Abstract
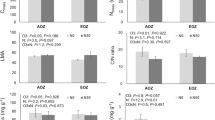
Long-chain fatty acids (LCFAs) in leaves have attracted attention as nutritious phytochemicals and olfactory signals that influence the behavior and growth of herbivorous insects. In recognition of the negative effects of increasing tropospheric ozone (O3) levels on plants, LCFAs can be altered through peroxidation by O3. However, how elevated O3 changes the amount and composition of LCFAs in field-grown plants is still unknown. We investigated palmitic, stearic, oleic, linoleic, linolenic LCFAs in the two leaf types (spring and summer) and two stages (early and late stage after expansion) of Japanese white birch (Betula platyphylla var. japonica) after a multi-year O3 exposure on the field. Summer leaves exhibited a distinct composition of LCFAs under elevated O3 at the early stage, whereas both stages of spring leaves did not exhibit significant changes in LCFAs composition by elevated O3. In the spring leaves, the amounts of saturated LCFAs significantly increased at the early stage, however, the amount of total, palmitic, and linoleic acids at the late stage were significantly decreased by elevated O3. Summer leaves had a lower amount of all LCFAs at both leaf stages. Regarding the early stage of summer leaves, the lower amount of LCFAs under elevated O3 was possibly due to O3-suppressed photosynthesis in the current spring leaves. Furthermore, the decrease ratio of spring leaves over time was significantly increased by elevated O3 in all LCFAs, whereas summer leaves did not exhibit such an effect. These findings suggest that further studies should be conducted to reveal the biological functions of LCFAs under elevated O3, considering the leaf type- and stage-dependent changes of LCFAs.



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abu ElEla SA, Agathokleous E, Koike T (2018) Growth and nutrition of Agelastica coerulea (Coleoptera: Chrysomelidae) larvae changed when fed with leaves obtained from an O3-enriched atmosphere. Environ Sci Pollut Res 25:13186–13194. https://doi.org/10.1007/s11356-018-1683-1
Agathokleous E, Sakikawa T, Abu ElEla SA, Mochizuki T, Nakamura M, Watanabe M, Kawamura K, Koike T (2017) Ozone alters the feeding behavior of the leaf beetle Agelastica coerulea (Coleoptera: Chrysomelidae) into leaves of Japanese white birch (Betula platyphylla var. japonica). Environ Sci Pollut Res 24:17577–17583. https://doi.org/10.1007/s11356-017-9369-7
Agathokleous E, Kitao M, Wang X, Mao Q, Harayama H, Manning WJ, Koike T (2021) Ethylenediurea (EDU) effects on Japanese larch: an one growing season experiment with simulated regenerating communities and a four growing season application to individual saplings. J For Res 32:2047–2057. https://doi.org/10.1007/s11676-020-01223-6
Agathokleous E, Kitao M, Shi C, Masui N, Abu ElEla SA, Hikino K, Satoh F, Koike T (2022) Ethylenediurea (EDU) spray effects on willows (Salix sachalinensis F. Schmid) grown in ambient or ozone-enriched air: implications for renewable biomass production. J For Res 33:397–422. https://doi.org/10.1007/s11676-021-01400-1
Aljbory Z, Chen MS (2018) Indirect plant defense against insect herbivores: a review. Insect Sci 25:2–23. https://doi.org/10.1111/1744-7917.12436
Bosch OJ, Geier M, Boeckh J (2000) Contribution of fatty acids to olfactory host finding of female Aedes aegypti. Chem Senses 25:323–330. https://doi.org/10.1093/oxfordjournals.chemse.a014042
Canavoso LE, Jouni ZE, Karnas KJ, Pennington JE, Wells MA (2001) Fat metabolism in insects. Annu Rev Nutr 21:23–46. https://doi.org/10.1146/annurev.nutr.21.1.23
Chu Y, Cheng TF, Gen M, Chan CK, Lee AKY, Chan MN (2019) Effect of ozone concentration and relative humidity on the heterogeneous oxidation of linoleic acid particles by ozone: an insight into the interchangeability of ozone concentration and time. ACS Earth Space Chem 3:779–788. https://doi.org/10.1021/acsearthspacechem.9b00002
Das S, Koner A, Barik A (2019) A beetle biocontrol agent of rice-field weeds recognizes its host plants by surface wax long-chain alkanes and free fatty acids. Chemoecology 29:155–170. https://doi.org/10.1007/s00049-019-00285-1
Downer RGH, Matthews JR (1976) Patterns of lipid distribution and utilisation in insects. Am Zool 16:733–745. https://doi.org/10.1093/icb/16.4.733
Duque L, Poelman EH, Steffan-Dewenter I (2019) Plant-mediated effects of ozone on herbivores depend on exposure duration and temperature. Sci Rep 9:19891. https://doi.org/10.1038/s41598-019-56234-z
Eigenbrode SD, Espelie KE (1995) Effects of plant epicuticular lipids on insect herbivores. Annu Rev Entomol 40:171–194. https://doi.org/10.1146/annurev.en.40.010195.001131
Erb M, Huber M, Robert CAM, Ferrieri AP, Machado RAR, Arce CCM (2013) The role of plant primary and secondary metabolites in root-herbivore behaviour, nutrition and physiology. In: Advances in Insect Physiology. Vol 45. Elsevier Ltd, pp 53–95. https://doi.org/10.1016/B978-0-12-417165-7.00002-7
Feng Z, Agathokleous E, Yue X, Oksanen E, Paoletti E, Sase H, Gandin A, Koike T, Calatayud V, Yuan X, Liu X, De Marco A, Jolivet Y, Kontunen-Soppela S, Hoshika Y, Saji H, Li P, Li Z, Watanabe M, Kobayashi K (2021) Emerging challenges of ozone impacts on Asian plants: actions are needed to protect ecosystem health. Ecosyst Health Sustain 7:1911602. https://doi.org/10.1080/20964129.2021.1911602
Friend WG (1958) Nutritional requirements of phytophagous insects. Annu Rev Entomol 3:57–74. https://doi.org/10.1146/annurev.en.03.010158.000421
Fuentes JD, Roulston TH, Zenker J (2013) Ozone impedes the ability of a herbivore to find its host. Environ Res Lett 8:014048. https://doi.org/10.1088/1748-9326/8/1/014048
Fürstenberg-Hägg J, Zagrobelny M, Bak S (2013) Plant defense against insect herbivores. Int J Mol Sci 14:10242–10297. https://doi.org/10.3390/ijms140510242
Hara H (2000) Major insects injuries to the trees of Betula spp. and Fraxinus mandshurica var. japonica in Hokkaido and their managements. Bull Hokkaido For Res Inst 37:67–74 [in Japanese]
Hoshika Y, Watanabe M, Inada N, Mao Q, Koike T (2013) Photosynthetic response of early and late leaves of white birch (Betula platyphylla var. japonica) grown under free-air ozone exposure. Environ Pollut 182:242–247. https://doi.org/10.1016/j.envpol.2013.07.033
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66. https://doi.org/10.1146/annurev.arplant.59.032607.092825
Ishangulyyeva G, Najar A, Curtis JM, Erbilgin N (2016) Fatty acid composition of novel host jack pine do not prevent host acceptance and colonization by the invasive mountain pine beetle and its symbiotic fungus. PLoS One 11:e0162046. https://doi.org/10.1371/journal.pone.0162046
Karabourniotis G, Liakopoulos G, Nikolopoulos D, Bresta P (2020) Protective and defensive roles of non-glandular trichomes against multiple stresses: structure–function coordination. J For Res 31:1–12. https://doi.org/10.1007/s11676-019-01034-4
Kobayashi K (2022) Effects of ozone on agricultural crops. In: Akimoto H, Tanimoto H (eds) Handbook of air quality and climate change. Springer Nature Singapore, Singapore, pp 1–44. https://doi.org/10.1007/978-981-15-2527-8
Kurokawa J, Ohara T (2020) Long-term historical trends in air pollutant emissions in Asia: regional Emission inventory in ASia (REAS) version 3. Atmos Chem Phys 20:12761–12793. https://doi.org/10.5194/acp-20-12761-2020
Lee AKY, Chan CK (2007) Heterogeneous reactions of linoleic acid and linolenic acid particles with ozone: reaction pathways and changes in particle mass, hygroscopicity, and morphology. J Phys Chem A 111:6285–6295. https://doi.org/10.1021/jp071812l
Liechti R, Farmer EE (2002) The jasmonate pathway. Science 296:1649–1650. https://doi.org/10.1126/science.1071547
Lindroth RL (2010) Impacts of elevated atmospheric CO2 and O3 on forests: phytochemistry, trophic interactions, and ecosystem dynamics. J Chem Ecol 36:2–21. https://doi.org/10.1007/s10886-009-9731-4
Malik U, Karmakar A, Barik A (2016) Variation in fatty acids throughout the developmental stages of Vigna unguiculata (L.) Walp. Leaves Bot Lett 163:461–468. https://doi.org/10.1080/23818107.2016.1235991
Manosalva L, Pardo F, Perich F, Mutis A, Parra L, Ortega F, Isaacs R, Quiroz A (2011) Behavioral responses of clover root borer to long-chain fatty acids from young red clover (Trifolium pratense) roots. Environ Entomol 40:399–404. https://doi.org/10.1603/en10008
Masui N, Mochizuki T, Tani A, Matsuura H, Agathokleous E, Watanabe T, Koike T (2020) Does ozone alter the attractiveness of Japanese white birch leaves to the leaf beetle Agelastica coerulea via changes in biogenic volatile organic compounds (BVOCs): an examination with the y-tube test. Forests 11:58. https://doi.org/10.3390/f11010058
Masui N, Agathokleous E, Tani A, Matsuura H, Koike T (2022) Plant-insect communication in urban forests: similarities of plant volatile compositions among tree species (host vs non-host trees) for alder leaf beetle Agelastica coerulea. Environ Res 204:111996. https://doi.org/10.1016/j.envres.2021.111996
Matsuki S, Sano Y, Koike T (2004) Chemical and physical defence in early and late leaves in three heterophyllous birch species native to northern Japan. Ann Bot 93:141–147. https://doi.org/10.1093/aob/mch022
Mitra S, Sarkar N, Barik A (2017) Long-chain alkanes and fatty acids from Ludwigia octovalvis weed leaf surface waxes as short-range attractant and ovipositional stimulant to Altica cyanea (Weber) (Coleoptera: Chrysomelidae). Bull Entomol Res 107:391–400. https://doi.org/10.1017/S0007485316001012
Mitra P, Das S, Barik A (2020) Leaf waxes from Lathyrus sativus: short-range attractant and stimulant for nymph laying in a viviparous insect. Chemoecology 30:117–129. https://doi.org/10.1007/s00049-020-00303-7
Moise T, Rudich Y (2002) Reactive uptake of ozone by aerosol-associated unsaturated fatty acids: kinetics, mechanism, and products. J Phys Chem A 106:6469–6476. https://doi.org/10.1021/jp025597e
Moser-Reischl A, Rötzer T, Biber P, Ulbricht M, Uhl E, Qu L, Koike T, Pretzsch H (2019) Growth of Abies sachalinensis along an urban gradient affected by environmental pollution in Sapporo. Japan Forests 10:707. https://doi.org/10.3390/f10080707
Mukherjee A, Sarkar N, Barik A (2014) Long-chain free fatty acids from Momordica cochinchinensis leaves as attractants to its insect pest, Aulacophora foveicollis Lucas (Coleoptera: Chrysomelidae). J Asia Pac Entomol 17:229–234. https://doi.org/10.1016/j.aspen.2014.01.010
Parr MJ, Tran BMD, Simmonds MSJ, Kite GC, Credland PF (1998) Influence of some fatty acids on oviposition by the bruchid beetle, Callosobruchus maculatus. J Chem Ecol 24:1577–1593. https://doi.org/10.1023/A:1020894410107
Qu L, Wang Y, Masyagina O, Kitaoka S, Fujita S, Kita K, Prokushkin A, Koike T (2022) Larch: a promising deciduous conifer as an eco-environmental resource. In: Gonçalves AC and Fonseca TF (eds) Conifers - recent advances. IntechOpen. pp 1–37. https://doi.org/10.5772/intechopen.101887
Roy N, Laskar S, Barik A (2012) The attractiveness of odorous esterified fatty acids to the potential biocontrol agent, Altica cyanea. J Asia Pac Entomol 15:277–282. https://doi.org/10.1016/j.aspen.2012.03.001
Sakikawa T, Shi C, Nakamura M, Watanabe M, Oikawa M, Satoh F, Koike T (2016) Leaf phenology and insect grazing of Japanese white birch saplings grown under free-air ozone exposure. J Agric Meteorol 72:80–84. https://doi.org/10.2480/agrmet.D-14-00031
Sarkar N, Barik A (2015) Free fatty acids from Momordica charantia L. flower surface waxes influencing attraction of Epilachna dodecastigma (Wied.) (Coleoptera: Coccinellidae). Int J Pest Manage 61:47–53. https://doi.org/10.1080/09670874.2014.990946
Sarkar N, Mukherjee A, Barik A (2013) Olfactory responses of Epilachna dodecastigma (Coleoptera: Coccinellidae) to long-chain fatty acids from Momordica charantia leaves. Arthropod Plant Interact 7:339–348. https://doi.org/10.1007/s11829-013-9249-0
Schoonhoven LM, van Loon JJA, Dicke M (2005) Insetc-plant biology. Oxford University Press, Oxford
Shashikumar A, Bičárová S, Laurence D-R (2022) The effect of ozone on pine forests in South-Eastern France from 2017 to 2019. J for Res. https://doi.org/10.1007/s11676-022-01496-z
Sicard P, Hoshika Y, Carrari E, De Marco A, Paoletti E (2021) Testing visible ozone injury within a light exposed sampling site as a proxy for ozone risk assessment for European forests. J for Res 32:1351–1359. https://doi.org/10.1007/s11676-021-01327-7
Trowbridge AM, Stoy PC (2013) BVOC-mediated plant-herbivore interactions. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions. Springer, Dordrecht, pp 21–46
Yan B, Dai Q, Liu X, Huang S, Wang Z (1996) Flooding-induced membrane damage, lipid oxidation and activated oxygen generation in corn leaves. Plant Soil 179:261–268. https://doi.org/10.1007/BF00009336
Yan K, Chen W, He X, Zhang G, Xu S, Wang L (2010) Responses of photosynthesis, lipid peroxidation and antioxidant system in leaves of Quercus mongolica to elevated O3. Environ Exp Bot 69:198–204. https://doi.org/10.1016/j.envexpbot.2010.03.008
Acknowledgements
We thank late K. Ichikawa and Prof. F. Satoh for their support in managing the experimental nursery and appreciate Prof. A. Tani for his kind help in discussing the results. This research was funded by the Kuribayashi Educational Scholarship and Academic Foundation (Sapporo, Japan) to N.M., grant number PK341563, and by Japan Science and Technology Agency (JST) SICORP Grant Number JPMJSC18HB, to T.K. E.A. acknowledges support from National Natural Science Foundation of China (No. 4210070867) and The Startup Foundation for Introducing Talent of Nanjing University of Information Science & Technology (NUIST) (Grant Number 003080), Nanjing, China.
Funding
This research was funded by the Kuribayashi Educational Scholarship and Academic Foundation (Sapporo, Japan) to N.M., grant number PK341563, and by Japan Science and Technology Agency (JST) SICORP Grant Number JPMJSC18HB, Japan to T.K. E.A. acknowledges support from National Natural Science Foundation of China (No. 4210070867) and The Startup Foundation for Introducing Talent of Nanjing University of Information Science & Technology (NUIST) (Grant Number 003080), Nanjing, China.
Author information
Authors and Affiliations
Contributions
Conceptualization: Noboru Masui; Methodology: Noboru Masui, Shiro Inoue, Hideyuki Matsuura; Formal analysis: Shiro Inoue, Hideyuki Matsuura; Investigation: Noboru Masui; Software: Evgenios Agathokleous; Resources: Evgenios Agathokleous, Takayoshi Koike; Funding acquisition: Noboru Masui, Evgenios Agathokleous, Takayoshi Koike; Writing—Original Draft: Noboru Masui; Writing—Review & Editing: Noboru Masui, Shiro Inoue, Evgenios Agathokleous, Hideyuki Matsuura, Takayoshi Koike.
Corresponding author
Ethics declarations
Ethical approval
Not applicable
Consent to participate
Not applicable
Consent to publish
Not applicable
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Gangrong Shi
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Graduate School of Agriculture, Hokkaido University, Kita 9 Nishi 9, Kita-Ku, Sapporo 0608589, Japan is where this research was conducted.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Masui, N., Inoue, S., Agathokleous, E. et al. Elevated ozone alters long-chain fatty acids in leaves of Japanese white birch saplings. Environ Sci Pollut Res 30, 79161–79170 (2023). https://doi.org/10.1007/s11356-023-28056-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-28056-0