Abstract
Aims
Cadmium (Cd) is the most widespread toxic heavy metal to plant growth. As the second leading oil crop, some genotypes of Brassica napus (B. napus) are potential Cd accumulators. However, the Cd translocation mechanism from root to shoot in B. napus in response to Cd toxicity remains unknown.
Methods
In the present study, a couple of B. napus genotypes with contrasting Cd uptake and root-to-shoot translocation abilities, named P78 (the high Cd accumulator, HC) and P72 (the low Cd accumulator, LC), were chosen from 39 B. napus genotypes with various Cd accumulation features.
Results
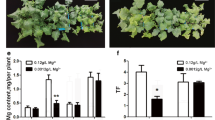
Physiological comparison of P78 and P72 reveals that P72 is more sensitive to Cd toxicity than P78. With genomic resequencing, transcriptomics and qRT-PCR assay, BnNramp2;1 and BnNramp4;2 were focused with highly upregulation in shoot of P78 under Cd treatment condition. Furthermore, BnNramp2;1 and BnNramp4;2 can successfully complement the function of tonoplast localized Cd transporter YCF1. And when BnNramp2;1 and BnNramp4;2 were transferred in Arabidopsis atnramp mutants, the transgenic plants showed better growth rate than mutants under higher Cd stress conditions.
Conclusions
The results reveals that BnNramp2;1 and BnNramp4;2 were two main Cd transporters associated with enhanced root-to-shoot translocation and accumulation of Cd in shoot of B. napus.








Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- AsA:
-
Ascorbic acid
- BCF:
-
Biological enrichment factor
- B.napus :
-
Brassica napus
- Ca:
-
Calcium
- CAT:
-
Catalase
- Cd:
-
Cadmium
- Ci:
-
Intercellular CO2 concentration
- Cu:
-
Copper
- DEGs:
-
Differentially expressed genes
- Fe:
-
iron
- GO:
-
Gene ontology
- GR:
-
Glutathione reductase
- Gs:
-
Stomatal conductance
- GSH:
-
Glutataione
- HC:
-
High Cd accumulator
- LC:
-
Low Cd accumulator
- MDA:
-
Malondialdehyde
- MDHAR:
-
Monodehydroascorbate reductase
- Mg:
-
Magnesium
- Mn:
-
Manganese
- Pn:
-
Variation of net CO2 assimilation
- POD:
-
Peroxidase
- R:
-
Root
- S:
-
Shoot
- SOD:
-
Superoxide dismutase
- TF:
-
Translocation factor
- Tr:
-
Transpiration
- Zn:
-
Zinc
References
Anjum NA, Ahmad I, Rodrigues SM, Henriques B, Cruz N, Coelho C, Pacheco M, Duarte AC, Pereira E (2013) Eriophorum angustifolium and Lolium perenne metabolic adaptations to metals- and metalloids-induced anomalies in the vicinity of a chemical industrial complex. Environ Sci Pollut Res Int 20(1):568–581
Cailliatte R, Lapeyre B, Briat JF, Mari S, Curie C (2009) The NRAMP6 metal transporter contributes to cadmium toxicity. Biochem J 422(2):217–228
Chen L, Wan H, Qian J, Guo J, Sun C, Wen J, Yi B, Ma C, Tu J, Song L, Fu T, Shen J (2018) Genome-wide association study of cadmium accumulation at the seedling stage in rapeseed (Brassica napus L.). Front Plant Sci 9:375
Choppala G, Saifullah BN, Bibi S, Iqbal M, Rengel Z, Kunhikrishnan A, Ashwath N, Ok YS (2014) Cellular mechanisms in higher plants governing tolerance to cadmium toxicity. Crit Rev Plant Sci 33(5):374–391
Curie C, Alonso JM, Jean ML, Ecker JR, Briat J-F (2000) Involvement of NRAMP1 from Arabidopsis thaliana in iron transport. Biochem J 347:749–755
D'Alessandro A, Taamalli M, Gevi F, Timperio AM, Zolla L, Ghnaya T (2013) Cadmium stress responses in Brassica juncea: hints from proteomics and metabolomics. J Proteome Res 12(11):4979–4997
Fan W, Liu C, Cao B, Qin M, Long D, Xiang Z, Zhao A (2018) Genome-wide identification and characterization of four gene families putatively involved in cadmium uptake, translocation and sequestration in mulberry. Front Plant Sci 9:879
Feng J, Jia W, Lv S, Bao H, Miao F, Zhang X, Wang J, Li J, Li D, Zhu C, Li S, Li Y (2018) Comparative transcriptome combined with morpho-physiological analyses revealed key factors for differential cadmium accumulation in two contrasting sweet sorghum genotypes. Plant Biotechnol J 16(2):558–571
Gao H, Xie W, Yang C, Xu J, Li J, Wang H, Chen X, Huang CF (2018) NRAMP2, a trans-Golgi network-localized manganese transporter, is required for Arabidopsis root growth under manganese deficiency. New Phytol 217(1):179–193
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48(12):909–930
Gravot A, Lieutaud A, Verret F, Auroy P, Vavasseur A, Richaud P (2004) AtHMA3, a plant P1B-ATPase, functions as a cd/Pb transporter in yeast. FEBS Lett 561(1–3):22–28
Gupta DK, Pena LB, Romero-Puertas MC, Hernandez A, Inouhe M, Sandalio LM (2017) NADPH oxidases differentially regulate ROS metabolism and nutrient uptake under cadmium toxicity. Plant Cell Environ 40(4):509–526
Hua Y, Zhou T, Ding G, Yang Q, Shi L, Xu F (2016) Physiological, genomic and transcriptional diversity in responses to boron deficiency in rapeseed genotypes. J Exp Bot 67(19):5769–5784
Ishimaru Y, Takahashi R, Bashir K, Shimo H, Senoura T, Sugimoto K, Ono K, Yano M, Ishikawa S, Arao T, Nakanishi H, Nishizawa NK (2012) Characterizing the role of rice NRAMP5 in manganese, Iron and cadmium transport. Sci Rep 2(1)
Jia W, Miao F, Lv S, Feng J, Zhou S, Zhang X, Wang D, Li S, Li Y (2017) Identification for the capability of cd-tolerance, accumulation and translocation of 96 sorghum genotypes. Ecotoxicol Environ Saf 145:391–397
Lanquar V, Lelièvre F, Barbier-Brygoo H, Thomine S (2004) Regulation and function of AtNRAMP4 metal transporter protein. Soil Sci Plant Nutr 50(7):1141–1150
Lanquar V, Leliévre F, Bolte S, Hamés C, Alcon C, Neumann D, Vansuyt G. 2005. Mobilization of vacuolar iron by AtNRAMP3 and AtNRAMP4. EMBO J 24: 4041–4051, 2005
Li B, Dewey CN (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinf 12(323)
Li ZS, Lu YP, Zhen RG, Szczypka M, Thiele DJ, Rea PA (1997) A new pathway for vacuolar cadmium sequestration in Saccharomyces cerevisiae: YCF1-catalyzed transport of bis(glutathionato)cadmium. Proc Natl Acad Sci 94(1):42–47
Li N, Xiao H, Sun J, Wang S, Wang J, Chang P, Zhou X, Lei B, Lu K, Luo F, Shi X, Li J (2018) Genome-wide analysis and expression profiling of the HMA gene family in Brassica napus under cd stress. Plant Soil 426(1–2):365–381
Lu G, Casaretto JA, Ying S, Mahmood K, Liu F, Bi YM, Rothstein SJ (2017) Overexpression of OsGATA12 regulates chlorophyll content, delays plant senescence and improves rice yield under high density planting. Plant Mol Biol 94(1–2):215–227
Luo JS, Huang J, Zeng DL, Peng JS, Zhang GB, Ma HL, Guan Y, Yi HY, Fu YL, Han B, Lin HX, Qian Q, Gong JM (2018) A defensin-like protein drives cadmium efflux and allocation in rice. Nat Commun 9(1):645
Martinka M, Vaculík M, Lux A (2014) Plant cell responses to cadmium and zinc. Appl Plant Cell Biol 22:209–246
Mills RF, Francini A, Ferreira da Rocha PS, Baccarini PJ, Aylett M, Krijger GC, Williams LE (2005) The plant P1B-type ATPase AtHMA4 transports Zn and cd and plays a role in detoxification of transition metals supplied at elevated levels. FEBS Lett 579(3):783–791
Miyadate H, Adachi S, Hiraizumi A, Tezuka K, Nakazawa N, Kawamoto T, Katou K, Kodama I, Sakurai K, Takahashi H, Satoh-Nagasawa N, Watanabe A, Fujimura T, Akagi H (2011) OsHMA3, a P1B-type of ATPase affects root-to-shoot cadmium translocation in rice by mediating efflux into vacuoles. New Phytol 189(1):190–199
Mwamba TM, Li L, Gill RA, Islam F, Nawaz A, Ali B, Farooq MA, Lwalaba JL, Zhou W (2016) Differential subcellular distribution and chemical forms of cadmium and copper in Brassica napus. Ecotoxicol Environ Saf 134P1:239–249
Nevo Y, Nelson N (2006) The NRAMP family of metal-ion transporters. Biochim Biophys Acta 1763(7):609–620
Okubo M, Yamada K, Hosoyamada M, Shibasaki T, Endou H (2003) Cadmium transport by human Nramp 2 expressed in Xenopus laevis oocytes. Toxicol Appl Pharmacol 187(3):162–167
Oomen RJ, Wu J, Lelievre F, Blanchet S, Richaud P, Barbier-Brygoo H, Aarts MG, Thomine S (2009) Functional characterization of NRAMP3 and NRAMP4 from the metal hyperaccumulator Thlaspi caerulescens. New Phytol 181(3):637–650
Pottier M, Oomen R, Picco C, Giraudat J, Scholz-Starke J, Richaud P, Carpaneto A, Thomine S (2015) Identification of mutations allowing natural resistance associated macrophage proteins (NRAMP) to discriminate against cadmium. Plant J 83(4):625–637
Sasaki A, Yamaji N, Yokosho K, Ma JF (2012) Nramp5 is a major transporter responsible for manganese and cadmium uptake in rice. Plant Cell 24(5):2155–2167
Seth CS, Remans T, Keunen E, Jozefczak M, Gielen H, Opdenakker K, Weyens N, Vangronsveld J, Cuypers A (2012) Phytoextraction of toxic metals: a central role for glutathione. Plant Cell Environ 35(2):334–346
Takahashi R, Ishimaru Y, Senoura T, Shimo H, Ishikawa S, Arao T, Nakanishi H, Nishizawa NK (2011) The OsNRAMP1 iron transporter is involved in cd accumulation in rice. J Exp Bot 62(14):4843–4850
Thomine S, vre FL, Debarbieux E, Schroeder JI, Barbier-Brygoo H (2003) AtNRAMP3, a multispecifc vacuolar metal transporter involved in plant responses to iron deficiency. Plant J 34:685–695
Vatansever R, Filiz E, Ozyigit II (2016) In silico analysis of Mn transporters (NRAMP1) in various plant species. Mol Biol Rep 43(3):151–163
Verret F, Gravot A, Auroy P, Leonhardt N, David P, Nussaume L, Vavasseur A, Richaud P (2004) Overexpression of AtHMA4 enhances root-to-shoot translocation of zinc and cadmium and plant metal tolerance. FEBS Lett 576(3):306–312
Wang L, Feng Z, Wang X, Wang X, Zhang X (2010) DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 26(1):136–138
Wu Z, Zhao X, Sun X, Tan Q, Tang Y, Nie Z, Hu C (2015a) Xylem transport and gene expression play decisive roles in cadmium accumulation in shoots of two oilseed rape cultivars (Brassica napus). Chemosphere 119:1217–1223
Wu Z, Zhao X, Sun X, Tan Q, Tang Y, Nie Z, Qu C, Chen Z, Hu C (2015b) Antioxidant enzyme systems and the ascorbate-glutathione cycle as contributing factors to cadmium accumulation and tolerance in two oilseed rape cultivars (Brassica napus L.) under moderate cadmium stress. Chemosphere 138:526–536
Wu D, Yamaji N, Yamane M, Kashino-Fujii M, Sato K, Feng Ma J (2016) The HvNramp5 transporter mediates uptake of cadmium and manganese, but not Iron. Plant Physiol 172(3):1899–1910
Zhao FJ, Huang XY (2018) Cadmium phytoremediation: call Rice CAL1. Mol Plant 11(5):640–642
Zhou H, Zeng M, Zhou X, Liao B-H, Peng P-Q, Hu M, Zhu W, Wu Y-J, Zou Z-J (2014) Heavy metal translocation and accumulation in iron plaques and plant tissues for 32 hybrid rice (Oryza sativa L.) cultivars. Plant Soil 386(1–2):317–329
Acknowledgements
This work was supported by National Key R & D Program of China (2018YFD0800600, 2018YFD0200903), National Natural Science Foundation of China (31870587; 31400063; 31500038) and Fundamental Research Funds for the Central Universities (XDJK2017B030; SWU116021; XDJK2018C095; SWU118114; SWU115018), Research Funds of Scientific Platform and Base Construction (cstc2014pt-sy0017), and The Recruitment Program for Foreign Experts (No. WQ20125500073).
Author information
Authors and Affiliations
Contributions
SW and NL designed and conceived the study and drafted the manuscript. SW, JS, KL and SL performed experiments and data analysis. NL, SW, KL, HM, ZX, JL, FL and SL coordinated the research and helped to finalize the manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Additional information
Responsible Editor: Juan Barcelo.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlight
Physiological, genomic and transcriptomic comparison of two rapeseed genotypes and functional characterization reveals that two BnNramps play important role in resistance to Cadmium toxicity
Electronic supplementary material
ESM 1
(DOCX 5260 kb)
Rights and permissions
About this article
Cite this article
Wang, S., Sun, J., Li, S. et al. Physiological, genomic and transcriptomic comparison of two Brassica napus cultivars with contrasting cadmium tolerance. Plant Soil 441, 71–87 (2019). https://doi.org/10.1007/s11104-019-04083-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04083-0