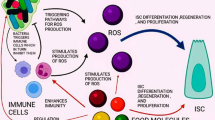
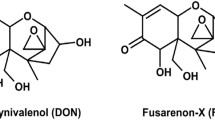
Abstract
Mycotoxins are fungal secondary metabolites that frequently occur in human and animal diets. Deoxynivalenol (DON) is one of the most widely occurring mycotoxins globally and poses significant harm to the animal husbandry industry and human health. People are increasingly aware of the adverse effects of DON on vulnerable structures and functions in the intestine, especially in the field of intestinal stem cells (ISCs). In this review, we present insights into DON that induces oxidative stress and affects the expansion of ISCs. Related studies of strategies for reducing its harm are summarized. We also discussed promising approaches such as regulation of microbiota, molecular docking, and modulation of the redox status via reducing the expression of Keap1 protein and single-cell sequencing, which may be critical for further revealing the mechanism of DON that induces oxidative stress and affects the expansion of ISCs.
Graphical abstract






Similar content being viewed by others
Availability of data and material
Not applicable
References
Abdel-Wahhab MA, El-Kady AA, Hassan AM et al (2015) Effectiveness of activated carbon and Egyptian montmorillonite in the protection against deoxynivalenol-induced cytotoxicity and genotoxicity in rats. Food Chem Toxicol 83:174–182
Adesso S, Autore G, Quaroni A et al (2017) The food contaminants nivalenol and deoxynivalenol induce inflammation in intestinal epithelial cells by regulating reactive oxygen species release. Nutrients 9c:1343
Ahad R, Zhou T, Lepp D et al (2017) Microbial detoxification of eleven food and feed contaminating trichothecene mycotoxins. BMC Biotechnol 17(1):30
Alassane-Kpembi I, Pinton P, Hupé JF et al (2018) Saccharomyces cerevisiae boulardii reduces the deoxynivalenol-induced alteration of the intestinal transcriptome. Toxins (Basel) 10(5):199
Al-Saeedi FJ (2021) Mangiferin protects oxidative stress against deoxynivalenol induced damages through Nrf2 signalling pathways in endothelial cells. Clin Exp Pharmacol 48(3):389–400
Awad WA, Böhm J, Razzazi-Fazeli E et al (2006) Effect of addition of a probiotic microorganism to broiler diets contaminated with deoxynivalenol on performance and histological alterations of intestinal villi of broiler chickens. Poult Sci 85(6):974–979
Bai X, Sun C, Liu D et al (2017) Photocatalytic degradation of deoxynivalenol using graphene/ZnO hybrids in aqueous suspension. Appl Catal B-Environ. 204:11–20
Bankaitis ED, Ha A, Kuo CJ et al (2018) Reserve stem cells in intestinal homeostasis and injury. Gastroenterology 155(5):1348–1361
Barker N, van Es JH, Kuipers J et al (2007) Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449(7165):1003–1007
Basso K, Gomes F, Bracarense AP (2013) Deoxynivanelol and fumonisin, alone or in combination, induce changes on intestinal junction complexes and in E-cadherin expression. Toxins (Basel) 5(12):2341–2352
Bensassi F, Gallerne C, Sharaf El DO et al (2012) Involvement of mitochondria-mediated apoptosis in deoxynivalenol cytotoxicity. Food Chem Toxicol 50(5):1680–1689
Bracarense APFL, Pierron A, Pinton P et al (2020) Reduced toxicity of 3-epi- deoxynivalenol and de-epoxy-deoxynivalenol through deoxynivalenol bacterial biotransformation: in vivo analysis in piglets. Food Chem Toxicol 140:111241
Broekaert N, Devreese M, Demeyere K et al (2016) Comparative in vitro cytotoxicity of modified deoxynivalenol on porcine intestinal epithelial cells. Food Chem. Toxicol 95:103–109
Burclaff J, Bliton RJ, Breau KA et al (2022) A proximal-to-distal survey of healthy adult human small intestine and colon epithelium by single-cell transcriptomics. Cell Mol Gastroenterol Hepatol 13(5):1554–1589
Carere J, Hassan YI, Lepp D et al (2018) The enzymatic detoxification of the mycotoxin deoxynivalenol: identification of DepA from the DON epimerization pathway. Microb Biotechnol 11(6):1106–1111
Chen J, Huang Z, Cao X et al (2022) Plant-derived polyphenols as Nrf2 activators to counteract oxidative stress and intestinal toxicity induced by deoxynivalenol in swine: an emerging research direction. Antioxidants (Basel) 11(12):2379
Dänicke S, Goyarts T, Valenta H et al (2005) On the effects of a hydrothermal treatment of deoxynivalenol (DON)-contaminated wheat in the presence of sodium metabisulphite (Na2S2O5) on DON reduction and on piglet performance. Anim Feed Sci Tech 118:93–108
Del Favero G, Woelflingseder L, Braun D et al (2018) Response of intestinal HT-29 cells to the trichothecene mycotoxin deoxynivalenol and its sulfated conjugates. Toxicol Lett 295:424–437
Deng Z, Yu H, Yang Z et al (2020) Gly-Pro-Ala peptide and FGSHF3 exert protective effects in DON-induced toxicity and intestinal damage via decreasing oxidative stress. Food Res Int 139:109840
Dong P, Zhang Y, Yan DY et al (2020) Protective effects of human milk-derived exosomes on intestinal stem cells damaged by oxidative stress. Cell Transplant 29:963689720912690
Fang HQ, Zhi Y, Yu Z et al (2018) The embryonic toxicity evaluation of deoxynivalenol (DON) by murine embryonic stem cell test and human embryonic stem cell test models. Food Control 86:234–240
Frobose HL, Erceg JA, Fowler SQ et al (2016) The progression of deoxynivalenol-induced growth suppression in nursery pigs and the potential of an algae-modified montmorillonite clay to mitigate these effects. J Anim Sci 94(9):3746–3759
Fuchs E, Binder EM, Heidler D et al (2002) Structural characterization of metabolites after the microbial degradation of type A trichothecenes by the bacterial strain BBSH 797. Food Addit Contam 19(4):379–386
Ganesan AR, Mohan K, Karthick Rajan D et al (2022) Distribution, toxicity, interactive effects, and detection of ochratoxin and deoxynivalenol in food: a review. Food Chem 378:131978
Garofalo M, Payros D, Oswald E et al (2022) The foodborne contaminant deoxynivalenol exacerbates DNA damage caused by a broad spectrum of genotoxic agents. Sci Total Environ 820:153280
Gu MJ, Song SK, Park SM et al (2014) Bacillus subtilis protects porcine intestinal barrier from deoxynivalenol via improved zonula occludens-1 expression. Asian-Australas J Anim Sci 27(4):580–586
Gu X, Guo W, Zhao Y et al (2019) Deoxynivalenol-induced cytotoxicity and apoptosis in IPEC-J2 cells through the activation of autophagy by inhibiting PI3K-AKT-mTOR signaling pathway. ACS Omega 4(19):18478–18486
Hanyu H, Yokoi Y, Nakamura K et al (2020) Mycotoxin deoxynivalenol has different impacts on intestinal barrier and stem cells by its route of exposure. Toxins (Basel) 12(10):610
Hou S, Ma J, Cheng Y et al (2021) The toxicity mechanisms of DON to humans and animals and potential biological treatment strategies. Crit Rev Food Sci Nutr 14:1–23
Ivanova L, Fæste CK, Solhaug A et al (2017) Role of P-glycoprotein in deoxynivalenol- mediated in vitro toxicity. Toxicol Lett 284:21–28
Ji X, Zheng W, Yao W (2020) Protective role of hydrogen gas on oxidative damage and apoptosis in intestinal porcine epithelial cells (IPEC-J2) induced by deoxynivalenol: a preliminary study. Toxins (Basel) 12(1):5
Kalaiselvi P, Rajashree K, Bharathi Priya L et al (2013) Cytoprotective effect of epigallocatechin-3-gallate against deoxynivalenol-induced toxicity through anti-oxidative and anti-inflammatory mechanisms in HT-29 cells. Food Chem Toxicol 56:110–118
Kang R, Li R, Dai P et al (2019) Deoxynivalenol induced apoptosis and inflammation of IPEC-J2 cells by promoting ROS production. Environ Pollut 251:689–698
Karlovsky P (2011) Biological detoxification of the mycotoxin deoxynivalenol and its use in genetically engineered crops and feed additives. Appl Microbiol Biot 91(3):491–504
Lau A, Wang XJ, Zhao FA et al (2010) Noncanonical mechanism of Nrf2 activation by autophagy deficiency: direct interaction between Keap1 and p62. Mol Cell Biol 30(13):3275–3285
Li D, Ye Y, Lin S et al (2014) Evaluation of deoxynivalenol-induced toxic effects on DF-1 cells in vitro: cell-cycle arrest, oxidative stress, and apoptosis. Environ Toxicol Phar 37(1):141–149
Li X, Mu P, Wen J, Deng Y (2017) Carrier-mediated and energy-dependent uptake and efflux of deoxynivalenol in mammalian cells. Sci Rep 7(1):5889
Li R, Li Y, Su Y et al (2018) Short-term ingestion of deoxynivalenol in naturally contaminated feed alters piglet performance and gut hormone secretion. Anim Sci J 89(8):1134–1143
Li XG, Zhu M, Chen MX et al (2019) Acute exposure to deoxynivalenol inhibits porcine enteroid activity via suppression of the Wnt/β-catenin pathway. Toxicol Lett 305:19–31
Li XG, Chen MX, Zhao SQ et al (2021) Intestinal models for personalized medicine: from conventional models to microfluidic primary intestine-on-a-chip. Stem Cell Rev Rep 28:1–15
Liang SJ, Li XG, Wang XQ (2019) Notch signaling in mammalian intestinal stem cells: determining cell fate and maintaining homeostasis. Curr Stem Cell Res Ther 14(7):583–590
Liao Y, Peng Z, Chen L et al (2018) Deoxynivalenol, gut microbiota and immunotoxicity: a potential approach? Food Chem Toxicol 112:342–354
Liao P, Li Y, Li M et al (2020) Baicalin alleviates deoxynivalenol-induced intestinal inflammation and oxidative stress damage by inhibiting NF-κB and increasing mTOR signaling pathways in piglets. Food Chem Toxicol 140:111326
Lin R, Sun Y, Mu P et al (2020) Lactobacillus rhamnosus GG supplementation modulates the gut microbiota to promote butyrate production, protecting against deoxynivalenol exposure in nude mice. Biochem Pharmacol 175:113868
Liu ZH, Xie WW, Zan GX et al (2021) Lauric acid alleviates deoxynivalenol-induced intestinal stem cell damage by potentiating the Akt/mTORC1/S6K1 signaling axis. Chem-Biol Interact 348:109640
Loboda A, Damulewicz M, Pyza E et al (2016) Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cell Mol Life Sci 73(17):3221–3247
Lucke A, Böhm J, Zebeli Q et al (2018) Dietary deoxynivalenol contamination and oral lipopolysaccharide challenge alters the cecal microbiota of broiler chickens. Front Microbiol 9:804
Luo S, Terciolo C, Bracarense APFL et al (2019) In vitro and in vivo effects of a mycotoxin, deoxynivalenol, and a trace metal, cadmium, alone or in a mixture on the intestinal barrier. Environ Int. 132:105082
Madden SK, Itzhaki LS (2020) Proteomics, structural and mechanistic insights into the Keap1-Nrf2 system as a route to drug discovery. BBA-Proteins Proteom 1868(7):140405
Maidana LG, Gerez J, Pinho F et al (2017) Lactobacillus plantarum culture supernatants improve intestinal tissue exposed to deoxynivalenol. Exp Toxicol Pathol 69(8):666–671
Meng XY, Zhang HX, Mezei M et al (2011) Molecular docking: a powerful approach for structure-based drug discovery. Curr Comput-Aid drug 7(2):146–157
Miró-Abella E, Torrell H, Herrero P et al (2018) Monitoring and evaluation of the interaction between deoxynivalenol and gut microbiota in Wistar rats by mass spectrometry-based metabolomics and next-generation sequencing. Food Chem Toxicol 121:124–130
Moore SR, Guedes MM, Costa TB et al (2015) Glutamine and alanyl-glutamine promote crypt expansion and mTOR signaling in murine enteroids. Am J Physiol-Gastr L 308(10):G831–G839
Myant KB, Cammareri P, McGhee EJ et al (2013) ROS production and NF-κB activation triggered by RAC1 facilitate Wnt-driven intestinal stem cell proliferation and colorectal cancer initiation. Cell Stem Cell 12(6):761–773
Osselaere A, Santos R, Hautekiet V et al (2013) Deoxynivalenol impairs hepatic and intestinal gene expression of selected oxidative stress, tight junction and inflammation proteins in broiler chickens, but addition of an adsorbing agent shifts the effects to the distal parts of the small intestine. PLoS One 8(7):e69014
Pan X, Whitten DA, Wilkerson CG et al (2014) Dynamic changes in ribosome-associated proteome and phosphoproteome during deoxynivalenol-induced translation inhibition and ribotoxic stress. Toxicol Sci 138(1):217–233
Park BJ, Takatori K, Sugita-Konishi Y et al (2007) Degradation of mycotoxins using microwave-induced argon plasma at atmospheric pressure. Surf Coat Tech 201:5733–5737
Paul MK, Bisht B, Darmawan DO et al (2014) Dynamic changes in intracellular ROS levels regulate airway basal stem cell homeostasis through Nrf2-dependent Notch signaling. Cell Stem Cell 15(2):199–214
Payros D, Alassane-Kpembi I, Pierron A et al (2016) Toxicology of deoxynivalenol and its acetylated and modified forms. Arch Toxicol 90(12):2931–2957
Payros D, Dobrindt U, Martin P et al (2017) The food contaminant deoxynivalenol exacerbates the genotoxicity of gut microbiota. mBio 8(2):e00007–e00017
Peng Z, Chen L, Xiao J et al (2017) Review of mechanisms of deoxynivalenol-induced anorexia: the role of gut microbiota. J Appl Toxicol 37(9):1021–1029
Peng Z, Liao Y, Wang X et al (2020) Heme oxygenase-1 regulates autophagy through carbon–oxygen to alleviate deoxynivalenol-induced hepatic damage. Arch Toxicol 94(2):573–588
Pinton P, Graziani F, Pujol A et al (2015) Deoxynivalenol inhibits the expression by goblet cells of intestinal mucins through a PKR and MAP kinase dependent repression of the resistin-like molecule β. Mol Nutr Food Res 59(6):1076–1087
Podgórska-Kryszczuk I, Solarska E, Kordowska-Wiater M (2022) Reduction of the Fusarium mycotoxins: deoxynivalenol, nivalenol and zearalenone by selected non-conventional yeast strains in wheat grains and bread. Molecules 27(5):1578
Qin T, Liu X, Luo Y et al (2020) Characterization of polysaccharides isolated from Hericium erinaceus and their protective effects on the DON-induced oxidative stress. Int J Biol Macromol 152:1265–1273
Rajput SA, Liang SJ, Wang XQ et al (2021) Lycopene protects intestinal epithelium from deoxynivalenol-induced oxidative damage via regulating Keap1/Nrf2 signaling. Antioxidants 10(9):1493
Rohweder D, Kersten S, Valenta H et al (2017) Bioavailability of the Fusarium toxin deoxynivalenol (DON) from wheat straw and chaff in pigs. Arch Anim Nutr 67(1):37–47
Sahu SC, Wiesenfeld PL, Kim CS et al (2010) Validation of an in vitro model for assessment of androstenedione hepatotoxicity using the rat liver cell line clone-9. J Appl Toxicol 228(5):703–709
Saint-Cyr MJ, Perrin-Guyomard A, Houée P et al (2013) Evaluation of an oral subchronic exposure of deoxynivalenol on the composition of human gut microbiota in a model of human microbiota-associated rats. Plos One 8(11):e80578
Santos-Ledo A, Luxán-Delgado B, Caballero B et al (2021) Melatonin ameliorates autophagy impairment in a metabolic syndrome model. Antioxidants 10(5):796
Sayyari A, Fæste CK, Hansen U et al (2018) Effects and biotransformation of the mycotoxin deoxynivalenol in growing pigs fed with naturally contaminated pelleted grains with and without the addition of Coriobacteriaceum DSM 11798. Food Addit Contam A 35(7):1394–1409
Schatzmayr G, Zehner F, Täubel M et al (2006) Microbiologicals for deactivating mycotoxins. Mol Nutr Food Res 50(6):543–551
Schultze N, Wanka H, Zwicker P et al (2017) Mitochondrial functions of THP-1 monocytes following the exposure to selected natural compounds. Toxicology 377:57–63
Schwartz-Zimmermann HE, Hametner C, Nagl V et al (2014) Deoxynivalenol (DON) sulfonates as major DON metabolites in rats: from identification to biomarker method development, validation and application. Anal Bioanal Chem 406(30):7911–7924
Song X, Qiao L, Chang J et al (2022) Dietary supplementation with selenium nanoparticles-enriched Lactobacillus casei ATCC 393 alleviates intestinal barrier dysfunction of mice exposed to deoxynivalenol by regulating endoplasmic reticulum stress and gut microbiota. Ecotoxicol Environ Saf 248:114276
Springler A, Hessenberger S, Schatzmayr G et al (2016) Early activation of MAPK p44/42 is partially involved in don-induced disruption of the intestinal barrier function and tight junction network. Toxins (Basel) 8(9):264
Springler A, Hessenberger S, Reisinger N et al (2017) Deoxynivalenol and its metabolite deepoxy-deoxynivalenol: multi-parameter analysis for the evaluation of cytotoxicity and cellular effects. Mycotoxin Res 33(1):25–37
Stanger BZ, Datar R, Murtaugh LC et al (2005) Direct regulation of intestinal fate by Notch. PNAS 102(35):12443–12448
Tang Y, Li J, Li F et al (2015) Autophagy protects intestinal epithelial cells against deoxynivalenol toxicity by alleviating oxidative stress via IKK signaling pathway. Free Radical Bio Med 89:944–951
Tang S, Chen S, Huang B et al (2019) Deoxynivalenol induces inhibition of cell proliferation via the Wnt/β-catenin signaling pathway. Biochem Pharmacol 166:12–22
Tian H, Biehs B, Chiu C et al (2015) Opposing activities of Notch and Wnt signaling regulate intestinal stem cells and gut homeostasis. Cell Reports 11(1):33–42
Tian Y, Tan Y, Liu N et al (2016) Detoxification of deoxynivalenol via glycosylation represents novel insights on antagonistic activities of trichoderma when confronted with fusarium graminearum. Toxins (Basel) 8(11):335
Vanhoutte I, De Mets L, De Boevre M et al (2017) Microbial detoxification of deoxynivalenol (DON), assessed via a Lemna minor l. bioassay, through biotransformation to 3-epi-DON and 3-epi-DOM-1. Toxins (Basel) 9(2):63
Videmann B, Tep J, Cavret S et al (2007) Epithelial transport of deoxynivalenol: involvement of human P-glycoprotein (ABCB1) and multidrug resistance-associated protein 2 (ABCC2). Food Chem Toxicol 45(10):1938–1947
Wang S, Yang J, Zhang B et al (2019) The potential link between gut microbiota and deoxynivalenol-induced feed refusal in weaned piglets. J Agr Food Chem 67(17):4976–4986
Wang X, Li L, Zhang G et al (2020a) A proteomic study on the protective effect of kaempferol pretreatment against deoxynivalenol-induced intestinal barrier dysfunction in a Caco-2 cell model. Food Funct 11(8):7266–7279
Wang S, Zhang C, Yang J et al (2020b) Sodium butyrate protects the intestinal barrier by modulating intestinal host defense peptide expression and gut microbiota after a challenge with deoxynivalenol in weaned piglets. J Agr Food Chem 68(15):4515–4527
Wang S, Wu K, Xue D et al (2021) Mechanism of deoxynivalenol mediated gastrointestinal toxicity: insights from mitochondrial dysfunction. Food Chem Toxicol 153:112214
Wu W, Bates MA, Bursian SJ et al (2013) Peptide YY3–36 and 5-hydroxytryptamine mediate emesis induction by trichothecene deoxynivalenol (vomitoxin). Toxicol Sci 133(1):186–195
Wu M, Xiao H, Ren W et al (2014) Therapeutic effects of glutamic acid in piglets challenged with deoxynivalenol. Plos One 9(7):e100591
Wu L, Liao P, He L et al (2015) Dietary L-arginine supplementation protects weanling pigs from deoxynivalenol-induced toxicity. Toxins (Basel) 7(4):1341–1354
Xiao K, Liu C, Qin Q et al (2020) EPA and DHA attenuate deoxynivalenol-induced intestinal porcine epithelial cell injury and protective barrier function integrity by inhibiting necroptosis signaling pathway. FASEB J 34(2):2483–2496
Yang J, Zhu C, Ye JL et al (2019) Resveratrol protects porcine intestinal epithelial cells from deoxynivalenol induced damage via the Nrf2 signaling pathway. J Agr Food Chem 67(6):1726–1735
Yang X, Liang S, Guo F et al (2020) Gut microbiota mediates the protective role of Lactobacillus plantarum in ameliorating deoxynivalenol-induced apoptosis and intestinal inflammation of broiler chickens. Poultry Sci 99(5):2395–2406
Zhang X, Jiang L, Geng C et al (2009) The role of oxidative stress in deoxynivalenol-induced DNA damage in HepG2 cells. Toxicon 54(4):513–518
Zhou JY, Wang Z, Zhang SW et al (2019a) Methionine and its hydroxyl analogues improve stem cell activity to eliminate deoxynivalenol-induced intestinal injury by reactivating Wnt/β-catenin signaling. J Agr Food Chem 67(41):11464–11473
Zhou JY, Zhang SW, Lin HL et al (2019b) Hydrolyzed wheat gluten alleviates deoxynivalenol-induced intestinal injury by promoting intestinal stem cell proliferation and differentiation via upregulation of Wnt/β-catenin signaling in mice. Food Chem Toxicol 131:110579
Zhou JY, Lin HL, Wang Z et al (2020) Zinc L-aspartate enhances intestinal stem cell activity to protect the integrity of the intestinal mucosa against deoxynivalenol through activation of the Wnt/β-catenin signaling pathway. Environ Pollut 262:114290
Zhou JY, Huang DG, Gao CQ et al (2021a) Heat-stable enterotoxin inhibits intestinal stem cell expansion to disrupt the intestinal integrity by downregulating the Wnt/beta-catenin pathway. Stem Cells 39(4):482–496
Zhou JY, Lin HL, Qin YC et al (2021b) L-carnosine protects against deoxynivalenol-induced oxidative stress in intestinal stem cells by regulating the Keap1/Nrf2 signaling pathway. Mol Nutr Food Res 65(17):e2100406
Zhu M, Wang XQ (2020) Critical review regulation of mTORC1 by small GTpases in response to nutrients. J Nutr 150(5):1004–1011
Zhu M, Qin YC, Gao CQ et al (2020) L-glutamate drives porcine intestinal epithelial renewal by increasing stem cell activity via upregulation of the EGFR-ERK-mTORC1 pathway. Food Funct 11(3):2714–2724
Zhu C, Liang SJ, Zan GX et al (2023) Selenomethionine alleviates DON-induced oxidative stress via modulating Keap1/Nrf2 signaling in the small intestinal epithelium. J Agric Food Chem 71(1):895–904
Funding
This work was supported by the National Natural Science Foundation of China (32072777, 31872389), the Basic and Applied Basic Research Foundation of Guangdong Province (2019B1515210021), and the Science and Technology Planning Project of Guangzhou (SL2022B03J01334).
Author information
Authors and Affiliations
Contributions
Xiu-qi Wang had the idea for the article and critically revised the work and Shao-jie Liang performed the literature search and finished the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent to participate
The authors declare that they consent to participate.
Consent for publication
The authors declare that they consent to publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liang, Sj., Wang, Xq. Deoxynivalenol induces intestinal injury: insights from oxidative stress and intestinal stem cells. Environ Sci Pollut Res 30, 48676–48685 (2023). https://doi.org/10.1007/s11356-023-26084-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26084-4