Abstract
Allelochemicals have been shown to inhibit cyanobacterial blooms for several years. In view of the disadvantages of “direct-added” mode, natural and pollution-free tea polyphenolic allelochemicals with good inhibitory effect on cyanobacteria were selected to prepare sustained-release particles by microcapsule technology. Results showed that the encapsulation efficiency of tea polyphenols sustained-release particles (TPSPs) was 50.6% and the particle size ranged from 700 to 970 nm, which reached the nanoscale under optimum preparation condition. Physical and chemical properties of TPSPs were characterized to prove that tea polyphenols were well encapsulated and the particles had good thermal stability. The optimal dosage of TPSPs was determined to be 0.3 g/L, at which the inhibition rate on Microcystis aeruginosa in logarithmic growth period could be maintained above 95%. Simultaneous decrease in algal density and chlorophyll-a content indicated that the photosynthesis of algal cells was affected leading to cell death. Significant changes of antioxidant enzyme activities suggested that Microcystis aeruginosa’s antioxidant systems had been disrupted. Furthermore, TPSPs increased the concentration of O2− which led to lipid peroxidation of cell membrane and a subsequent increase in malondialdehyde (MDA) content. Meanwhile, the protein content, nucleic acid content, and electrical conductivity in culture medium rose significantly indicating the cell membrane was irreversibly damaged. This work can provide a basis for the utilization of environmentally friendly algal suppressants.
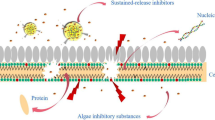
Graphical abstract








Similar content being viewed by others
Data availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
References
Ahmad P, Jaleel CA, Salem MA, Nabi G, Sharma S (2010) Roles of enzymatic and nonenzymatic antioxidants in plants during abiotic stress. Crit Rev Biotechnol 30:161–175. https://doi.org/10.3109/07388550903524243
Ahn CY, Joung SH, Choi A, Kim HS, Jang KY, Oh HM (2007) Selective control of cyanobacteria in eutrophic pond by a combined device of ultrasonication and water pumps. Environ Technol 28:371–379. https://doi.org/10.1080/09593332808618800
Ali MB, Hahn EJ, Paek KY (2006) Antioxidative responses of Echinacea angustifolia cultured roots to different levels of CO2 in bioreactor liquid cultures. Enzyme Microb Tech 39:982–990. https://doi.org/10.1016/j.enzmictec.2006.02.002
Allen RD (1995) Dissection of oxidative stress tolerance using transgenic plants. Plant Physiol 107:1049–1054. https://doi.org/10.1104/pp.107.4.1049
Anesini C, Ferraro GE, Filip R (2008) Total polyphenol content and antioxidant capacity of commercially available tea (Camellia sinensis) in Argentina. J Agric Food Chem 56:9225–9229. https://doi.org/10.1021/jf8022782
Aranganathan L, Rajasree SRR, Suman TY, Remya RR, Gayathri S, Jayaseelan C, Karthih MG, Gobalakrishnan M (2019) Comparison of molecular characteristics of type A humic acids derived from fish waste and sugarcane bagasse co-compost influenced by various alkaline extraction protocols. Microchem J 149:104038. https://doi.org/10.1016/j.microc.2019.104038
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bharati DC, Kumar H, Saroj AL (2019) Chitosan-PEG-NaI based bio-polymer electrolytes: structural, thermal and ion dynamics studies. Mater Res Express 6:12. https://doi.org/10.1088/2053-1591/ab66a3
Chen YD, Zhao C, Zhu XY, Zhu Y, Tian RN (2022) Multiple inhibitory effects of succinic acid on Microcystis aeruginosa: morphology, metabolomics, and gene expression. Environ Technol 43:3121–3130. https://doi.org/10.1080/09593330.2021.1916090
Evans JJ (1968) Peroxidases from the extreme dwarf tomato plant. Identification, isolation, and partial purification. Plant Physiol 43:1037–1041. https://doi.org/10.1104/pp.43.7.1037
Fosca M, Rau JV, Uskokovi V (2021) Factors influencing the drug release from calcium phosphate cements. Bioactive Mater 7:341–363. https://doi.org/10.1016/j.bioactmat.2021.05.032
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. II. Role of electron transfer. Arch Biochem Biophys 125:850–857. https://doi.org/10.1016/0003-9861(68)90523-7
Hejjaji EMA, Smith AM, Morris GA (2018) Evaluation of the mucoadhesive properties of chitosan nanoparticles prepared using different chitosan to tripolyphosphate (CS:TPP) ratios. Int J Biol Macromol 120:1610–1617. https://doi.org/10.1016/j.ijbiomac.2018.09.185
Hu XM, Ma XJ, Tang PW, Yuan QP (2013) Improved beta-carotene production by oxidative stress in Blakeslea trispora induced by liquid paraffin. Biotechnol Lett 35:559–563. https://doi.org/10.1007/s10529-012-1102-5
Huang HM, Xiao X, Lin F, Grossart HP, Nie ZY, Sun LJ, Xu C, Shi JY (2016) Continuous-release beads of natural allelochemicals for the long-term control of cyanobacterial growth: preparation, release dynamics and inhibitory effects. Water Res 95:113–123. https://doi.org/10.1016/j.watres.2016.02.058
ISO, 8692 (2012) Water quality - fresh water algal growth inhibition test with unicellular green algae. https://www.iso.org/standard/54150.html
Jin P, Wang H, Huang W, Liu W, Fan Y, Miao W (2018) The allelopathic effect and safety evaluation of 3,4-dihydroxybenzalacetone on Microcystis aeruginosa. Pestic Biochem Phys 147:145–152. https://doi.org/10.1016/j.pestbp.2017.08.011
Joshua S, Bailey S, Mann NH, Mullineaux CW (2005) Involvement of phycobilisome diffusion in energy quenching in cyanobacteria. Plant Physiol 138:1577–1585. https://doi.org/10.1104/pp.105.061168
Komanapalli IR, Lau BHS (1996) Ozone-induced damage of Escherichia coli K-12. Appl Microbiol Biotechnol 46:610–614. https://doi.org/10.1007/s002530050869
Laue P, Bahrs H, Chakrabarti S, Steinberg CEW (2014) Natural xenobiotics to prevent cyanobacterial and algal growth in freshwater: contrasting efficacy of tannic acid, gallic acid, and gramine. Chemosphere 104:212–220. https://doi.org/10.1016/j.chemosphere.2013.11.029
Li F, Hu H, Chong Y, Men Y, Guo M (2007) Effects of allelochemical EMA isolated from Phragmites communis on algal cell membrane lipid and ultrastructure. Huan jing ke xue = Huan jing kexue 28:1534–1538. https://doi.org/10.13227/j.hjkx.2007.07.022. (in Chinese)
Li ZH, Wang QA, Ruan XA, Pan CD, Jiang DA (2010) Phenolics and plant allelopathy. Molecules 15:8933–8952. https://doi.org/10.3390/molecules15128933
Li C, Li B, Zhu C, Meng X (2020) Optimization and characterization of magnetic tea polyphenols microcapsules. Food Fermentation Industries 46(9):128–134. https://doi.org/10.13995/j.cnki.11-1802/ts.022484
Li BH, Yin YJ, Kang LF, Feng L, Liu YZ, Du ZW, Tian YJ, Zhang LQ (2021) A review: application of allelochemicals in water ecological restoration–algal inhibition. Chemosphere 267:13. https://doi.org/10.1016/j.chemosphere.2020.128869
Liang J, Li F, Fang Y, Yang WJ, An XX, Zhao LY, Xin ZH, Cao L, Hu QH (2011) Synthesis, characterization and cytotoxicity studies of chitosan-coated tea polyphenols nanoparticles. Colloid Surf B-Biointerfaces 82:297–301. https://doi.org/10.1016/j.colsurfb.2010.08.045
Liu Y, Wang Z, Zhang L (2013) A novel method for assaying catalase activity. West China Journal of Pharmaceutical Sciences 28:403–405. https://doi.org/10.13375/j.cnki.wcjps.2013.04.032
Lu YP, Wang J, Yu Y, Shi LM, Kong FX (2014) Changes in the physiology and gene expression of Microcystis aeruginosa under EGCG stress. Chemosphere 117:164–169. https://doi.org/10.1016/j.chemosphere.2014.06.040
Lu YP, Wang J, Zhang XQ, Kong FX (2016) Inhibition of the growth of cyanobacteria during the recruitment stage in Lake Taihu. Environ Sci Pollut Res 23:5830–5838. https://doi.org/10.1007/s11356-015-5821-8
Lv LP, Zhang XD, Qiao JL (2018) Flocculation of low algae concentration water using polydiallyldimethylammonium chloride coupled with polysilicate aluminum ferrite. Environ Technol 39:83–90. https://doi.org/10.1080/09593330.2017.1296028
Ma R, Zhang D, Xu Y, Wang J, Cheng S (2019) Tea-polyphenols embedded by Pichia kudriavzevii A16 ghost and its stability. Food and Fermentation Industries 45:63–69. https://doi.org/10.13995/j.cnki.11-1802/ts.020322
Matshetshe KI, Parani S, Manki SM, Oluwafemi OS (2018) Preparation, characterization and in vitro release study of beta-cyclodextrin/chitosan nanoparticles loaded Cinnamomum zeylanicum essential oil. Int J Biol Macromol 118:676–682. https://doi.org/10.1016/j.ijbiomac.2018.06.125
Meng QH, Hu JL, Ho KC, Ji FL, Chen SJ (2009) The shape memory properties of biodegradable chitosan/poly(L-lactide) composites. J Polym Environ 17:212–224. https://doi.org/10.1007/s10924-009-0141-z
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410. https://doi.org/10.1016/s1360-1385(02)02312-9
Nakai S, Inoue Y, Hosomi M (2001) Algal growth inhibition effects and inducement modes by plant-producing phenols. Water Res 35:1855–1859. https://doi.org/10.1016/s0043-1354(00)00444-9
Ni LX, Jie XT, Wang PF, Li SY, Hu SZ, Li YP, Li Y, Acharya K (2015a) Characterization of unsaturated fatty acid sustained-release microspheres for long-term algal inhibition. Chemosphere 120:383–390. https://doi.org/10.1016/j.chemosphere.2014.07.098
Ni LX, Jie XT, Wang PF, Li SY, Wang GX, Li YP, Li Y, Acharya K (2015b) Effect of linoleic acid sustained-release microspheres on Microcystis aeruginosa antioxidant enzymes activity and microcystins production and release. Chemosphere 121:110–116. https://doi.org/10.1016/j.chemosphere.2014.11.056
Ni LX, Rong SY, Gu GX, Hu LL, Wang PF, Li DY, Yue FF, Wang N, Wu HQ, Li SY (2018) Inhibitory effect and mechanism of linoleic acid sustained-release microspheres on Microcystis aeruginosa at different growth phases. Chemosphere 212:654–661. https://doi.org/10.1016/j.chemosphere.2018.08.045
Ni L, Yue F, Zhang J, Rong S, Liu X, Wang Y, Wang P, Li D, Wang N, Wu H, Li S (2020) Cell membrane damage induced by continuous stress of artemisinin sustained-release microspheres (ASMs) on Microcystis aeruginosa at different physiological stages. Environ Sci Pollut Res Int 27:12624–12634. https://doi.org/10.1007/s11356-020-07813-5
Pal M, Yesankar PJ, Dwivedi A, Qureshi A (2020) Biotic control of harmful algal blooms (HABs): a brief review. J Environ Manage 268:10. https://doi.org/10.1016/j.jenvman.2020.110687
Pant A, Negi JS (2018) Novel controlled ionic gelation strategy for chitosan nanoparticles preparation using TPP-beta-CD inclusion complex. Eur J Pharm Sci 112:180–185. https://doi.org/10.1016/j.ejps.2017.11.020
Park J, Church J, Son Y, Kim KT, Lee WH (2017) Recent advances in ultrasonic treatment: challenges and field applications for controlling harmful algal blooms (HABs). Ultrason Sonochem 38:326–334. https://doi.org/10.1016/j.ultsonch.2017.03.003
Pei HY, Ma CX, Hu WR, Sun F (2014) The behaviors of Microcystis aeruginosa cells and extracellular microcystins during chitosan flocculation and flocs storage processes. Bioresour Technol 151:314–322. https://doi.org/10.1016/j.biortech.2013.10.077
Peng JN, Wang K, Feng TY, Zhang HZ, Li XH, Qi ZQ (2020) The effect of (1S,2R-((3-bromophenethyl)amino)-N-(4-chloro-2-trifluoromethylphenyl) cyclohexane-1-sulfonamide) on Botrytis cinerea through the membrane damage mechanism. Molecules 25:11. https://doi.org/10.3390/molecules25010094
Poulin RX, Hogan S, Poulson-Ellestad KL, Brown E, Fernández FM, Kubanek J (2018) Karenia brevis allelopathy compromises the lipidome, membrane integrity, and photosynthesis of competitors. Sci Rep-UK 8:9572. https://doi.org/10.1038/s41598-018-27845-9
Qin H, Li S, Li D (2013) An improved method for determining phytoplankton chlorophyll a concentration without filtration. Hydrobiologia 707:81–95. https://doi.org/10.1007/s10750-012-1412-6
Raven JA, Brownlee C (2001) Understanding membrane function. J Phycol 37:960–967. https://doi.org/10.1046/j.1529-8817.2001.01078.x
Rodea-Palomares I, Gonzalo S, Santiago-Morales J, Leganes F, Garcia-Calvo E, Rosal R, Fernandez-Pinas F (2012) An insight into the mechanisms of nanoceria toxicity in aquatic photosynthetic organisms. Aquat Toxicol 122:133–143. https://doi.org/10.1016/j.aquatox.2012.06.005
Romagni JG, Allen SN, Dayan FE (2000) Allelopathic effects of volatile cineoles on two weedy plant species. J Chem Ecol 26:303–313. https://doi.org/10.1023/a:1005414216848
Savela H, Spoof L, Perala N, Preede M, Lamminmaki U, Nybom S, Haggqvist K, Meriluoto J, Vehniainen M (2015) Detection of cyanobacterial sxt genes and paralytic shellfish toxins in freshwater lakes and brackish waters on Aland Islands, Finland. Harmful Algae 46:1–10. https://doi.org/10.1016/j.hal.2015.04.005
Shao JH, Wu ZX, Yu GL, Peng X, Li RH (2009) Allelopathic mechanism of pyrogallol to Microcystis aeruginosa PCC7806 (Cyanobacteria): from views of gene expression and antioxidant system. Chemosphere 75:924–928. https://doi.org/10.1016/j.chemosphere.2009.01.021
Skopinska-Wisniewska J, De la Flor S, Kozlowska J (2021) From supramolecular hydrogels to multifunctional carriers for biologically active substances. Int J Mol Sci 22:7402. https://doi.org/10.3390/ijms22147402
Spiese CE, Le T, Zimmer RL, Kieber DJ (2016) Dimethylsulfide membrane permeability, cellular concentrations and implications for physiological functions in marine algae. J Plankton Res 38:41–54. https://doi.org/10.1093/plankt/fbv106
Su W, Chen J, Zhang S-P, Kong F-X (2017) Selective inhibition of rice straw extract on growth of cyanobacteria and chlorophyta. Huan jing ke xue= Huanjing kexue 38:2901–2909. https://doi.org/10.13227/j.hjkx.201612017. (in Chinese)
Szterner P, Legendre B, Sghaier M (2010) Thermodynamic properties of polymorphic forms of theophylline. Part I: DSC, TG, X-ray study. J Therm Anal Calorim 99:325–335. https://doi.org/10.1007/s10973-009-0186-1
Tan TY, Yin Z (2009) Determination of free radicals in vivo by hydroxylamine methods. Chin J Biochem Pharmaceutics 30:330–332 (in Chinese)
Waglewska E, Pucek-Kaczmarek A, Bazylinska U (2022) Self-assembled bilosomes with stimuli-responsive properties as bioinspired dual-tunable nanoplatform for pH/temperature-triggered release of hybrid cargo. Colloids Surf B Biointerfaces 215:112524. https://doi.org/10.1016/j.colsurfb.2022.112524
Wan JJ, Guo PY, Zhang SX (2014) Response of the cyanobacterium Microcystis flos-aquae to levofloxacin. Environ Sci Pollut Res 21:3858–3865. https://doi.org/10.1007/s11356-013-2340-3
Wang J, Chen G, Zhang C (2002) The effects of water stress on soluble protein content, the activity of SOD, POD and CAT of two ecotypes of reeds (Phragmites communis). Acta Bot Boreali-Occident Sin 22:561–565 (in Chinese)
Wang J, Du M, Yu Y, Lu Y (2014) The inhibitory effect of plant extracts on Microcystis aeruginosa and its algal inhibiting characteristics. Journal of Nanjing Agricultural University 37:91–96 (in Chinese)
Wang J, Liu Q, Feng J, Lv JP, Xie SL (2016) Effect of high-doses pyrogallol on oxidative damage, transcriptional responses and microcystins synthesis in Microcystis aeruginosa TY001 (Cyanobacteria). Ecotoxicol Environ Saf 134:273–279. https://doi.org/10.1016/j.ecoenv.2016.09.010
Wang X, Xiang P, Zhang Y, Wan Y, Lian H (2018) The inhibition of Microcystis aeruginosa by electrochemical oxidation using boron-doped diamond electrode. Environ Sci Pollut Res 25:20631–20639. https://doi.org/10.1007/s11356-018-1977-3
Wu X, Wu H, Chen JR, Ye JY (2013) Effects of allelochemical extracted from water lettuce (Pistia stratiotes Linn.) on the growth, microcystin production and release of Microcystis aeruginosa. Environ Sci Pollut Res 20:8192–8201. https://doi.org/10.1007/s11356-013-1783-x
Yang H, Li QY, Yang LL, Sun T, Li XP, Zhou B, Li JR (2020) The competitive release kinetics and synergistic antibacterial characteristics of tea polyphenols/epsilon-poly-l-lysine hydrochloride core-shell microcapsules against Shewanella putrefaciens. Int J Food Sci Technol 55:3542–3552. https://doi.org/10.1111/ijfs.14687
Yuan YT, Zhang HG, Pan G (2016) Flocculation of cyanobacterial cells using coal fly ash modified chitosan. Water Res 97:11–18. https://doi.org/10.1016/j.watres.2015.12.003
Zha F, Li SG, Chang Y (2008) Preparation and adsorption property of chitosan beads bearing beta-cyclodextrin cross-linked by 1,6-hexamethylene diisocyanate. Carbohydr Polym 72:456–461. https://doi.org/10.1016/j.carbpol.2007.09.013
Zhang SL, Zhang B, Dai W, Zhang XM (2011) Oxidative damage and antioxidant responses in Microcystis aeruginosa exposed to the allelochemical berberine isolated from golden thread. J Plant Physiol 168:639–643. https://doi.org/10.1016/j.jplph.2010.10.005
Zhang S, Xia W, Yang X, Zhang T (2016) Inhibition effect of aquaculture water of Salvinia natans (L.) All. on Microcystis aeruginosa PCC7806. Wei sheng yan jiu = Journal of Hygiene Research 45:81–86. https://doi.org/10.19813/j.cnki.weishengyanjiu.2016.01.013. (in Chinese)
Zhu XQ, Dao GH, Tao Y, Zhan XM, Hu HY (2021) A review on control of harmful algal blooms by plant-derived allelochemicals. J Hazard Mater 401:11. https://doi.org/10.1016/j.jhazmat.2020.123403
Zischka H, Larochette N, Hoffmann F, Hamoller D, Jagemann N, Lichtmannegger J, Jennen L, Muller-Hocker J, Roggel F, Gottlicher M, Vollmar AM, Kroemer G (2008) Electrophoretic analysis of the mitochondrial outer membrane rupture induced by permeability transition. Anal Chem 80:5051–5058. https://doi.org/10.1021/ac800173r
Acknowledgements
The authors acknowledge the financial support received from the National Natural Science Foundation of China (Grant Nos. 51779079,51979137) and the Natural Science Foundation of Jiangsu Province (Grant No. BK20181313); the project was Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD) and the Top-notch Academic Programs Project of Jiangsu Higher Education Institutions (TAPP).
Funding
All sources of funding for the research reported have been declared in the “Acknowledgements” section.
Author information
Authors and Affiliations
Contributions
Lixiao Ni: conceptualization, methodology, investigation writing—original draft, and funding acquisition. Jiajia Wang: investigation writing—original draft, investigation, and writing—review and editing. Yuanyi Fang: formal analysis and investigation. Chengjie Zhu: data curation and investigation. Jakpa Wizi: data curation and writing—review and editing. Zhiyun Jiang: validation and data curation. Cunhao Du: writing—review and editing and visualization. Shiyin Li: conceptualization, supervision, project administration, and resources. Xuqing Chen: resources. Jian Xu: resources. Hua Su: resources.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Harmful algal blooms, especially Microcystis aeruginosa blooms, can induce several negative impacts to the aquatic ecosystem.
• As an effective allelochemical, tea polyphenols were used to prepared sustained-release particles with nano-size and high encapsulation efficiency.
• Tea polyphenols sustained-release particles (TPSPs) had the optimal dose (0.3 g/L) with high inhibitory ratio and less cost.
• TPSPs affected the photosynthesis, caused oxidative stress, disrupted antioxidant systems, and destroyed the cell membrane of cyanobacteria cells.
• TPSPs can provide a theoretical basis for the practical application of environmentally friendly sustained-release particles for algal inhibition.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ni, L., Wang, J., Fang, Y. et al. An innovative strategy to control Microcystis growth using tea polyphenols sustained-release particles: preparation, characterization, and inhibition mechanism. Environ Sci Pollut Res 30, 43113–43125 (2023). https://doi.org/10.1007/s11356-023-25255-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-25255-7