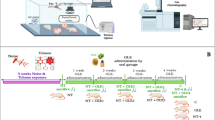
Abstract
Mosquito coil repellents are well-known indoor air pollutant with significant health concerns. The present study investigated the toxic effects of mosquito coil smoke on the heart of young and adult male rats. The animals were subjected to the smoke for 6 h/day, 6 days/week, for 4 weeks. Within the first hour after lighting the coil, significant amounts of formaldehyde, total volatile organic compounds, and particulate matter (PM2.5 and PM10) were detected. Both exposed ages, particularly the young group, showed a significant increase in the activities of serum aspartate aminotransferase, lactate dehydrogenase, creatine kinase-MB, and the levels of troponin I, myoglobin, Na+ levels, lipid profile, and inflammatory markers (interleukin-6 and C-reactive protein) as well as a significant decrease in K+ levels and cardiac Na–K ATPase activity, indicating development of cardiac inflammation and dysfunction. Furthermore, the toxic stress response was validated by significant downregulation at expression of the detoxifying enzyme cytochrome p450. Histopathological studies in both age groups, especially the young group, revealed cardiomyocyte degeneration and necrotic areas. Moreover, upregulation at the pro-apoptotic markers, caspase3, P53, and cytochrome C expressions, was detected by immunohistochemical approach in heart sections of the exposed groups. Finally, the myocardial dysfunctional effects of the coil active ingredient, meperfluthrin, were confirmed by the docking results which indicated a high binding affinity of meperfluthrin, with Na–K ATPase and caspase 3. In conclusion, both the young and adult exposed groups experienced significant cardiac toxicity changes evidenced by cell apoptosis and histopathological alterations as well as disruption of biochemical indicators.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Indoor air pollution is considered among the most prominent driver of environment-induced health risks. The main types of residential insecticide products include aerosols, liquid vaporizers, and mosquito coils. Pyrethrin/pyrethroids are the most widely used active ingredients of mosquito coils that were believed to have low toxicity in humans and untargeted animals (Li et al. 2016). However, recent studies showed adverse effects of these components on human and animal wellness (Hassan et al. 2019; Liu et al. 2022; Badr and Vornanen 2023). Mosquito coils are generally manufactured from materials such as sawdust, coconut shell powder, wrappings, dyes, oxidants such as nitrate, and other additives that control smoldering (Pierre et al. 2017). Combustion of these materials releases insecticide ingredients as well as large amounts of submicrometer particles and gaseous pollutants into the environment (Singh et al. 2020; Elehinafe et al. 2022). Naz et al. (2019) indicated that the gas phase of mosquito coil smoke contains carbonyl compounds including formaldehyde and acetaldehyde. Burning a single mosquito coil has the same bad effects as burning 75–137 cigarettes on health, in addition to the emission of formaldehyde that is equivalent to 51 cigarettes (Uthman et al. 2016; Andini et al. 2022).
A large population is routinely using mosquito coils on daily bases in closed environment to get rid of mosquito biting in complete unawareness of the potential hazard impact, especially during summer (Karim et al. 2020; Anyabolu et al. 2021). Due to the economic affordability and easy market accessibility to this type of mosquito repellent, it is widely used in Asia, South America, and Africa, including Egypt, especially in rural areas (Abdrabouh 2021). Approximately 2 billion people are exposed to elevated smoke resulting from incomplete biomass combustion of 50 billion mosquito coils burned, resulting in indoor air pollution (Syed et al. 2019). Epidemiological studies showed that children and their parents exposed to mosquito coil smoke are more susceptible to the released chemicals overnight during sleeping quarters (Hassan et al. 2019). This may cause respiratory problems (Hogarh et al. 2016) and mutagenic effects (Chen et al. 2008).
The World Health Organization suggested that early exposure to pollutants during childhood and adolescent years can alter the trajectory of the child’s health and increase the incidence of cardiac diseases (World Health Organization 2018). Moreover, Schraufnagel (2020) and Yang et al. (2020) reported that air pollutants could virtually contribute to the onset of cardiovascular diseases even at low levels of exposure. Several biomarkers are effectively used for the diagnosis of cardiac diseases. For instance, liver transaminases, especially aspartate transaminase (AST), was first described in myocardial injury and necrosis in 1954 and remained among the most important biomarkers for several years (Alvarez and Mukherjee 2011; Abdulla Al-Mamun et al. 2017). Creatine kinase-myoglobin binding (CK-MB), which is mostly found in the heart and lactate dehydrogenase (LDH) which plays an important role in the cellular production of energy, are also classical cytosolic enzymes, serving as biomarkers for the diagnosis of myocardial damage. In normal physiological conditions, they are constitutively retained within the endochylema of cardiomyocytes. When the cell membrane becomes permeable or destroyed during myocardial injury, CK-MB and LDH can easily transit through the cytoplasmic membrane into the circulation. Thus, their activity indirectly indicates the extent of myocardial damage (Xu et al. 2020).
Cardiac troponins (cTn) are specific proteins constitute of thin filaments that aggregate to form thick filaments responsible for cardiomyocytes’ contractility (Hammarstena et al. 2018). Due to its cardiac specificity, the cTn is a preferred biomarker for laboratory diagnosis of myocardial infarction (Aydin et al. 2019; Parsanathan and Jain 2020). Moreover, Na–k ATPase is related to the cardiac contractile function either in humans or animals (Yan et al. 2016). Furthermore, exposure to air pollutants may lead to immune suppression, producing an accumulation of immune complexes through different mechanisms, including the uncontrolled release of inflammatory markers (Hamanaka and Mutlu 2018; Kunovac et al. 2020; Hung et al. 2021).
On the other hand, although programmed cell death (apoptosis) is an essential process for cellular and tissue homeostasis, maintaining cell growth and differentiation, and controlling tissue repair, however, excessive heart apoptosis induces cardiac dysfunction (Del Re et al. 2019). Apoptosis and inflammation were assigned as the two major pathological mechanisms of cardiac function deterioration (Sun et al. 2017). Cardiomyocyte apoptosis is a low-abundance process, identified in the diseased heart of humans and rodents. The activation of such process is considered sufficient to lead to heart failure (Bogazzi et al. 2011; Feridooni et al. 2011).
Therefore, the present study aims to investigate the possible toxicological changes at the structure and function of the heart of young and adult male rats upon exposure to mosquito coil smoke using biochemical, histopathological, immunohistochemical, and bioinformatics approaches.
Materials and methods
Experimental protocol
Forty male Wistar rats of two ages, young rats aged 3 weeks weighing 75 ± 5 g and adults aged 9 weeks weighing 220 ± 5 g, were purchased from the Egyptian Institute for Serological and Vaccine Production, Helwan, Egypt. The animals were maintained in stainless steel cages at room temperature (25 ± 5 ℃) and humidity 50 ± 5% with a natural day and night cycle. Rats were provided food and water ad libitum under the approval of Animal Care Committee of Mansoura University, Egypt (No. Sci-Z-P-2022–82). After an acclimation period of a week, each age group was randomly subdivided into two groups, each of 10 rats: control young (CN-Y) and exposed young (EX-Y) groups, as well as control adult (CN-A) and exposed adult (EX-A) groups. Control groups inhaled ambient air, while exposed groups were allowed to inhale mosquito coil smoke in an exposure chamber of dimensions 1.5 × 0.9 × 2.1 m3 previously described in Abdrabouh (2021) for 6 h (8 am–2 pm)/day, 6 days/week for 4 weeks.
Mosquito coil characters
A commercially available brand of mosquito repellent coil (Jinjiang Laojun Chemical Co., Ltd, China) with productive approved certificate number HNP35042-I2707 was purchased from retrial outlets at Aga villages, Mansoura, Dakahlia, Egypt, was used in this study. The repellent black coils were declared to contain 0.05% meperfluthrin as an active ingredient. Each coil was approximately 15 cm in diameter, weighed 24–32 g, and was alight for 7–10 h. The same brand of repellent coils was used throughout the study period to avoid variations in commercial products.
Detection of indoor air quality
DI. ZENE portable air quality detector (model: DZ-8600, USA) was used to detect levels of particulate matter, PM2.5 and PM10 (μg/m3), total volatile organic compounds (TVOCs), and formaldehyde (HCHO) mg/m3, inside the exposure chamber at zero time and after each hour of the exposure period (6 h/day). Also, the quality of the ambient air in the control chamber was detected.
Blood sampling
After the experimental period, each animal group was sacrificed under anesthesia by intraperitoneal (I.P.) injection by a mixture of ketamine (0.08 mL/g) and xylazine (0.008 mL/g), where each rat received 0.001 mL/g of this mixture.
Two blood samples were collected per rat. The first sample was pooled in ethylenediaminetetraacetic acid (EDTA) tubes for investigating hematological parameters, including red blood cell (RBCs) count, hemoglobin (Hb) content, hematocrit (HCT)%, platelet (PLT) count, and total white blood cell (WBCs) count, using a fully automatic hematological analyzer (Sysmex XE-2100, Corp., Kobe, Japan), as described by Dacie and Lewis (2001). The second blood sample was centrifuged for 15 min at 855 × g, and the separated serum was preserved at – 20 °C until analysis.
Serum biochemical studies
In serum samples of each group, erythropoietin hormone (EPO), interleukin-6 (IL-6) as an inflammatory marker, and serum levels of cardiac troponin-I (cTn-I) were determined using the enclosed methods of ELISA kit of Cusabio, Houston, USA, with catalog numbers CSB-E07323r, CSB-E04640r, and CSB-E08594r, respectively. However, the Spinreact Kit, Ctra Santa Coloma, Spain, was used to detect serum levels of C-reactive protein (CRP), lipid profile including total cholesterol (TC), total triglycerides (TG), low- and high-density lipoprotein cholesterols (LDL) and (HDL), as well as aspartate aminotransferase (AST), lactate dehydrogenase (LDH), creatine kinase-MB (CK-MB) enzymes, in addition to Na+ and K+ ions according to the enclosed method of each parameter. Each with catalog number of MDTLIS40-P, TKBSIS48-E, MXBSIS49-I, MXBSIS51-I, MDBSIS37-I, MDBEIS46-I, TKBEIS43-I, MXBEIS31-I, BSIS54-I, and BSIS53-I, respectively. However, serum myoglobin (MB) was determined according to the included method of BioVision (Milpitas, CA, USA), catalog number E4328-100.
Heart Na–K ATPase
Half of the heart tissue from each rat was detached to estimate heart Na–K ATPase activity. Approximately 0.5 g of heart tissue was homogenized in phosphate buffer (0.1 M, pH 7.4) and centrifuged at 855 × g for 10 min. Then clear supernatants were collected and frozen at – 80 °C until detection using the method enclosed in the ELISA kit (MyBiosource, Inc., San Diego, USA), catalog number MBS7245054.
Heart histopathological examinations
The other half of the detached heart tissue was washed through a saline solution, fixed in 10% formaldehyde for 72 h, then dehydrated through an alcohol series, cleared in xylene, embedded in paraffin wax, and cut into sections of 5 μm thickness to be used in histological and immunohistochemical assays.
Histological studies
Hematoxylin and eosin (H&E) staining was used for evaluation of the heart histopathological alterations. Paraffin sections were cleared in xylene for 2 min, three times, then hydrated by three changes of ethanol (100%, 95%, 70%), 2 min for each concentration. Slides were rinsed by running tap water for 2 min. Heart sections were stained with hematoxylin solution for 3 min, then rinsed with tap water for 5 min. After that, a working eosin solution was applied for 2 min. Dehydration of heart sections was achieved by dipping slides in 95% ethanol for 5 min, then transferred to 100% ethanol twice for 2 min per each. Consequently, heart sections were cleared in xylene 3 times, 2 min per each, and a drop of mounting medium was placed over the slide and covered by a coverslip (Cardiff et al. 2014).
Histopathological evaluation and semiquantitative scoring
For H&E heart sections in both young and adult groups, the extent of cardiac tissue injury was evaluated via a semi-quantitative scoring system; five randomly selected fields were evaluated for each section in each group. The scoring of cardiac lesion severity depended on the percentage of tissue involvement, as described by Khafaga and El-Sayed (2018): none (0), no involvement of evaluated field; mild (1), involvement of 0–25% of evaluated field; moderate (2), involvement of 25–50% of evaluated field; and severe (3), involvement of 50–100% of the evaluated field.
Immunohistochemical studies
Five-micrometer-thick positively charged slides were first processed with xylene and alcohol as in histopathological procedures. Antigen retrieval was performed by boiling the samples in 9 mmol/L citrate buffer (PH 6) (Invitrogen, CA, USA) for 30 min. Heart sections were examined for expression of caspase 3, P53, cytochrome C (Cytc) (1:100 dilution; Genemed), and anti-SVV polyclonal antibody (1:75 dilution; Santa Cruz, CA) by overnight incubation with different antibodies at 4 °C. Afterward, slides were incubated with HRP conjugated secondary antibody (1:500 dilution, Santa Cruz, CA) for 1 h (Magaki et al. 2019). Staining with DAB chromogen was conducted using the Vector lab detection kit according to the manufacturers’ instructions (Vector Lab, CA, USA).
For antigen retrieval of the detoxifying enzyme, cytochrome P450 (CYP450), heart tissue slides were placed in antigen retrieval solution (citrate buffer solution, pH 6), then slides were microwaved at power 10 for 5 min two times with adding water if necessary to avoid dryness. Slides were allowed to cool for 15 min, then were washed in deionized water 5 times, and incubated with an endogenous peroxidase blocking reagent containing hydrogen peroxide and sodium azide (DAKO peroxidase blocking reagent, Cat. No. S 2001). One to two drops of the supersensitive primary monoclonal antibody against CYP450 were then put on the sections. Slides were incubated horizontally in humid chamber at room temperature for 60 min. Excess reagent was thrown off and slides were rinsed twice for 5 min in phosphate buffer solution. After blotting excess buffer, 1–2 drops of the ready-to-use DAKO EnVision + system were applied for 20 min at room temperature. Chromogen used was DAB (diaminobenzidine), 1–2 drops for 10–20 min until a desirable brown color was obtained; the slides were then washed in buffer. Sections were taken to distilled water, then nuclear counter staining was done using Mayer’s hematoxylin (Hx) solution for 3–5 min according to degree of nuclear staining. Then, sections were washed in tap water, differentiated in acid-alcohol, and washed again in water. Slides were left to dry in air, then mounted in Canada balsam (Abdraboh et al. 2011).
Quantitative morphometric measurements
Quantitative morphometric measurements by Leica Quin 500^ image analyzer computer system (Leica image system Ltd.; Cambridge, England) were used to detect the percent of area occupied by expressions of caspase 3, P53, Cytc, and CYP450 in heart sections in each group at a magnification of × 400, according to Shi et al. (2007).
Molecular docking study of meperfluthrin
The crystal structures were obtained from Protein Data Bank (https://www.rcsb.org/) and meperfluthrin structure from PubChem (https://pubchem.ncbi.nlm.nih.gov/). The docking study was performed using MOE software 2022.02(1), the protein structures were prepared, optimized as well as the ligand structure, and the docking was set to perform 100 runs keeping the top 10 poses for visual inspection.
Statistical analysis
One-way analysis of variance (ANOVA) followed by Tukey’s post hoc test was used with GraphPad Prism software (v 5.04, GraphPad Software Inc., La Jolla, CA, USA) to statistically analyze the obtained data. The results are expressed as the mean ± standard deviation (SD), and significant values were recorded at p < 0.05. The semi-quantitative scoring of cardiac injury parameters was analyzed using Kruskal–Wallis test followed by Dunn’s test to assess the significance between mean scores.
Results
Detection of air quality
As seen in Table 1, the portable air quality detector recorded low levels of PM2.5 and PM10 and zero values of TVOC and HCHO at zero time before starting the mosquito coil burn. However, upon ignition of the mosquito coil, PM2.5 and PM10 started to increase from the first hour of exposure to severely polluted limits (> 250 µg/m3 and 420 μg/m3), respectively. Additionally, TVOCs and HCHO exhibited heavily polluted categories (> 5.00 mg/m3 and 0.500 mg/m3), respectively. However, at the second hour of exposure, emissions increased, where PM2.5, PM10, and TVOCs recorded stable values of maximal readings, while HCHO increased with time, indicating heavily polluted ambient air. It should also be mentioned that the ambient air in the control chamber was of good quality.
Serum biochemical studies
The obtained data showed significantly increased levels of EPO hormone, TC, TG, LDL, AST, LDH, CK-MB, cTnI, MB, and Na+ along with significantly decreased levels of K+ in both exposed young and adult groups compared to their respective control groups. However, HDL levels were non-significantly decreased in both exposed ages. The investigated serum biochemical parameters showed no significant difference between young and adult exposed groups, except for LDH and CK-MB levels. However, the latter was also significantly decreased in the control adult group compared to its respective young group. The % change was more remarkable in TC, LDH, cTnI, Na+, and K+ in the serum of exposed young group (43.74%, 116.04%, 92.61%, 98.52%, and − 33.21%) compared to the exposed adult (37.75%, 91.72%, 74.51%, 71.28%, and − 25.45%), respectively. However, the adult exposed group showed an obvious increase in the % change for EPO, TG, LDL, HDL, CK-MB, and MB (123.53%, 94.65%, 84.49%, − 21.40%, 103.35%, and 106.40%) compared to the young exposed group (85.71%, 83.96%, 63.98%, − 7.33%, 70.03%, and 87.86%), respectively (Table 2).
The present study also showed a significant increase in serum inflammatory markers (IL-6 and CRP) in both young and adult groups exposed to mosquito coil smoke compared to their controls. This increase was more pronounced in the exposed young group, where the percentages of increases in IL-6 and CRP in the young group were 118.5% and 167.1%, while in the adult exposed group were 62.1% and 46.3%, respectively (Fig. 1). The present data also showed no significant difference in either inflammatory marker between the two exposed ages. Moreover, IL-6 showed a nonsignificant difference between the control group of both studied ages. However, CRP levels in the adult control group increased significantly compared to the control young group.
Heart Na–K ATPase
The levels of Na–K ATPase in the heart of the investigated groups exhibited a significant decrease at both exposed ages compared to the control. However, the % change was higher in the young-exposed group than in the adult-exposed group. It should also be mentioned that no significant difference was recorded either between the exposed groups or the control groups of both ages (Fig. 2).
Hematological parameters
As shown in Table 3, exposure to mosquito coil smoke for 4 weeks exhibited a non-significant increase in hematological parameters, including RBC count, Hb content, and HCT% along with a significant decrease in total WBC count in both the young and adult exposed groups compared to their respective controls. However, the total PLT count was significantly elevated in the young exposed group, but nonsignificant in the adult group. RBC count in the exposed adult group was significantly elevated compared to the exposed young group. The percentage of change (% change) between exposed young and adult groups exhibited some increase in RBC count in the adult exposed group (10.22%) compared to the young exposed group (4.21%). However, the % change in HCT% and total PLT count was increased in the young exposed group (5.68%, 23.06%) compared with the adult exposed group (0.51%, 13.17%), respectively.
Heart histopathological examination
Histological studies
The heart of the control groups in both young and adult ages stained by H&E showed regular striation and normally branched cardiomyocytes with intact intercalated discs and acidophilic sarcoplasm with centrally located single or binucleus (Fig. 3a, c). In contrast, both exposed ages, especially young group, showed disarrangement of cardiomyocytes characterized by degenerated muscle fibers, dilated, and congested blood vessels. This was accompanied by marked cytoplasmic vacuolation, pyknotic nuclei, and distinguished necrotic spots infiltrated by inflammatory cells (Fig. 3b, d). Heart sections from both young and adult control groups appeared with no lesions (no involvement of evaluated field). However, the heart of young exposed group was involved in mild vasculitis, moderate to severe degeneration, and necrosis, in addition to moderate congestion. The adult exposed group showed less involvement in histopathological lesions, appeared in mild vasculitis, degeneration, necrosis, and congestion in heart tissue (Fig. 3e). Statistically all these changes were significantly increased in young exposed rats compared to adults.
Photomicrographs of longitudinal sections of cardiac muscles stained by H&E, showing young and adult control groups (a, c) with normal branched striated cardiomyocytes with normal muscle fibers (MF) and intact intercalated discs containing connective tissue and blood vessels (arrow), acidophilic sarcoplasm, and centrally located nuclei (N). Both the exposed young and adult groups (b, d) showed disarrangement of cardiomyocytes with degenerated muscle fibers (DMF), dilated and congested blood vessels (DBV), pyknotic nuclei (curved arrow), necrotic areas (tailed arrow), and infiltration zones (star), especially in the heart of exposed young group. e Semi-quantitative scoring of heart injury in young and adult rats using Kruskal–Wallis test followed by Dunn’ test to compare all means. Letters a, b, and c means significant difference when P < 0.05; a, between CN-Y and EX-Y; b, between CN-A and EX-A; c, between Ex-Y and Ex-A
Immunohistochemical studies
The expressions of caspase 3, P53, and Cytc in cardiomyocytes of young and adult groups were evaluated. The heart sections immunostained with caspase 3 showed both the young and adult control groups with negatively stained cardiomyocytes (Fig. 4a, c). However, exposed young and adult groups showed positive expression in cardiomyocytes that was more markedly expressed in the young exposed group (Fig. 4b, d). Quantitative morphometric measurements using image analysis supported these observations, where the percent area of caspase 3 expression was increased in the young exposed group (2.217%) compared to the adult (1.664%) (Fig. 4e). However, the respective control groups were 0.134% and 0.129%, respectively. The heart sections immunostained with P53 in both young and adult control groups showed mild reactivity in cardiomyocytes (Fig. 5a, c), while exposed young and adult groups showed increased positive P53 expression (Fig. 5b, d). This was more illustrated through Fig. (5e), where morphometric measurements showed the percent area of P53 expression increased in young and adult exposed groups (1.344% and 1.364%) compared to respective control groups (0.191% and 0.141%). Moreover, a negative immune reaction of Cytc was found in cardiac muscle fibers of the control group of both studied ages (Fig. 6a, c). However, positive expression of Cytc was observed in both the exposed groups (Fig. 6b, d). Image analysis showed the percent area of such positivity as 2.463% and 2.218% in young and adult exposed groups compared to control groups (0.174% and 0.096%), respectively (Fig. 6e).
Photomicrographs of heart sections immunostained with caspase 3. Both the young and adult control groups (a, c) showed negative staining in cardiomyocytes. Exposed young and adult groups (b, d) showed positive brown expression in cardiomyocytes (arrowheads) that is markedly expressed in young-exposed rats. e Histogram showing the percent of caspase 3 in the cardiac muscle fibers of rat groups using image analysis
Photomicrographs of heart sections immunostained with P53. Both the young and adult control groups (a, c) showed mild positive brown staining in cardiomyocytes (arrowheads). Exposed young and adult groups (b, d) showed increased positive brown expression in cardiomyocytes (arrowheads). e Histogram showing the percent of P53 in the cardiac muscle fibers of rat groups using image analysis
Photomicrographs of heart sections immunostained with Cytc. Both the young and adult control groups (a, c) showed negative staining in cardiomyocytes. Exposed young and adult groups (b, d) showed positive brown expression in cardiomyocytes (arrowheads). e Histogram showing the percent of cytochrome C in the cardiac muscle fibers of rat groups using image analysis
Heart tissues in control young group showed few numbers of cardiomyocytes with a positive brownish cytoplasmic reactivity to CYP450 indicated by a moderate to marked positively stained sarcoplasm, sarcolemmal membrane, and occasionally vascular endothelial cells (Fig. 7a). Comparatively, an increased number of cells with positive brownish cytoplasmic staining reaction were recorded in the control adult group (Fig. 7c). However, expressions of positive immuno-stained cardiomyocytes in both young and adult exposed groups were lower as compared with their respective control groups with few moderately reactive cells (Fig. 7b, d). Quantitative morphometric measurements using image analysis supported these observations, where the percent area of CYP450 expression was decreased in the young control group (3.38%) compared to the control adult group (11.48%). However, the expressions of CYP450 in exposed young and adult groups were 2.04% and 2.36%, respectively (Fig. 7e).
Photomicrographs of heart sections immunostained with CYP450 showing positive brownish cytoplasmic reactivities in different experimental groups. Control young and adult groups showed higher expression (a, c); meanwhile, exposed young and adult groups showed the least reactivities (b, d). Positive cardiomyocytes are indicated by arrow and negative cells are indicated by arrowhead. e Histogram showing the percent area of CYP450 in the cardiomyocytes of rat groups using image analysis
Molecular docking study of meperfluthrin
With Na–K pump (Na–K ATPase)
Meperfluthrin showed good binding with sodium–potassium pump (Na/K-ATPase, PDB: 3KDP)(4) where it formed two hydrogen bonds with Lys437, which is involved in substrate recognition and binding, and helps to stabilize the transition state during transport, and Asn747. The binding score was − 6.57 (Fig. 8).
With Caspase-3
As shown in Fig. 9, meperfluthrin showed good binding with the pocket of caspase-3 (PDB: 5I9B)(2) with a binding score of − 6.011 through two hydrogen bonds with Gly122, which is important for the flexibility of the active site and Arg207, which is essential in the ligand recognition and binding.
Discussion
Pyrethroids are extensively used worldwide as commercial and household insecticides (Liu et al. 2022). Continuous inhalation of mosquito coil smoke containing pyrethroid may harm the hearts of both young and adults. The mosquito coils used in the present study were declared to contain meperfluthrin, as one of pyrethroids. However, the air quality assessment recorded significant elevation at the levels of PM2.5, PM10, TVOCs, and HCHO contaminants which were emitted with mosquito coil smoke within the first hour of exposure, indicating rapidly polluted ambient air. This finding supported the study of Kumar et al. (2014), Taylor et al. (2017), and Singh et al. (2020) who attributed this to the aromatization process of organic (base) material involved in the burning process, which included a complex reaction result in different emissions. Other studies attributed the emissions of the mosquito coils to the binding materials which include carbon and other various compounds (Hogarh et al. 2018; Wang et al. 2018; Karim et al. 2020). Furthermore, other findings indicated that burning one mosquito coil might release a comparable amount of PM2.5 resulting from burning 75–137 cigarettes and emitting formaldehyde equivalent to that released from burning 51 cigarettes (Hassan et al. 2019; Andini et al. 2022). On the other hand, Nascimento et al. (2017) revealed that pyrethroids and other hazardous materials can be absorbed on particulate matter and become hazardous through inhalation.
For a future effective therapy, a well-defined understanding of the molecular mechanisms behind the negative effects brought on by exposure to pyrethroids is required. The current study showed sever heart dysfunction manifested by disrupted heart function parameters, altered histological architecture of heart tissue, and decreased CYP 450 in young and adult exposed rats. These effects were coupled with increased inflammatory indicators in the serum of the same animals.
Recent study reported that regular exposure to a common home insecticide may increase the risk of cardiovascular disease and early mortality (Xue et al. 2021). The present study showed extensive cardiac histopathological alterations with increased activities of AST, CK-MB, and LDH as well as troponin I levels in the serum of young and adult animals exposed to mosquito coil smoke for 4 weeks compared to the controls. This suggests that myocardial injury was developed due to a damage of the cardiomyocytes and upregulation of the enzyme level. These results supported the earlier research findings that illustrated the chronic exposure to pyrethroid induced heart remodeling and impairment through oxidative stress, suggesting a possible positive association between pyrethroid exposure and the risk of coronary heart disease (Han et al. 2017; Marques et al. 2022). Additionally, AbouZied et al. (2021) related the elevation in CK-MB to hypoxic stress as an adaptive factor in cases of pathophysiologic myocardial hypoxia. In support, Juhász et al. (2022) elucidated that the increase in myocardial enzymes in serum was linked to a certain extent of myocardial damage after exposure to organic compounds.
Furthermore, MB is one of the cytoplasmic heme proteins, which is used for storing intracellular oxygen and facilitating the distribution of oxygen to the mitochondria in the energy formation process (Herawati et al. 2018). The obtained data revealed a significant elevation in MB levels in both exposed young and adult groups compared to the control groups. Levett et al. (2011) attributed the increase in MB to regulate the blood flow to the heart during hypoxia.
Since the changes in cellular ion fluxes and inhibition of membrane-bound ATPases can alter myocardial contractility, the effect of pyrethroid on cardiac tissue was examined. The current study showed a significant inhibition of Na–K ATPase in the heart and marked disturbance of Na+ and K + ions in the pyrethroid-exposed rats. This effect might change the transmembrane ion transport with increased Na+ influx, leading to changed membrane potential which in turn causes enhancement of catecholamine release leading to increased myocardium contraction and development of heart dysfunction. Furthermore, the molecular docking data in the present study may explain the significant decrease in Na–K ATPase activity in both young and adult exposed rats through high binding affinity with meperfluthrin.
Moreover, young and adult animals exposed to pyrethroid showed a significant increase in serum lipid fractions (TC,TG, and LDL) compared to the control groups, confirming recent study in fish (Bej et al. 2021) indicating dyslipidemia due to pyrethroid exposure. This might be attributed to the established links between hyperlipidemia and development of atherosclerosis, and the prevalence of cardiovascular diseases (Bhakkiyalakshmi et al. 2016). Taiwo et al. (2008) reported the inability of the mitochondria to utilize cholesterol, phospholipids, and fatty acids for ATP production, causing fatty degeneration of parenchymal cells. This could explain histological alterations observed in the H&E-stained heart sections, especially in young exposed rats compared to the control group. These results were in accordance with other studies that demonstrated histopathological changes in different organs, including heart and lung of rats, exposed to mosquito coil smoke (Taiwo et al. 2008; Andini et al. 2022).
To further disclose the mechanism implicated in disruption of heart structure and function after exposure to pyrethroid, apoptotic regulating proteins were evaluated. The implication of meperfluthrin in cell apoptosis was first validated using molecular docking approach which revealed a high affinity of meperfluthrin for binding with caspas-3. The current study showed induction of myocardial cell apoptosis in exposed groups which pointed out by elevation of pro-apoptotic markers, including caspase 3, P53, and Cytc in both exposed groups compared to control groups. The same observation was obtained by Urich et al. (2009) after exposing mice to PM 2.5. This effect was related to the induction of mitochondrial oxidative stress which activates P53 in the intrinsic apoptotic pathway (Reed et al. 2015; Liu et al. 2019). In this concern, Kelley et al. (2013) suggested that deleterious effects of reactive oxygen species (ROS) may be also induced by inhibiting the expression of CYP450 and compromising the ability of an organism to metabolize and eliminate xenobiotics (Andreau et al. 2012). This response was validated in the present study by significant downregulation at expression of the detoxifying enzyme CYP450 in the heart of exposed groups. This agreed with Jamieson et al. (2017) who suggested that the exposure to chemicals and other environmental agents can independently alter the expression and activity of CYP450 in the detoxification process. On the other hand, the elevated expression of P53 after stress exposure could activate caspase 3 in cardiomyocytes via induction of Cytc release from mitochondria during heart failure (Birks et al. 2008).
Recently, de Oliveira et al. (2020) added that the increase in apoptotic cells may be related to the increase at cell inflammation. The present study showed a marked increase in the serum levels of inflammatory markers, including IL6 and CRP, in young and adult exposed groups after exposure to mosquito coil smoke. These findings agreed with Castrogiovanni et al. (2016), Schraufnagel (2020), and Hung et al. (2021) who found a positive association between exposure to air pollutants and inflammatory mediators as a pathway, through which airborne particulate matter may lead to short-term increases in cardiac risk.
Furthermore, estimation of hematological parameters usually reflects the general health of the experimental animals. The present study showed insignificant changes in RBC count, Hb content, and Hct% in both the exposed young and adult groups compared to their respective control groups, confirming recent study of Anyabolu et al. (2021) who reported that inhalation of mosquito coil smoke was not anemic. Moreover, obtained data also showed significant increase in serum EPO hormone in both exposed groups. Idowu et al. (2013) suggested that the increase in EPO hormone is responsible for stimulating bone marrow to produce RBCs to overcome hypoxia. In turn, the maintenance of normal levels of hematological parameters in the present study can be attributed to the increase of EPO hormone in the exposed groups.
The present study revealed that young rats were more vulnerable to the harmful effects of mosquito coil smoke that appeared in most biochemical and histopathological investigations compared to adult rats. Buka et al. (2006) attributed this to the rapid development of the cardiopulmonary system in young age that may make them more susceptible to injury and inflammation caused by pollutants. Other studies explained that young age has more immature immune system resulting in rapid inflammatory responses and autoimmune injuries (Burroughs Peña and Rollins 2017; Wang et al. 2019). In addition, Ntarladima et al. (2019) explained the sensitivity of young age to air pollutants as they breathe in more air per unit of body weight and consequently more air pollutants. This could be also supported by the pronounced decrease in CYP450 expression in the control young group compared to control adult group, referring to low detoxifying ability in young age, making them more vulnerable to injury. This agrees with Xu et al. (2019) who observed the same impact on the liver of rats. Subsequently, the heart of young exposed rats was more susceptible to hazards than the adult.
Conclusion
Various pollutants, including particulate matter, volatile organic compounds, and formaldehyde, were present in the generated smoke of mosquito coils that contained 0.05% meperfluthrin which achieved extremely contaminated standards within the first hour of exposure. Severe cardiac consequences include significant changes in biochemical parameters, inflammation, and apoptosis. The young rats were more vulnerable to negative consequences than adults. This study recommended a controllable usage of synthetic pyrethroid insecticides as their indoor use will result in unbearable side effects on the heart as shown in the present study. Moreover, additional research is advised and warranted for a future effective therapy to further elaborate on the reduction of harmful effects of mosquito coil smoke.
Data availability
All data generated and analyzed are available upon request.
References
Abdraboh ME, Gaur RL, Hollenbach AD, Sandquist D, Raj MH, Ouhtit A (2011) Survivin is a novel target of CD44-promoted breast tumor invasion. Am J Pathol 179(2):555–563. https://doi.org/10.1016/j.ajpath.2011.04.042
Abdrabouh AE (2021) Susceptibility of young and adult rat kidneys to impacts of mosquito coil fumes. Egypt j Basic Appl Sci 8(1):1–11. https://doi.org/10.1080/2314808X.2020.1839850
Abdulla Al-Mamun M, Rahman MA, Rahman MH, Hoque KMF, Ferdousi Z, Matin MN, Abu Reza M (2017) Biochemical and histological alterations induced by the smoke of allethrin based mosquito coil on mice model. BMC Clin Pathol 17:19. https://doi.org/10.1186/s12907-017-0057-9
AbouZied H, Khalifa NA, Loudeeni MH, El-Shaarawy SA (2021) Role of creatine kinase MB in diagnosis of myocardial injury after neonatal hypoxia-ischemia. Egypt J Hosp Med 85(2):3498–3502
Alvarez AM, Mukherjee D (2011) Liver abnormalities in cardiac diseases and heart failure. Int Angiol 20:135–142
Andini A, Hasanah M, Azizah SN, Rosyadahan AR, Rimasari D, Triapadma W, Ayu F, Syafiuddin A (2022) The effect of insect repellent exposure on leukocyte profile and histopathologic findings in lungs. Biointerface Res Appl Chem 12(6):7796–7803
Andreau K, Leroux M, Bouharrour A (2012) Health and cellular impacts of air pollutants: from cytoprotection to cytotoxicity. Biochem Res Int 2012:1–19. https://doi.org/10.1155/2012/493894
Anyabolu AE, Ezejindu DN, Obinwa BN (2021) Evaluation of toxic effect of d-alletrin based mosquito’s coil on the lungs and selected haematological parameters of adult wistar rats. J Adv Med Pharmac Sci 23(4):9–19
Aydin S, Ugur K, Aydin S, Sahin I, Yardim M (2019) Biomarkers in acute myocardial infarction: current perspectives. Vasc Health Risk Manag 15:1–10
Badr A, Vornanen M (2023) Deltamethrin and retene toxicity to excitability of ventricular myocytes in rainbow trout (Oncorhynchus mykiss). Sohag J Sci 8(2):101–106. https://doi.org/10.21608/sjsci.2023.180890.1047
Bej S, Ghosh K, Chatterjee A, Saha NC (2021) Assessment of biochemical, hematological and behavioral biomarkers of Cyprinus carpio on exposure to a type-II pyrethroid insecticide Alpha-cypermethrin. Environ Toxicol Pharmacol 87:103717
Bhakkiyalakshmi E, Sireesh D, Sakthivadivel M, Sivasubramanian S, Gunasekaran P, Ramkumar KM (2016) Anti-hyperlipidemic and anti-peroxidative role of pterostilbene via Nrf2 signaling in experimental diabetes. Eur J Pharmacol 777:9–16
Bogazzi F, Russo D, Raggi F, Bohlooly M, Tornel J et al (2011) Cardiac extrinsic apoptotic pathway is silent in young but activated in elder mice overexpressing bovine GH: interplay with the intrinsic pathway. J Endocrinol 210:231–238. https://doi.org/10.1530/JOE-10-040
Buka I, Koranteng S, Osornio-Vargas AR (2006) The effects of air pollution on the health of children. Paediatr Child Health 11(8):513–516
Burroughs Peña MS, Rollins A (2017) Environmental exposures and cardiovascular disease: a challenge for health and development in low- and middle-income countries. Cardiol Clin 35(1):71–86. https://doi.org/10.1016/j.ccl.2016.09.001
Cardiff RD, Miller CH, Munn RJ (2014) Manual hematoxylin and eosin staining of mouse tissue sections. Cold Spring Harb Protoc 6:655–658. https://doi.org/10.1101/pdb.prot073411
Castrogiovanni P, Trovato FM, Szychlinska MA, Loreto C et al (2016) Effects of synthetic anti-inflammatory sterol in cb3v-induced myocarditis: a morphological study on heart muscle tissue. J Funct Morphol Kinesiol 1:69–89. https://doi.org/10.3390/jfmk1010069
Chen SC, Wong RH, Shiu LJ, Chiou MC, Lee H (2008) Exposure to mosquito coil smoke may be a risk factor for lung cancer in Taiwan. J Epidemiol 18:19–25
Dacie JV, Lewis SM (2001) Practical haematology, 9th edition. Churchill Livingstone, London. 633
de Oliveira AN, Pereira GM, Di Domenico M, Costanzo G et al (2020) Inflammation response, oxidative stress and DNA damage caused by urban air pollution exposure increase in the lack of DNA repair XPC protein. Environ Intern 145:106150. https://doi.org/10.1016/j.envint.2020.106150
Del Re DP, Amgalan D, Linkermann A, Liu Q, Kitsis RN (2019) Fundamental mechanisms of regulated cell death and implications for heart disease. Physiol Rev 99:1765–1817. https://doi.org/10.1152/physrev.00022.2018
Ej B, Latif N, Enesa K, Folkvang T et al (2008) Elevated p53 expression is associated with dysregulation of the ubiquitin-proteasome system in dilated cardiomyopathy. Cardiovasc Res 79:472–480. https://doi.org/10.1093/cvr/cvn083
Elehinafe FB, Okedere OB, Oladimeji TE, Anabui SO (2022) Investigation of some gaseous and trace metal emissions with their emission factors from various brands of mosquito coils used in Nigeria. Environ Health Insights 16:1–6. https://doi.org/10.1177/11786302221091741
Feridooni T, Hotchkiss A, Remley-Carr S, Saga Y, Pasumarthi KBS (2011) Cardiomyocyte specific ablation of p53 is not sufficient to block doxorubicin induced cardiac fibrosis and associated cytoskeletal changes. PLoS ONE 6(7):e22801. https://doi.org/10.1371/journal.pone.0022801
Hamanaka RB, Mutlu GM (2018) Particulate matter air pollution: effects on the cardiovascular system. Front Endocrinol 9:680. https://doi.org/10.3389/fendo.2018.00680
Hammarstena O, Mairb J, Mockel M, Lindahld B, Jaffe AS (2018) Possible mechanisms behind cardiac troponin elevations. Biomark 23(8):725–734. https://doi.org/10.1080/1354750X.2018.1490969
Han J, Zhou L, Luo M, Liang Y, Zhao W, Wang P, Zhou Z, Liu D (2017) Nonoccupational exposure to pyrethroids and risk of coronary heart disease in the Chinese population. Environ Sci Technol 51(1):664–670
Hassan Y, Ibrahim M, Abubakar UA (2019) Unpremeditated indoor pollution: a glance at recent use of mosquito coil in northern Nigeria. Int J Sci Res Rev 7(3):3570–3577
Herawati M, Mulyawan W, Farhan FS, Ferdinal F, Jusman SWA, Sadikin M (2018) Expression of hypoxia-inducible factor-1a and myoglobin in rat heart as adaptive response to intermittent hypobaric hypoxia exposure. HAYATI J Biosc 24(3):131. https://doi.org/10.4308/hjb.24.3.131
Hogarh JN, Antwi-Agyei P, Obiri-Danso K (2016) Application of mosquito repellent coils and associated self reported health issues in Ghana. Malaria J 15: Hayati J Biosci 24:131–135
Hogarh JN, Agyekum TP, Bempah CK, Emmanuel DJ et al (2018) Environmental health risks and benefits of the use of mosquito coils as malaria prevention and control strategy. Malar J 17:265–277. https://doi.org/10.1186/s12936-018-2412-4
Hung SC, Cheng HY, Yang CC, Lin CI, Hohdh CK, Lee WH, Cheng FJ, Li CJ, Chuang HY (2021) The association of white blood cells and air pollutants—a population-based study. Int J Environ Res Public Health 18:2370. https://doi.org/10.3390/ijerph18052370
Idowu ET, Aimufua OJ, Ejovwoke YO, Akinsanya B, Otubanjo AO (2013) Toxicological effects of prolonged and intense use of mosquito coil emission in rats and its implications on malaria control. Int J Trop Biol 61(3):1463–1473
Jamieson KL, Endo T, Darwesh AM, Samokhvalov V, Seubert JM (2017) Cytochrome P450-derived eicosanoids and heart function. Pharmacol Ther 179:47–83. https://doi.org/10.1016/j.pharmthera.2017.05.005
Juhász L, Tallósy SP, Nászai A, Varga G, Érces D, Boros M (2022) Bioactivity of inhaled methane and interactions with other biological gases. Front Cell Dev Biol 9:824749. https://doi.org/10.3389/fcell.2021.824749
Karim R, Ghose DK, Rahman F, Hossain T, Rahman R, Rahman A, Islam R (2020) Evidence of health complications caused by mosquito coil smoke inhalation in mouse model. J Adv Biotechnol Exp Ther 3(2):122–127. https://doi.org/10.5455/jabet.2020.d116
Katrukhaa IA, Katrukha AG (2021) Myocardial injury and the release of troponins I and T in the blood of patients. Clin Chem 67(1):124–130. https://doi.org/10.1093/clinchem/hvaa281
Kelley MA, Hebert VY, Thibeaux TM, Orchard MA, Hasan F et al (2013) Model combustion-generated particulate matter containing persistent free radicals redox cycle to produce reactive oxygen species. Chem Res Toxicol 26:1862–1871. https://doi.org/10.1021/tx400227s
Khafaga AF, El-Sayed YS (2018) Spirulina ameliorates methotrexate hepatotoxicity via antioxidant, immune stimulation, and proinflammatory cytokines and apoptotic proteins modulation. Life Sci 196:9–17
Kumar R, Gupta N, Kumar D, Kumar MA, Singh K, Kumar M (2014) Monitoring of indoor particulate matter during burning of mosquito coil, incense sticks and dhoop. Indian J Allergy Asthma Immun 28:68–73
Kunovac XA, Hathaway XQA, Pinti MV, Taylor AD, Hollander JM (2020) Cardiovascular adaptations to particle inhalation exposure: molecular mechanisms of the toxicology. Am J Physiol - Heart Circ Physiol 319:H282–H305. https://doi.org/10.1152/ajpheart.00026.2020
Levett DZ, Fernandez BO, Riley HL, Martin DS, Mitchell K, Leckstrom CA, Ince C et al (2011) The role of nitrogen oxides in human adaptation to hypoxia. Sci Rep 1:109–117. https://doi.org/10.1038/srep00109
Li H, Lydy MJ, You J (2016) Pyrethroids in indoor air during application of various mosquito repellents: occurrence, dissipation and potential exposure risk. Chemosphere 144:2427–2435
Liu J, Zhanga J, Rena L, Wei J et al (2019) Fine particulate matters induce apoptosis via the ATM/P53/CDK2 and mitochondria apoptosis pathway triggered by oxidative stress in rat and GC-2spd cell. Ecotoxicol Environ Saf 180:280–287. https://doi.org/10.1016/j.ecoenv.2010.05.013
Liu XC, Strodl E, Huang LH, Lu Q, Liang Y, Chen WQ (2022) First trimester of pregnancy as the sensitive period for the association between prenatal mosquito coil smoke exposure and preterm birth. Int J Environ Res Public Health 19:11771. https://doi.org/10.3390/ijerph191811771
Magaki S, Hojat SA, Wei B, So A, Yong WH (2019) An introduction to the performance of immunohistochemistry. Methods Mol Biol 1897:289–298. https://doi.org/10.1007/978-1-4939-8935-5_25
Marques LP, Joviano-Santos JV, Souza DS, Santos-Miranda A, Roman-Campos D (2022) Cardiotoxicity of pyrethroids: molecular mechanisms and therapeutic options for acute and long-term toxicity. Biochem Soc Trans 50(6):1737–1751
Nascimento MM, da Rocha GO, de Andrade JB (2017) Pesticides in fine airborne particles: from a green analysis method to atmospheric characterization and risk assessment. Sci Rep 7(1):2267. https://doi.org/10.1038/s41598-017-02518-1
Naz M, Rehman N, Ansari MN, Kamal M, Ganaie MA, Awaad AS, Alqasoumi SI (2019) Comparative study of subchronic toxicities of mosquito repellents (coils, mats and liquids) on vital organs in Swiss albino mice. Saudi Pharm J 27:348–353
Ntarladima A-M, Vaartjes I, Grobbee DE, Dijst M et al (2019) Relations between air pollution and vascular development in 5-year old children: a cross-sectional study in the Netherlands. Environ Health 18:50–61. https://doi.org/10.1186/s12940-019-0487-1
Parsanathan R, Jain SK (2020) Novel invasive and noninvasive cardiac-specific biomarkers in obesity and cardiovascular diseases. Metab Syndr Relat Disord 18(1):10–30. https://doi.org/10.1089/met.2019.0073
Pierre M, Didier AA, Baptiste AAJ (2017) Evaluation of the acute toxicity of a mosquito coil based-meperfluthrin. Moj Toxicol 3(7):167–170. https://doi.org/10.15406/mojt.2017.03.00076
Reed JR, dela Cruzb ALN, Lomnickib SM, Backes WL (2015) Inhibition of cytochrome P450 2B4 by environmentally persistent free radical-containing particulate matter. Biochem Pharmacol 95(2):126–132. https://doi.org/10.1016/j.bcp.2015.03.012
Schraufnagel DE (2020) The health effects of ultrafine particles. Exp Mol Med 52:311–317. https://doi.org/10.1038/s12276-020-0403-3
Shi SR, Liu C, Young L (2007) Development of an optimal antigen retrieval protocol for immunohistochemistry of retinoblastoma protein (pRB) in formalin fixed, paraffin sections based on comparison of different methods. Biotech Histochem 82(6):301–309
Singh R, Singh H, Ranjana MP, Diwan RK, Kumari S (2020) Protective role of vitamin c against toxic indoor pollutants released from pyrethroid based mosquito coil on a rat model: a histopathological study. Era’s J Med Res 7(1):1–6
Sun Y-H, Qiang SU, Lang LI, Wang X-T et al (2017) Expression of p53 in myocardium following coronary microembolization in rats and its significance. J Geriatr Cardiol 14:292–300
Syed M, Tousia S, Balowria KS, Jabeen N, Faruqi NA, Beg A (2019) Haematological and histopathological effects of mosquito coil smoke on kidney-an experimental study on albino rat. Acad Anatom Int 5(1):49–53. https://doi.org/10.21276/aanat.2019.5.1.11
Taiwo VO, Nwagbara ND, Suleiman R, Angbashim JE, Zarma MJ (2008) Clinical signs and organ pathology in rats exposed to graded dose of pyrethroids containing mosquito smoke and aerosolized insecticidal spray. Afr J Biomed Res 11:97–104
Taylor ET, Beah JM, Barrie M et al (2017) Characterizing emission of particulate matter from combusting different products of mosquito coils in southern Sierra Leone. JSM Environ Sci Ecol 5:1054–1059
Urich D, Soberanes S, Burgess Z, Chiarella SE et al (2009) Proapoptotic Noxa is required for particulate matter-induced cell death and lung inflammation. FASEB J 23:2055–2064
Uthman GS, Mihi UH, Musa HA (2016) Exposure to incense of meperfluthrin mosquito repellent smoke causes biochemical and histopathological changes in rabbits. Niger J Pharm Biomed Res 1:68–74
Wang L, Zheng X, Stevanovic S, Xiang Z, Liu J, Shi H et al (2018) Characterizing pollutant emissions from mosquito repellents incenses and implications in risk assessment of human health. Chemosphere 191:962–970
Wang J, Cao H, Sun D, Qi Z et al (2019) Associations between ambient air pollution and mortality from all causes, pneumonia, and congenital heart diseases among children aged under 5 years in Beijing, China: a population-based time series study. Environ Res. https://doi.org/10.1016/j.envres.2019.108531
World Health Organization (WHO) (2018) Report. Air pollution and child health—prescribing clean air. https://apps.who.int/iris/handle/10665/275545
Xu S-F, Hu A-L, Xie L, Liu J-J, Wu Q, Liu J (2019) Age-associated changes of cytochrome P450 and related phase-2 gene/proteins in livers of rats. Peer J 7:e7429–e7449. https://doi.org/10.7717/peerj.7429
Xu Y, Tang C, Tan S, Duan J, Tian H, Yang Y (2020) Cardioprotective effect of isorhamnetin against myocardial ischemia reperfusion (I/R) injury in isolated rat heart through attenuation of apoptosis. J Cell Mol Med 24:6253–6262
Xue Q, Pan A, Wen Y, Huang Y, Chen D, YangC-X WuJH, Yang J, PanJ Pan X-F (2021) Association between pyrethroid exposure and cardiovascular disease: a national population-based cross-sectional study in the US. Environ Int 153:106545. https://doi.org/10.1016/j.envint.2021.106545
Yan X, Xun M, Li J, Wu L, Dou X, Zheng J (2016) Activation of Na+/K+-ATPase attenuates high glucose-induced H9c2 cell apoptosis via suppressing ROS accumulation and MAPKs activities by DRm217. Acta Biochim Biophys Sin 48(10):883–893. https://doi.org/10.1093/abbs/gmw079
Yang AM, Lo K, Zheng TZ, Yang JL, Bai YN, Feng YQ, Cheng N, Liu SM (2020) Environmental heavy metals and cardiovascular diseases: status and future direction. Chronic Dis Transl Med 6(4):251–259
Acknowledgements
Faculty of Sciences, Mansoura University, provided all possible facilities and equipment to complete this research.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
The author conducted all the experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the Faculty of Science, Mansoura University (No. Sci-Z-P-2022–82).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The author declares no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdrabouh, A.ES. Toxicological and histopathological alterations in the heart of young and adult albino rats exposed to mosquito coil smoke. Environ Sci Pollut Res 30, 93070–93087 (2023). https://doi.org/10.1007/s11356-023-28812-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-28812-2