Abstract
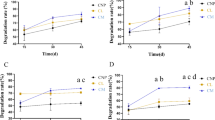
Chinese brake fern (Pteris vittata) can increase tolerance to arsenic (As) and cadmium (Cd) toxicity by regulating rhizosphere microbial diversity. However, effects of combined As–Cd stress on microbial diversity and plant uptake and transport remain poorly understood. Therefore, effects of different concentrations of As and Cd on Pteris vittata (P. vittata) metal uptake and translocation and rhizosphere microbial diversity were examined in a pot experiment. The results indicated that As primarily accumulated aboveground in P. vittata (bioconcentration factor (BCF) ≤ 51.3; translocation factor (TF) ≈ 4), whereas Cd primarily accumulated belowground (BCF ≤ 39.1; TF < 1). Under single As, single Cd, and As–Cd combined stress, the most dominant bacteria and fungi were Burkholderia-Caballeronia-P (6.62–27.92%) and Boeremia (4.61–30.42%), Massilia (8.07–11.51%) and Trichoderma (4.47–22.20%), and Bradyrhizobium (2.24–10.38%) and Boeremia (3.16–45.69%), respectively, and their abundance ratios had a significant impact on the efficiency of P. vittata for As and Cd accumulation. However, with increasing As and Cd concentrations, abundances of plant pathogenic bacteria such as Fusarium and Chaetomium (the highest abundances were 18.08% and 23.72%, respectively) increased, indicating that As and Cd concentrations reduced P. vittata resistance to pathogens. At high soil concentrations of As–Cd, although plant As and Cd contents increased and microbial diversity was highest, enrichment efficiency and transportability of As and Cd decreased substantially. Therefore, pollution intensity should be considered when evaluating P. vittata suitability for phytoremediation of combined As–Cd contaminated soils.



Similar content being viewed by others
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anderson MJ, Crist TO, Chase JM et al (2011) Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecol Lett 14(1):19–28
Apuan DA, Apuan MJB, Perez TR et al (2016) Propagation protocol of Pteris vittata L. using spores for phytoremediation. Int J Biol Sci 8:14–21
Ashraf MA, Hussain I, Rasheed R et al (2017) Advances in microbe-assisted reclamation of heavy metal contaminated soils over the last decade: a review. J Environ Manage 198:132–143
Ashraf S, Ali Q, Zahir ZA et al (2019) Phytoremediation: environmentally sustainable way for reclamation of heavy metal polluted soils. Ecotoxicol Environ Saf 174:714–727
Bai G, Shaner G (2004) Management and resistance in wheat and barley to Fusarium head blight. Annu Rev Phytopathol 42:135–161
Balestri M, Ceccarini A, Forino LMC et al (2014) Cadmium uptake, localization and stress-induced morphogenic response in the fern Pteris vittata. Planta 239(5):1055–1064
Barra Caracciolo A, Terenzi V (2021) Rhizosphere microbial communities and heavy metals. Microorganisms 9(7):1462
Biswal B, Singh SK, Patra A et al (2022) Evaluation of phytoremediation capability of French marigold (Tagetes patula) and African marigold (Tagetes erecta) under heavy metals contaminated soils. Int J Phytorem 24(9):945–954
Candeias C, Avila P, Coelho P et al (2019) Mining activities: health impacts. In: Nriagu J (ed) Encyclopedia of environmental health, 2nd edn. Elsevier, Oxford, pp 415–435
Chen T, Wei C, Huang Z et al (2002) Arsenic hyperaccumulator Pteris vittata L. and its arsenic accumulation. Chin Sci Bull 47(11):902–905
Chen S, Zhou Y, Chen Y et al (2018) FASTP: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34(17):884–890
Chi Y, Yu J, Guo Z et al (2022) First record of Chaetomium globosum causing leaf spot on Arachis hypogaea in China. J Plant Pathol 104(2):867–867
Das S, Chou ML, Jean JS et al (2017) Arsenic-enrichment enhanced root exudates and altered rhizosphere microbial communities and activities in hyperaccumulator Pteris vittata. J Hazard Mater 325:279–287
Drava G, Roccotiello E, Minganti V et al (2012) Effects of cadmium and arsenic on Pteris vittata under hydroponic conditions. Environ Toxicol Chem 31(6):1375–1380
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10(10):996–998
Fayiga AO, Ma LQ, Cao X et al (2004) Effects of heavy metals on growth and arsenic accumulation in the arsenic hyperaccumulator Pteris vittata L. Environ Pollut 132(2):289–296
Gabrijel O, Ivana JB, Robert P et al (2022) Humates and chlorides synergistically increase Cd phytoaccumulation in strawberry fruits, heightening health risk from Cd in human diet. Expo Health 14:393–410
GB 15618–2018 (2018) Soil environmental quality–risk control standard for soil contamination of agricultural land of China. Standards Press of China, Beijing, pp 1–4 (in Chinese)
Golui D, Datta SP, Dwivedi BS et al (2019) Assessing soil degradation in relation to metal pollution—a multivariate approach. Soil Sediment Contam: Int J 28(7):630–649
Gupta K, Srivastava S, Saxena G et al (2022) Application of Pteris vittata L. for phytoremediation of arsenic and biomonitoring of the process through cyto-genetic biomarkers of Trigonella foenum-graecum L. Physiol Mol Biol Plants 28(1):91–106
Han YH, Liu X, Rathinasabapathi B et al (2017) Mechanisms of efficient As solubilization in soils and As accumulation by As-hyperaccumulator Pteris vittata. Environ Pollut 227:569–577
Hibiki A, Arimura TH (2004) Environmental polices and firm-level management practices in Japan. OCED, Japan
Hou J, Fan L, Zhou M et al (2018) The combined electrokinetic and flushing remediation of multiple heavy metals co-contaminated soil enhanced with acid treatment. ES Energy Environ 2(2):82–89
Jia P, Li F, Zhang S et al (2022) Microbial community composition in the rhizosphere of Pteris vittata and its effects on arsenic phytoremediation under a natural arsenic contamination gradient. Front Microbiol 13:989272
Karim MDM (2000) Arsenic in groundwater and health problems in Bangladesh. Water Res 34(1):304–310
Laha A, Bhattacharyya S, Sengupta S et al (2021) Investigation of arsenic-resistant, arsenite-oxidizing bacteria for plant growth promoting traits isolated from arsenic contaminated soils. Arch Microbiol 203(7):4677–4692
Lampis S, Santi C, Ciurli A et al (2015) Promotion of arsenic phytoextraction efficiency in the fern Pteris vittata by the inoculation of As-resistant bacteria: a soil bioremediation perspective. Front Plant Sci 6:80
Li Y, Jia S, Liu J (2022) Solidification, remediation and long-term stability of heavy metal contaminated soil under the background of sustainable development. Sci Rep 12:10330
Lorenzo-Gutiérrez D, Gómez-Gil L, Guarro J et al (2019) Role of the Fusarium oxysporum metallothionein Mt1 in resistance to metal toxicity and virulence. Metallomics 11(7):1230–1240
Ma Y, Rajkumar M, Luo YM et al (2011) Inoculation of endophytic bacteria on host and non-host plants—effects on plant growth and Ni uptake. J Hazard Mater 195:230–237
Ma Y, Oliveira RS, Nai F et al (2015) The hyperaccumulator Sedum plumbizincicola harbors metal-resistant endophytic bacteria that improve its phytoextraction capacity in multi-metal contaminated soil. J Environ Manage 156:62–69
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27(21):2957–2963
Malekzadeh E, Alikhani HA, Savaghebi-Firoozabadi GR et al (2012) Bioremediation of cadmium-contaminated soil through cultivation of maize inoculated with plant growth-promoting rhizobacteria. Bioremediat J 16(4):204–211
Morkunas I, Woźniak A, Mai VC et al (2018) The role of heavy metals in plant response to biotic stress. Molecules 23(9):2320
Muszynska E, Hanus-Fajerska E (2015) Why are heavy metal hyperaccumulating plants so amazing? BioTechnologia. J Biotechnol Comput Biol Bionanotechnology 96(4):256–271
Nan G, Guo L, Gao Y et al (2019) Speciation analysis and dynamic absorption characteristics of heavy metals and deleterious element during growing period of Chinese peony. Int J Phytorem 21(14):1407–1414
Oladoye PO, Olowe OM, Asemoloye MD (2022) Phytoremediation technology and food security impacts of heavy metal contaminated soils: a review of literature. Chemosphere 288:132555
Peralta JM, Travaglia CN, Romero-Puertas MC et al (2020) Unraveling the impact of arsenic on the redox response of peanut plants inoculated with two different Bradyrhizobium sp. strains. Chemosphere 259:127410
Qian F, Huang X, Su X et al (2022) Responses of microbial communities and metabolic profiles to the rhizosphere of Tamarix ramosissima in soils contaminated by multiple heavy metals. J Hazard Mater 438:129469
Ren Y, Yu G, Shi C et al (2022) Majorbio Cloud: a one-stop, comprehensive bioinformatic platform for multiomics analyses. Imeta 1(2):e12
Saadaoui W, Gamboa-Rosales H, Sifuentes-Gallardo C et al (2022) Effects of lead, copper and cadmium on bioaccumulation and translocation factors and biosynthesis of photosynthetic pigments in Vicia faba L. (broad beans) at different stages of growth. Appl Sci 12(18):8941
Sarangi BK, Tiwari S, Pandey RA et al (2014) Efficacy of three different plant species for arsenic phyto-extraction from hydroponic system. Environ Eng Res 19(2):145–149
Thompson RS, Roller R, Mika A et al (2017) Dietary prebiotics and bioactive milk fractions improve NREM sleep, enhance REM sleep rebound and attenuate the stress-induced decrease in diurnal temperature and gut microbial alpha diversity. Front Behav Neurosci 10:240
Tirry N, Joutey NT, Sayel H et al (2018) Screening of plant growth promoting traits in heavy metals resistant bacteria: prospects in phytoremediation. J Genet Eng Biotechnol 16(2):613–619
Tu C, Ma LQ (2002) Effects of arsenic concentrations and forms on arsenic uptake by the hyperaccumulator ladder brake. J Environ Qual 31(2):641–647
USEPA (2007) Microwave assisted acid digestion of sediment, sludges, soils, and oils. U S Environ Protect Agency Method 3051A:1–30
Wang Q, Garrity GM, Tiedje JM et al (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73(16):5261–5267
Wang Y, Shen H, Xu L et al (2015) Transport, ultrastructural localization, and distribution of chemical forms of lead in radish (Raphanus sativus L.). Front Plant Sci 6:293
Wang X, Zhou C, Xiao X et al (2022) Phytoextraction potential of arsenic and cadmium and response of rhizosphere microbial community by intercropping with two types of hyperaccumulators. Environ Sci Pollut Res 29(60):91356–91367
Wei FS (1992) Modern analysis methods of soil elements. China Environmental Science Press, Beijing, pp 1–251 (in Chinese)
Wood JL, Zhang C, Mathews ER et al (2016) Microbial community dynamics in the rhizosphere of a cadmium hyper-accumulator. Sci Rep 6(1):1–10
Xia Q, Lamb D, Peng C et al (2017) Interaction effects of As, Cd and Pb on their respective bioaccessibility with time in co-contaminated soils assessed by the Unified BARGE Method. Environ Sci Pollut Res 24(6):5585–5594
Xiong J, Wu L, Tu S et al (2010) Microbial communities and functional genes associated with soil arsenic contamination and the rhizosphere of the arsenic-hyperaccumulating plant Pteris vittata L. Appl Environ Microbiol 76(21):7277–7284
Yan Y, Yang J, Wan X et al (2021) Temporal and spatial differentiation characteristics of soil arsenic during the remediation process of Pteris vittata L. and Citrus reticulata Blanco intercropping. Sci Total Environ 812:152475
Yu X, Li Y, Li Y et al (2017) Pongamia pinnata inoculated with Bradyrhizobium liaoningense PZHK1 shows potential for phytoremediation of mine tailings. Appl Microbiol Biotechnol 101(4):1739–1751
Zhang C, Nie S, Liang J et al (2016) Effects of heavy metals and soil physicochemical properties on wetland soil microbial biomass and bacterial community structure. Sci Total Environ 557:785–790
Acknowledgements
We thank LetPub (www.letpub.com) for the linguistic assistance and pre-submission expert review.
Funding
This research was financially supported by the National Natural Science Foundation of China (No. 31960057) and the Yunnan Key Laboratory of Metal–Organic Molecular Materials and Devices (YNMOMD2213).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Shizhan Cui, Han Xiao, Deren Miao, and Wanqiu Yang. The first draft of the manuscript was written by Shizhan Cui. The supervision and writing — review and editing were finished by Wanqiu Yang. All authors commented on previous versions of the manuscript and read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
The authors confirm that the work described has not been published before, that it is not under consideration for publication elsewhere. The manuscript has been approved by all co-authors. The manuscript has been approved by the responsible authorities at the institution where the work is carried out.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Diane Purchase
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cui, S., Xiao, H., Miao, D. et al. Metal uptake and translocation by Chinese brake fern (Pteris vittata) and diversity of rhizosphere microbial communities under single and combined arsenic and cadmium stress. Environ Sci Pollut Res 30, 85198–85209 (2023). https://doi.org/10.1007/s11356-023-28448-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-28448-2