi.org/10.1134/S1021443715050131
Shahid M, Dumat C, Khalid S, Niazi NK, Antunes PMC (2017) Cd Bioavailability, Uptake, Toxicity and Detoxification in Soil-Plant System. Rev Environ Contam Toxicol 241:73–137. https://doi.org/10.1007/398_2016_8
Sharma SS, Dietz KJ, Mimura T (2016) Vacuolar compartmentalization as indispensable component of heavy metal detoxification in plants. Plant Cell Environ 39(5):1112–1126. https://doi.org/10.1111/pce.12706
Shen C, Fu HL, Liao Q, Huang BF, Huang YY, Xin JL (2021) Selection for low-cadmium cultivars and cadmium subcellular distribution comparison between two selected cultivars of eggplant (Solanum melongena L.). Environ Sci Pollut Res Int 28(41):57739–57750. https://doi.org/10.1007/s11356-021-14652-5
Shen X, Dai M, Yang J, Sun L, Tan X, Peng C, Ali I, Naz I (2022) A critical review on the phytoremediation of heavy metals from environment: Performance and challenges. Chemosphere 291(Pt 3):132979. https://doi.org/10.1016/j.chemosphere.2021.132979
Song Y, Jin L, Wang X (2017) Cadmium absorption and transportation pathways in plants. Int J Phytoremediation 19(2):133–141. https://doi.org/10.1080/15226514.2016.1207598
Song ML, Jie J, Yang L, Jun M, Shou LX, Yuan YT, You FL, Qing YS, Jian ZH (2019) Characterization andevaluation of OsLCT1 and OsNramp5 mutants generated through CRISPR/Cas9-mediated mutagenesis for breeding low Cd rice. Rice Sci 26:88–97. https://doi.org/10.1016/j.rsci.2019.01.002
Su DC, Huang HZ (2002) The phytoremediation potential of oilseed rape (B. juncea) as a hyperaccumulator for Cd contaminated soil. China Environ Sci 22(1):48–51. https://doi.org/10.3321/j.issn:1000-6923.2002.01.012
Takahashi R, Ishimaru Y, Shimo H, Ogo Y, Senoura T, Nishizawa NK, Nakanishi H (2012) The OsHMA2 transporter is involved in root-to-shoot translocation of Zn and Cd in rice. Plant Cell Environ 35(11):1948–1957. https://doi.org/10.1111/j.1365-3040.2012.02527.x
Tang JH (2018) Effect of VIP+n technology on cadmium reduction in paddy fields polluted by heavy metals. Hunan Agric Sci 9:48–50. https://doi.org/10.16498/j.cnki.hnnykx.2018.009.014
Tang XW (2020) Technology mode and benefit analysis of “VIP + n” comprehensive treatment on polluted rice fields with light acid and moderate cadmium. Anhui Agri Sci Bull 26(09):145–146. https://doi.org/10.16377/j.cnki.issn1007-7731.2020.09.052
Tóth G, Hermann T, Da Silva MR, Montanarella L (2016) Heavy metals in agricultural soils of the European Union with implications for food safety. Environ Int 88:299–309. https://doi.org/10.1016/j.envint.2015.12.017
Wang F, Wang M, Liu Z, Shi Y, Han T, Ye Y, Gong N, Sun J, Zhu C (2015) Different responses of low grain-Cd-accumulating and high grain-Cd-accumulating rice cultivars to Cd stress. Plant Physiol Biochem 96:261–269. https://doi.org/10.1016/j.plaphy.2015.08.001
Wei S, Wang S, Li Y, Zhu J (2013) Root system responses of hyperaccumulator Solanum nigrum L. to Cd. J Soil Sedimentol 13:1069–1074. https://doi.org/10.1007/s11368-013-0687-1
Weigel HJ, Jager HJ (1980) Subcellular distribution and chemical form of Cd in bean plants. Plant Physiol 65(3):480–482. https://doi.org/10.1104/pp.65.3.480
Weng BS, Xie XY, Weiss DJ, Liu JC, Lu HL, Yan CL (2012) Kandelia obovata (S., L.) Yong tolerance mechanisms to Cd: subcellular distribution, chemical forms and thiol pools. Mar Pollut Bull 64:2453–2460. https://doi.org/10.1016/j.marpolbul.2012.07.047
Wong CKE, Cobbett CS (2009) HMA P-type ATPases are the major mechanism for root-to-shoot Cd translocation in Arabidopsis thaliana. New Phytol 181(1):71–78. https://doi.org/10.1111/j.1469-8137.2008.02638.x
Wu X, Song H, Guan C, Zhang Z (2020a) Boron mitigates cadmium toxicity to rapeseed (Brassica napus) shoots by relieving oxidative stress and enhancing cadmium chelation onto cell walls. Environ Pollut 263(Pt B):114546. https://doi.org/10.1016/j.envpol.2020.114546
Wu X, Song H, Guan C, Zhang Z (2020b) Boron alleviates cadmium toxicity in Brassica napus by promoting the chelation of cadmium onto the root cell wall components. Sci Total Environ 728:138833. https://doi.org/10.1016/j.scitotenv.2020.138833
Wu X, Tian H, Li L, Guan C, Zhang Z (2021) Higher Cd-accumulating oilseed rape has stronger Cd tolerance due to stronger Cd fixation in pectin and hemicellulose and higher Cd chelation. Environ Pollut 285:117218. https://doi.org/10.1016/j.envpol.2021.117218
Xin J, Huang B (2014) Subcellular distribution and chemical forms of cadmium in two hot pepper cultivars differing in cadmium accumulation. J Agric Food Chem 62(2):508–515. https://doi.org/10.1021/jf4044524
Xu PX, Wang ZL (2013) Physiological mechanism of hypertolerance of cadmium in Kentucky bluegrass and tall fescue: chemical forms and tissue distribution. Environ Exp Bot 96:35–42. https://doi.org/10.1016/j.envexpbot.2013.09.001
Yu R, Xia S, Liu C, Zhang Z, Shi G (2017) Variations in root morphology among 18 herbaceous species and their relationship with Cd accumulation. Environ Sci Pollut Res Int 24(5):4731–4740. https://doi.org/10.1007/s11356-016-8210-z
Zhang ZH, Zhou T, Tang TJ, Song HX, Guan CY, Huang JY, Hua YP (2019) A multiomics approach reveals the pivotal role of subcellular reallocation in determining rapeseed resistance to cadmium toxicity. J Exp Bot 70(19):5437–5455. https://doi.org/10.1093/jxb/erz295
Zhang D, Zhou H, Shao L, Wang H, Zhang Y, Zhu T, Ma L, Ding Q, Ma L (2022) Root characteristics critical for Cd tolerance and reduced accumulation in wheat (Triticum aestivum L.). J Environ Manag 305:114365. https://doi.org/10.1016/j.jenvman
Zhao J, Yang W, Zhang S, Yang T, Liu Q, Dong J, Fu H, Mao X, Liu B (2018) Genome-wide association study and candidate gene analysis of rice Cd accumulation in grain in a diverse rice collection. Rice (n Y) 11(1):61. https://doi.org/10.1186/s12284-018-0254-x
Zhu WB, Qiu BS (2005) Effects of Cd hyperaccumulation on physiological characteristics of Sedum alfredii Hance (Crassulaceae). Plant Sci 169(4):737–745. https://doi.org/10.1016/j.plantsci.2005.05.030
Funding
This study was supported by the Natural Science Foundation of Hunan Province, China (grant no. 2022JJ40139).
Author information
Authors and Affiliations
Contributions
Conception and design of the study were accomplished by Junliang Xin, Qiong Liao, and Yingying Huang; data collection and analysis was performed by Qiong Liao, Huiling Fu, Chongyang Hu, Xiaokang Xiong, and Yuxi Huang; first draft was written by Qiong Liao and Chuang Shen. Data interpretation and manuscript polishing was carried out by Baifei Huang and Junliang Xin. All the authors approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Roberto Terzano
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liao, Q., Fu, H., Shen, C. et al. Physiological and biochemical characteristics of high and low Cd accumulating Brassica napus genotypes. Environ Sci Pollut Res 31, 11873–11885 (2024). https://doi.org/10.1007/s11356-024-31942-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-31942-w