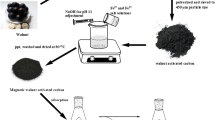
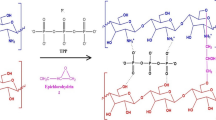
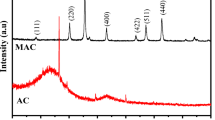
Abstract
Adsorption is a promising technology for removing several contaminants from aqueous matrices. In the last years, researchers worldwide have been working on developing composite adsorbents to overcome some limitations and drawbacks of conventional adsorbent materials, which depend on various factors, including the characteristics of the adsorbents. Therefore, it is essential to characterize the composite adsorbents to describe their properties and structure and elucidate the mechanisms, behavior, and phenomenons during the adsorption process. In this sense, this work aimed to review the main methods used for composite adsorbent characterization, providing valuable information on the importance of these techniques in developing new adsorbents. In this paper, we reviewed the following methods: X-Ray diffraction (XRD); spectroscopy; scanning electron microscopy (SEM); N2 adsorption/desorption isotherms (BET and BJH methods); thermogravimetry (TGA); point of zero charge (pHPZC); elemental analysis; proximate analysis; swelling and water retention capacities; desorption and reuse.
Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ahmadian M, Anbia M, Rezaie M (2020) Sulfur dioxide removal from flue gas by supported CuO nanoparticle adsorbents. Ind Eng Chem Res 59(50):21642–21653. https://doi.org/10.1021/acs.iecr.0c05629
Ahmedna M, Marshall WE, Rao RM (2000) Granular activated carbons from agricultural by-products: preparation, properties, and application in cane sugar refining. LSU Agricultural Experiment Station Reports 456. http://digitalcommons.lsu.edu/agexp/456
Alver E, Metin AÜ, Brouers F (2020) Methylene blue adsorption on magnetic alginate/rice husk bio-composite. Int J Biol Macromol 154:103–113. https://doi.org/10.1016/j.ijbiomac.2020.02.330
Amenabar I, Poly S, Goikoetxea M, Nuansing W, Lasch P, Hillenbrand R (2017) Hyperspectral infrared nanoimaging of organic samples based on Fourier transform infrared nanospectroscopy. Nat Commun 8:14402. https://doi.org/10.1038/ncomms14402
Arumugam TK, Krishnamoorthy P, Rajagopalan NR, Nanthini S, Vasudevan D (2019) Removal of malachite green from aqueous solutions using a modified chitosan composite. Int J Biol Macromol 128:655–664. https://doi.org/10.1016/j.ijbiomac.2019.01.185
Asadu CO, Anthony EC, Elijah OC, Ike IS, Onoghwarite OE, Okwudili UE (2021) Development of an adsorbent for the remediation of crude oil polluted water using stearic acid grafted coconut husk (Cocos nucifera) composite. Appl Surf Sci 6:100179. https://doi.org/10.1016/j.apsadv.2021.100179
Ashiq A, Sarkar B, Adassooriya N, Walpita J, Rajapaksha AU, Ok YS, Vithanage M (2019) Sorption process of municipal solid waste biochar-montmorillonite composite for ciprofloxacin removal in aqueous media. Chemosphere 236:124384. https://doi.org/10.1016/j.chemosphere.2019.124384
Askalany AA, Henninger SK, Ghazy M, Saha BB (2017) Effect of improving thermal conductivity of the adsorbent on performance of adsorption cooling system. Appl Therm Eng 110:695–702. https://doi.org/10.1016/j.applthermaleng.2016.08.075
Bardestani R, Patience GS, Kaliaguine S (2019) Experimental methods in chemical engineering: specific surface area and pore size distribution measurements—BET, BJH, and DFT. Can J Chem Eng 97(11):2781–2791. https://doi.org/10.1002/cjce.23632
Barrett EP, Joyner LG, Halenda PP (1951) The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J Am Chem Soc 73(1):373–380
Benhalima T, Ferfera-Harrar H (2019) Eco-friendly porous carboxymethyl cellulose/dextran sulfate composite beads as reusable and efficient adsorbents of cationic dye methylene blue. Int J Biol Macromol 132:126–141. https://doi.org/10.1016/j.ijbiomac.2019.03.164
Bergaya F, Lagaly G (2013) Handbook of clay science, vol 5, 2nd edn. Hardcover
Bish DL, Reynolds RC (1989) Sample preparation for x-ray diffraction. Modern Powder Diffr 73–100. https://doi.org/10.1515/9781501509018-007
De Blasio C (2019) Thermogravimetric Analysis (TGA). In: De Blasio C (Ed) Fundamentals of biofuels engineering and technology, Green Energy and Technology, pp 91–102. https://doi.org/10.1007/978-3-030-11599-9_7
Bottom R (2008) Thermogravimetric Analysis. In: Gabbott P (ed) Principles and applications of thermal analysis, Blackwell Publishing Ltd, pp. 88–118. https://doi.org/10.1002/9780470697702.ch3
Brown ME (1979) Determination of purity by differential scanning calorimetry (DSC). J Chem Educ 56(5):310. https://doi.org/10.1021/ed056p310
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60(2):309–319. https://doi.org/10.1021/ja01269a023
Bunaciu AA, UdriŞTioiu EG, Aboul-Enein HY (2015) X-ray diffraction: instrumentation and applications. Crit Rev Anal Chem 45(4):289–299. https://doi.org/10.1080/10408347.2014.949616
Chauhan M, Saini VK, Suthar S (2020) Ti-pillared montmorillonite clay for adsorptive removal of amoxicillin, imipramine, diclofenac-sodium, and paracetamol from water. J Hazard Mater 399:122832. https://doi.org/10.1016/j.jhazmat.2020.122832
Chen Y, Ma Y, Lu W, Guo Y, Zhu Y, Lu H, Song Y (2018) Environmentally friendly gelatin/β-cyclodextrin composite fiber adsorbents for the efficient removal of dyes from wastewater. Molecules 23(10):1–17. https://doi.org/10.3390/2Fmolecules23102473
Chen H, Zheng K, Zhu A, Meng Z, Li W, Qin C (2020) Preparation of bentonite/chitosan composite for bleaching of deteriorating transformer oil. Polymers 12(1):60. https://doi.org/10.3390/polym12010111060
Cuccarese M, Brutti S, De Bonis A, Teghil R, Mancini IM, Masi S, Caniani D (2021) Removal of diclofenac from aqueous solutions by adsorption on thermo-plasma expanded graphite. Sci Rep 11(1):1–15. https://doi.org/10.1038/s41598-021-83117-z
Davranche M, Lacour S, Bordas F, Bollinger JC (2003) An easy determination of the surface chemical properties of simple and natural solids. J Chem Educ 80(1):76. https://doi.org/10.1021/ed080p76
De Leon A, Tiu B, Mangadlao J, Pangilinan K, Cao P, Advincula R (2014) Applications of Fourier transform infrared (FTIR) imaging. In: Gauglitz G, Moore DS (eds) Handbook of Spectroscopy. Wiley Online Library Hoboken, Weinheim, pp 1179–1200. https://doi.org/10.1002/9783527654703.ch31
De Rossi A, Rigueto CVT, Dettmer A, Colla LM, Piccin JS (2020) Synthesis, characterization, and application of Saccharomyces cerevisiae/alginate composites beads for adsorption of heavy metals. J Environ Chem Eng 8(4):104009. https://doi.org/10.1016/j.jece.2020.104009
Dotto GL, McKay G (2020) Current scenario and challenges in adsorption for water treatment. J Environ Chem Eng 8:103988. https://doi.org/10.1016/j.jece.2020.103988
Ebele AJ, Oluseyi T, Drage DS, Harrad S, Abdallah MAE (2020) Occurrence, seasonal variation and human exposure to pharmaceuticals and personal care products in surface water, groundwater and drinking water in Lagos State, Nigeria. Emerg Contam 6:124–132. https://doi.org/10.1016/j.emcon.2020.02.004
Edathil AA, Kannan P, Banat F (2020) Adsorptive oxidation of sulfides catalysed by δ-MnO2 decorated porous graphitic carbon composite. Environ Pollut 266:115218. https://doi.org/10.1016/j.envpol.2020.115218
El-Kousy SM, El-Shorbagy HG, El-Ghaffar MAA (2020) Chitosan/montmorillonite composites for fast removal of methylene blue from aqueous solutions. Mater Chem Phys 254:123236. https://doi.org/10.1016/j.matchemphys.2020.123236
Eltaweil AS, Elgarhy GS, El-Subruiti GM, Omer AM (2020) Carboxymethyl cellulose/carboxylated graphene oxide composite microbeads for efficient adsorption of cationic methylene blue dye. Int J Biol Macromol 154:307–318. https://doi.org/10.1016/j.ijbiomac.2020.03.122
Faraj SS, Alkizwini RS, Al Juboury MF (2020) Simulate permeable reactive barrier by using a COMSOL model and comparison with the Thomas, Yoon-Nelson and Clark models for CR dye remediation by composite adsorbent (sewage and waterworks sludge). Water Sci Technol 82(12):2902–2919. https://doi.org/10.2166/wst.2020.500
Farhadi S, Sohrabi MR, Motiee F, Davallo M (2021) Organophosphorus Diazinon pesticide removing from aqueous solution by zero-valent iron supported on biopolymer chitosan: RSM optimization methodology. J Polym Environ 29(1):103–120. https://doi.org/10.1007/s10924-020-01855-z
Fatma S, Asif N, Ahmad R, Fatma T (2020) Toxicity of NSAID drug (paracetamol) to nontarget organism-Nostoc muscorum. Environ Sci Pollut Res 27:35208–35216. https://doi.org/10.1007/s11356-020-09802-0
Fissaha HT, Torrejos REC, Kim H, Chung WJ, Nisola GM (2020) Thia-crown ether functionalized mesoporous silica (SBA-15) adsorbent for selective recovery of gold (Au+3) ions from electronic waste leachate. Microporous Mesoporous Mater 305:110301. https://doi.org/10.1016/j.micromeso.2020.110301
Gao Z, Ma W, Huang S, Hua P, Lan C (2019) Deep learning for super-resolution in a field emission scanning electron microscope. Ai 1(1):1–9. https://doi.org/10.3390/ai1010001
Gauglitz G, Vo-Dihn T (2003) Handbook of spectroscop. Wiley-VCH Verlag GmbH & Co, KGaA, Weinheim. https://doi.org/10.1002/3527602305
Gedam AH, Dongre RS (2016) Activated carbon from Luffa cylindrica doped chitosan for mitigation of lead (II) from an aqueous solution. RSC Adv 6(27):22639–22652. https://doi.org/10.1039/c5ra22580a
Germer TA, Zwinkels JC, Tsai BK (2014) Introduction. In: Germer TA, Zwinkels JC, Tsai BK (Ed). Experimental methods in the physical sciences, Elsevier, pp 1–9. https://doi.org/10.1016/B978-0-12-386022-4.00001-7
Gharanjig H, Gharanjig K, Hosseinnezhad M, Jafari SM (2020) Differential scanning calorimetry (DSC) of nanoencapsulated food ingredients. Characterization of Nanoencapsulated Food Ingredients 295–346. https://doi.org/10.1016/b978-0-12-815667-4.00010-9.
Guillossou R, Roux JL, Goffin A, Mailler R, Varrault G, Vulliet E, Morlay C, Nauleau F, Guérin S, Rocher V, Gaspéri J (2021) Fluorescence excitation/emission matrices as a tool to monitor the removal of organic micropollutants from wastewater effluents by adsorption onto activated carbon. Water Res 190:116749. https://doi.org/10.1016/j.watres.2020.116749
Haas J, Mizaikoff B (2016) Advances in mid-infrared spectroscopy for chemical analysis. Annu Rev Anal Chem 9:45–68. https://doi.org/10.1146/annurev-anchem-071015-041507
Hokkanen S, Sillanpää M (2020) Nano- and microcellulose-based adsorption materials in water treatment Adv Water Treat 1–83. https://doi.org/10.1016/b978-0-12-819216-0.00001-1
Hoseinzadeh H, Hayati B, Ghaheh FS, Seifpanahi-Shabani K, Mahmoodi NM (2021) Development of room temperature synthesized and functionalized metal-organic framework/graphene oxide composite and pollutant adsorption ability. Mater Res Bull 142:111408. https://doi.org/10.1016/j.materresbull.2021.111408
Inkson BJ (2016) Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) for materials characterization. Mater Characterization Using Nondestr Eval (NDE) Methods 17–43. https://doi.org/10.1016/B978-0-08-100040-3.00002-X
Işık B, Kurtoğlu AE, Gürdağ G, Keçeli G (2021) Radioactive cesium ion removal from wastewater using polymer metal oxide composites J Hazard Mater 403. https://doi.org/10.1016/j.jhazmat.2020.123652
Işık B, Uğraşkan V (2021) Adsorption of methylene blue on sodium alginate–flax seed ash beads: Isotherm, kinetic and thermodynamic studies. Int J Biol Macromol 167:1156–1167. https://doi.org/10.1016/j.ijbiomac.2020.11.070
Jawad AH, Abdulhameed AS, Abdallah R, Yaseen ZM (2020) Zwitterion composite chitosan-epichlorohydrin/zeolite for adsorption of methylene blue and reactive red 120 dyes. Int J Biol Macromol 163:756–765. https://doi.org/10.1016/j.ijbiomac.2020.07.014
Kaliva M, Vamvakaki M (2020) Nanomaterials characterization. In: Kaliva M, Vamvakaki M (Ed.), Polymer Science and Nanotechnology, Elsevier, pp 401–433. https://doi.org/10.1016/B978-0-12-816806-6.00017-0
Karimi-Maleh H, Ayati A, Davoodi R, Tanhaei B, Karimi F, Malekmohammadi S, Orooji Y, Fu L, Sillanpää M (2021) Recent advances in using of chitosan-based adsorbents for removal of pharmaceutical contaminants: A review J Clean Prod 125880. https://doi.org/10.1016/j.jclepro.2021.125880
Kasap S, Nostar AE, Oztürk I (2019) Investigation of MnO2 nanoparticles-anchored 3D-graphene foam composites (3DGF-MnO2) as an adsorbent for strontium using the central composite design (CCD) method. New J Chem 43(7):2981–2989. https://doi.org/10.1039/c8nj05283b
Khan MN, Sarwar A (2007) Determination of points of zero charge of natural and treated adsorbents. Surf Rev Lett 14:461–469. https://doi.org/10.1142/S0218625X07009517
Khan SB, Ali F, Kamal T, Anwar Y, Asiri AM, Seo J (2016) CuO embedded chitosan spheres as antibacterial adsorbent for dyes. Int J Biol Macromol 88:113–119. https://doi.org/10.1016/j.ijbiomac.2016.03.026
Kim J, Lee K, Seo BK, Hyun JH (2020) Effective removal of radioactive cesium from contaminated water by synthesized composite adsorbent and its thermal treatment for enhanced storage stability. Environ Res 191:110099. https://doi.org/10.1016/j.envres.2020.110099
Kohli R, Mittal KL (2019) Methods for Assessment and Verification of Cleanliness of Surfaces and Characterization of Surface Contaminants. In: Kohli R, Mittal KL, Developments in Surface Contamination and Cleaning, v. 12, Elsevier. https://doi.org/10.1016/C2017-0-03847-8
Kowalczuk D, Pitucha M (2019) Application of FTIR method for the assessment of immobilization of active substances in the matrix of biomedical materials. Mater 12:2972. https://doi.org/10.3390/2Fma12182972
Kulkarni S, Kaware J (2014) Regeneration and recovery in adsorption-a review. Int J Innov Sci Eng Technol 1:61–65
Ladu F, Mwaffo V, Li J, Macrì S, Porfiri M (2015) Acute caffeine administration affects zebrafish response to a robotic stimulus. Behav Brain Res 289:48–54. https://doi.org/10.1016/j.bbr.2015.04.020
Lessa EF, Nunes ML, Fajardo AR (2018) Chitosan/waste coffee-grounds composite: an efficient and eco-friendly adsorbent for removal of pharmaceutical contaminants from water. Carbohydr Polym 189:257–266. https://doi.org/10.1016/j.carbpol.2018.02.018
Lowell S, Shields JE (2013) Powder surface area and porosity (Vol. 2). Springer Science & Business Media
Lütke SF, Oliveira MLS, Silva LFO, Cadaval TRS Jr, Dotto GL (2020) Nanominerals assemblages and hazardous elements assessment in phosphogypsum from an abandoned phosphate fertilizer industry. Chemosphere 256:127138. https://doi.org/10.1016/j.chemosphere.2020.127138
Ma TS, Gutterson M (1974) Organic elemental analysis. Anal Chem 46(5):437–451. https://doi.org/10.1021/ac60341a032
Machado LDB, Matos JR (2004) Differential thermal analysis and differential scanning calorimetry. In: Canevarolo Junior SV (ed) Polymer characterization techniques, Artliber, pp 229–261
Mahboub MJD, Dubois JL, Cavani F, Rostamizadeh M, Patience GS (2018) Catalysis for the synthesis of methacrylic acid and methyl methacrylate. Chem Soc Rev 47(20):7703–7738. https://doi.org/10.1039/C8CS00117K
Mahmood T, Saddique MT, Naeem A, Westerhoff P, Mustafa S, Alum A (2011) Comparison of different methods for the point of zero charge determination of NiO. Ind Eng Chem Res 50(17):10017–10023. https://doi.org/10.1021/ie200271d
Malek MA, Nakazawa T, Kang HW, Tsuji K, Ro CU (2019) Multi-modal compositional analysis of layered paint chips of automobiles by the combined application of ATR-FTIR imaging, Raman Microspectrometry, and SEM/EDX. Molecules 24:1381. https://doi.org/10.3390/molecules24071381
Manyangadze M, Chikuruwo NHM, Chakra CS, Narsaiah TB, Radhakumari M, Danha G (2020) Enhancing adsorption capacity of nano-adsorbents via surface modification: A review. S Afr J Chem Eng 31(1):25–32. https://doi.org/10.1016/j.sajce.2019.11.003
Melara F, Machado TS, Alessandretti I, Manera C, Perondi D, Godinho M, Piccin JS (2021) Synergistic effect of the activated carbon addition from leather wastes in chitosan/alginate-based composites. Environ Sci Pollut Res 28:48666–48680. https://doi.org/10.1007/s11356-021-14150-8
Mendham J, Denney RC, Barnes JD, Thomas MJK (2000) Vogel’s quantitative chemical analysis, 6th edn. Prentice Hall, New York
Meng F, Schricker S, Brantley W, Mendel D, Rashid R, Fields H Jr, Vig K, Alapati S (2007) Differential scanning calorimetry (DSC) and temperature-modulated DSC study of three mouthguard materials. Dent Mater 23(12):1492–1499. https://doi.org/10.1016/j.dental.2007.01.006
Morhardt C, Ketterer B, Heißler S, Franzreb M (2014) Direct quantification of immobilized enzymes by means of FTIR ATR spectroscopy – A process analytics tool for biotransformations applying non-porous magnetic enzyme carriers. J Mol Catal b: Enzym 107:55–63. https://doi.org/10.1016/j.molcatb.2014.05.018
Nabers A, Ollesch J, Schartner J, Kötting C, Genius J, Haußmann U, Klafki H, Wiltfang J, Gerwert K (2015) An infrared sensor analyzing label-free the secondary structure of the Abeta peptide in presence of complex fluids. J Biophotonics 9:224–234. https://doi.org/10.1002/jbio.201400145
Naderi M (2015) Surface Area: Brunauer-Emmett-Teller (BET). Progr Filtr Sep 585–608. https://doi.org/10.1016/B978-0-12-384746-1.00014-8
Nagpal M, Kakkar R (2020) Facile synthesis of mesoporous magnesium oxide–graphene oxide composite for efficient and highly selective adsorption of hazardous anionic dyes. Res Chem Intermed 46(5):2497–2521. https://doi.org/10.1007/s11164-020-04103-0
Newbury DE, Ritchie NWM (2013) Is scanning electron microscopy/energy dispersive X-ray spectrometry (SEM/EDS) quantitative? Scanning 35(3):141–168. https://doi.org/10.1002/sca.21041
Nguyen DT, Tran HN, Juang RS, Dat ND, Tomul F, Ivanets A, Woo SH, Hosseini-Bandegharaei A, Nguyen VP, Chao HP (2020) Adsorption process and mechanism of acetaminophen onto commercial activated carbon. J Environ Chem Eng 8(6):104408. https://doi.org/10.1016/j.jece.2020.104408
Ngwenya S, Guyo U, Zinyama NP, Chigondo F, Nyamunda BC, Muchanyereyi N (2019) Response surface methodology for optimization of Cd (II) adsorption from wastewaters by fabricated tartaric acid-maize tassel magnetic hybrid sorbent. Biointerface Res Appl Chem 9(4):3996–4005. https://doi.org/10.33263/BRIAC94.996005
Nieto E, Corada-Fernández C, Hampel M, Lara-Martín PA, Sánchez-Argüello P, Blasco J (2017) Effects of exposure to pharmaceuticals (diclofenac and carbamazepine) spiked sediments in the midge, Chironomus riparius (Diptera, Chironomidae). Sci Total Environ 609:715–723. https://doi.org/10.1016/j.scitotenv.2017.07.171
Noh JS, Schwarz JA (1989) Estimation of the point of zero charge of simple oxides by mass titration. J Colloid Interface Sci 130(1):157–164. https://doi.org/10.1016/0021-9797(89)90086-6
Ogbodo NO, Asadu CO, Ezema CA, Onoh MI, Elijah OC, Ike IS, Onoghwarite OE (2021) Preparation and characterization of activated carbon from agricultural waste (Musa-paradisiaca peels) for the remediation of crude oil contaminated water. J Hazard Mater Adv 2:100010. https://doi.org/10.1016/j.hazadv.2021.100010
Osayi JI, Iyuke S, Ogbeide SE (2014) Biocrude production through pyrolysis of used tyres. J Catal 2014:386371. https://doi.org/10.1155/2014/386371
Oshagbemia AA, Autaa M, Kovoa AS, Yahyaa MD, Abdulkareema AS, Ibrahima MA, Obayomib KS (2020) Synthesized chitosan–zeolite composite matrix for the adsorption of chromium (VI) ions. Desalin Water Treat 195:377–388. https://doi.org/10.5004/dwt.2020.25885
Ounphikul B, Chantarasombat N, Hunt AJ, Ngernyen Y (2021) A new low-cost carbon–silica composite adsorbent from a by-product of the sugar industry. Mater Today: Proc 54(5):1884–1887. https://doi.org/10.1016/j.matpr.2021.10.113
Patel JP, Parsania PH (2018) Characterization, testing, and reinforcing materials of biodegradable composites Biodegrad Biocompat Polym Compos 55–79. https://doi.org/10.1016/B978-0-08-100970-3.00003-1
Pavia DL, Lampman GM, Kriz DS, Vyvyan JR (2009) introduction to Spectroscopy: A guide for students of organic chemistry, 4th edn. Thomson Learning Academic Resource Center, Belmont
Peuravuori J, Žbánková P, Pihlaja K (2006) Aspects of structural features in lignite and lignite humic acids. Fuel Process Technol 87(9):829–839. https://doi.org/10.1016/j.fuproc.2006.05.003
Poiana MA, Alexa E, Munteanu MF, Gligor R, Moigradean D, Mateescu C (2015) Use of ATR-FTIR spectroscopy to detect the changes in extra virgin olive oil by adulteration with soybean oil and high temperature heat treatment. Open Chem 13:689–698. https://doi.org/10.1515/chem-2015-0110
Pourebrahimi S, Kazemeini M, Ganji BE, Taheri A (2015) Removal of the CO2 from flue gas utilizing hybrid composite adsorbent MIL-53(Al)/GNP metal-organic framework. Microporous Mesoporous Mater 218:144–152. https://doi.org/10.1016/j.micromeso.2015.07.013
Prime RB, Bair HE, Vyazovkin S, Gallagher PK, Riga A (2009) Thermogravimetric Analysis (TGA). In: Menczel JD, Prime RB (eds) Thermal Analysis of Polymers: Fundamentals and Applications. John Wiley & Sons, Hoboken, pp 241–317. https://doi.org/10.1002/9780470423837.ch3
Pryia SAK, Kaith BS, Tanwar V, Bhatia JK, Sharma N, Bajaj S, Panchal S (2019) RSM-CCD optimized sodium alginate/gelatin based ZnS-nanocomposite hydrogel for the effective removal of biebrich scarlet and crystal violet dyes. Int J Biol Macromol 129:214–226. https://doi.org/10.1016/j.ijbiomac.2019.02.034
Rajeswari A, Christy EJS, Gopi S, Jayaraj K, Pius A (2020) Characterization studies of polymer-based composites related to functionalized filler-matrix interface. In: Goh KL, Aswathi MK, Silva RT, Thomas S (ed) Interfaces in Particle and Fibre Reinforced Composites, Elsevier, pp 219–250. https://doi.org/10.1016/B978-0-08-102665-6.00009-1
Rasoulzadeh H, Sheikhmohammadi A, Abtahi M, Roshan B (2021) Eco-friendly rapid removal of palladium from aqueous solutions using alginate-diatomite magnano composite. J Environ Chem Eng 9(5):105954. https://doi.org/10.1016/j.jece.2021.105954
Rastegar A, Farzadkia M, Jafari JA, Esrafili A (2019) Preparation of Carbon-Alumina (C/Al2O3) aerogel nanocomposite for benzene adsorption from flow gas in fixed bed reactor. MethodsX 6:2476–2483. https://doi.org/10.1016/j.mex.2019.10.019
Rigueto CVT, Nazari MT, Massuda LA, Ostwald BEP, Piccin JS, Dettmer A (2021a) Production and environmental applications of gelatin-based composite adsorbents for contaminants removal: a review. Environ Chem Lett 19:2465–2486. https://doi.org/10.1007/s10311-021-01184-0
Rigueto CVT, Nazari MT, Rosseto M, Massuda LA, Alessandretti I, Piccin JS, Dettmer A (2021b) Emerging contaminants adsorption by beads from chromium (III) tanned leather waste recovered gelatin. J Mol Liq 330:115638. https://doi.org/10.1016/j.molliq.2021.115638
Rigueto CVT, Rosseto M, Nazari MT, Ostwald BEP, Alessandretti I, Manera C, Piccin JS, Dettmer A (2021c) adsorption of diclofenac sodium by composite beads prepared from tannery wastes-derived gelatin and carbon nanotubes. J Environ Chem Eng 9(1):105030. https://doi.org/10.1016/j.jece.2021.105030
Rivera-Jaimes JA, Postigo C, Melgoza-Alemán RM, Aceña J, Barceló D, de Alda ML (2018) Study of pharmaceuticals in surface and wastewater from Cuernavaca, Morelos, Mexico: Occurrence and environmental risk assessment. Sci Total Environ 613:1263–1274. https://doi.org/10.1016/j.scitotenv.2017.09.134
Rodrigues A (2020) Chemical engineering and environmental challenges cyclic adsorption/reaction technologies: materials and process together! J Environ Chem Eng 8:103926. https://doi.org/10.1016/j.jece.2020.103926
Rouquerol J, Llewellyn P, Rouquerol F (2007) Is the BET equation applicable to microporous adsorbents? Stud Surf Sci Catal 160:49–56. https://doi.org/10.1016/s0167-2991(07)80008-5
Rytwo G, Zakai R, Wicklein B (2015) The use of ATR-FTIR spectroscopy for quantification of adsorbed compounds. J Spectrosc 727595. https://doi.org/10.1155/2015/727595
Saadatkhah N, Garcia AC, Ackermann S, Leclerc P, Latifi M, Samih S, Patience GS, Chaouki J (2019) Experimental methods in chemical engineering: thermogravimetric analysis-TGA. Can J Chem Eng 98:34–43. https://doi.org/10.1002/cjce.23673
Saber-Samandari S, Saber-Samandari S, Joneidi-Yekta H, Mohseni M (2017) Adsorption of anionic and cationic dyes from aqueous solution using gelatin-based magnetic nanocomposite beads comprising carboxylic acid functionalized carbon nanotube. Chem Eng J 308:1133–1144. https://doi.org/10.1016/j.cej.2016.10.017
Sanad MMS, Farahat MM, Abdel Khalek MA (2021) One-step processing of low-cost and superb natural magnetic adsorbent: kinetics and thermodynamics investigation for dye removal from textile wastewater. Adv Powder Technol 32:1573–1583. https://doi.org/10.1016/j.apt.2021.03.013
Saraydın D, Işıkver Y, Karadağ E (2018) A Study on the correlation between adsorption and swelling for poly (Hydroxamic Acid) hydrogels-triarylmethane dyes systems. J Polym Environ 26:3924–3936. https://doi.org/10.1007/s10924-018-1257-9
Scheverín N, Fossati A, Horst F, Lassalle V, Jacobo S (2020) Magnetic hybrid gels for emulsified oil adsorption: an overview of their potential to solve environmental problems associated to petroleum spills. Environ Sci Pollut Res 27:861–872. https://doi.org/10.1007/s11356-019-06752-0
Shah IK, Pre P, Alappat BJ (2013) Steam regeneration of adsorbents: an experimental and technical review. Chem Sci Trans 2:1078–1088. https://doi.org/10.7598/cst2013.545
Sharma M, Joshi M, Nigam S, Shree S, Avasthi DK, Adelung R, Srivastava SK, Mishra YK (2019) ZnO tetrapods and activated carbon based hybrid composite: adsorbents for enhanced decontamination of hexavalent chromium from aqueous solution. Chem Eng J 358:540–551. https://doi.org/10.1016/j.cej.2018.10.031
Shukla NB, Rattan S, Madras G (2012) Swelling and dye-adsorption characteristics of an amphoteric superabsorbent polymer. Ind Eng Chem Res 51:14941–14948. https://doi.org/10.1021/ie301839z
Silva AC, Silva VE, Silva BP, Camargo PB, Pereira RC, Barra UM, Botelho AMM, Torrado PV (2013) Lignocellulosic and isotopic composition of vegetation and soil organic matter in a tropical peatland. II - humic substances and humification processes. Rev Bras Cienc Solo 37:134–144. https://doi.org/10.1590/S0100-06832013000100014
Silva RR, Lucena GN, Machado AF, Freitas GA, Matos AT, Abrahão WAP (2018) Spectroscopic and elementar characterization of humic substances in organic substrates. Comun Sci 9:264–274. https://doi.org/10.14295/cs.v9i2.2734
Silva LFO, Pinto D, Neckel A, Dotto GL, Oliveira MLS (2020) The impact of air pollution on the rate of degradation of the fortress of Florianópolis Island, Brazil. Chemosphere 251:126838. https://doi.org/10.1016/j.chemosphere.2020.126838
Sing KSW (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl Chem 57:603–619. https://doi.org/10.1351/pac198557040603
Sing K (2001) The use of nitrogen adsorption for the characterisation of porous materials. Colloids Surf A 187–188:3–9. https://doi.org/10.1016/S0927-7757(01)00612-4
Soliman EM, Ahmed SA, Fadl AA (2020) Microwave-enforced green synthesis of novel magnetic nano composite adsorbents based on functionalization of wood sawdust for fast removal of calcium hardness from water samples. Water Environ Res 92:2112–2128. https://doi.org/10.1002/wer.1383
Song T, Luo W, Mu J, Cai Y, Wei J, Li H (2019) Preparation of polyacrylic-acid/palygorskite composite particles via inverse-suspension polymerization for efficient separation of Ce3+ from aqueous solution. J Colloid Interface Sci 535:371–379. https://doi.org/10.1016/j.jcis.2018.09.103
Soni P, Sur A, Gaba VK (2022) Experimental Investigation of silica-gel based composite adsorbent for adsorption refrigeration system. Therm Sci Eng Prog 29:101194. https://doi.org/10.1016/j.tsep.2022.101194
Sousa JCG, Ribeiro AR, Barbosa MO, Pereira MFR, Silva AMT (2018) A review on environmental monitoring of water organic pollutants identified by EU guidelines. J Hazard Mater 334:146–162. https://doi.org/10.1016/j.jhazmat.2017.09.058
Speight JG (2015) Proximate Analysis. In: Speight JG (ed) Handbook of Coal Analisys, 2nd edn. Wiley, New York, pp 116–142
Spiridon I, Teaca CA, Bodirlau R (2011) Structural changes evidenced by FTIR spectroscopy in cellulosic materials after pre-treatment with ionic liquid and enzymatic hydrolysis. BioResources 6:400–413
Streit AFM, Collazzo GC, Druzian SP, Verdi RS, Foletto EL, Oliveira LFS, Dotto GL (2021) Adsorption of ibuprofen, ketoprofen, and paracetamol onto activated carbon prepared from effluent treatment plant sludge of the beverage industry. Chemosphere 262:128322. https://doi.org/10.1016/j.chemosphere.2020.128322
Stuart BH (2004) Infrared spectroscopy: fundamentals and applications. John Wiley & Sons, Hoboken
Tam NTM, Liu YG, Van Thom N (2020) Magnetic gelatin-activated biochar synthesis from agricultural biomass for the removal of sodium diclofenac from aqueous solution: adsorption performance and external influence. Int J Environ Anal Chem. https://doi.org/10.1080/03067319.2020.1832483
Thirumoorthy K, Krishna SK (2020) Removal of cationic and anionic dyes from aqueous phase by Ball clay - Manganese dioxide nanocomposites. J Environ Chem Eng 8:103582. https://doi.org/10.1016/j.jece.2019.103582
Thompson M (2008) CHNS elemental analysers. Analytical methods committee technical briefs. The Royal Society of Chemistry, United Kingdown. https://www.rsc.org/images/CHNS-elemental-analysers-technical-brief-29_tcm18-214833.pdf
Tran S, Fulcher N, Nowicki M, Desai P, Tsang B, Facciol A, Chow H, Gerlai R (2017) Time-dependent interacting effects of caffeine, diazepam, and ethanol on zebrafish behaviour. Prog in Neuro-Psychopharmacol Biol Psychiatry 75:16–27. https://doi.org/10.1016/j.pnpbp.2016.12.004
Valand R, Tanna S, Lawson G, Bengtström L (2020) A review of Fourier Transform Infrared (FTIR) spectroscopy used in food adulteration and authenticity investigations. Food Addit Contam Part A 37:19–38. https://doi.org/10.1080/19440049.2019.1675909
Valentini A, Carreño NLV, Probst LFD, Leite ER, Longo E (2004) Synthesis of Ni nanoparticles in microporous and mesoporous Al and Mg oxides. Microporous Mesoporous Mater 68:151–157. https://doi.org/10.1016/j.micromeso.2003.12.021
Verdonck E, Schaap K, Thomas LC (1999) A discussion of the principles and applications of Modulated Temperature DSC (MTDSC). Int J Pharm 192:3–20. https://doi.org/10.1016/S0378-5173(99)00267-7
Vernon-Parry KD (2000) Scanning electron microscopy: an introduction. III-Vs Review 13:40–44. https://doi.org/10.1016/S0961-1290(00)80006-X
Villar A, Gorritxategi E, Aranzabe E, Fernández S, Otaduy D, Fernández LA (2012) Low-cost visible–near infrared sensor for on-line monitoring of fat and fatty acids content during the manufacturing process of the milk. Food Chem 135:2756–2760. https://doi.org/10.1016/j.foodchem.2012.07.074
Vishwakarma V, Uthaman S (2020) Environmental impact of sustainable green concrete. In: Liew MS, Nguyen-Tri P, Nguyen TA, Kakooei S (eds) Smart Nanoconcretes and Cement-Based Materials: Properties, Modelling and Applications. Elsevier, Amsterdam, pp 241–255. https://doi.org/10.1016/B978-0-12-817854-6.00009-X
Vyazovkin S, Burnham AK, Criado JM, Pérez-Maqueda LA, Popescu C, Sbirrazzuoli N (2011) ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta 520:1–19. https://doi.org/10.1016/j.tca.2011.03.034
Wagner M (2018) Thermogravimetric Analysis. In: Wagner M (ed) Thermal Analysis in Practice. Hanser Publications, Liberty Twp, pp 162–186
Wang S, Gao B, Zimmerman AR, Li Y, Ma L, Harris WG, Migliaccio KW (2015) Removal of arsenic by magnetic biochar prepared from pinewood and natural hematite. Bioresour Technol 175:391–395. https://doi.org/10.1016/j.biortech.2014.10.104
Wang W, Lu T, Chen Y, Tian G, Sharma VK, Zhu Y, Zong L, Wang A (2019) Mesoporous silicate/carbon composites derived from dye-loaded palygorskite clay waste for efficient removal of organic contaminants. Sci Total Environ 696:133955. https://doi.org/10.1016/j.scitotenv.2019.133955
Wang RF, Deng LG, Li K, Fan XJ, Li W, Lu HQ (2020) Fabrication and characterization of sugarcane bagasse–calcium carbonate composite for the efficient removal of crystal violet dye from wastewater. Ceram Int 46:27484–27492. https://doi.org/10.1016/j.ceramint.2020.07.237
Wang Y, He L, Dang G, Li H, Li X (2021) Polypyrrole-functionalized magnetic Bi2MoO6 nanocomposites as a fast, efficient and reusable adsorbent for removal of ketoprofen and indomethacin from aqueous solution. J Colloid Interface Sci 592:51–65. https://doi.org/10.1016/j.jcis.2021.02.033
Wen J, Arthur K, Chemmalil L, Muzammil S, Gabrielson J, Jiang Y (2012) Applications of differential scanning calorimetry for thermal stability analysis of proteins: qualification of DSC. J Pharm Sci 101:955–964. https://doi.org/10.1002/jps.22820
Wendlandt WW (1986) Thermal analysis. John Wiley & Sons, New York
Wu M, Chen W, Mao Q, Bai Y, Ma H (2019) Facile synthesis of chitosan/gelatin filled with graphene bead adsorbent for orange II removal. Chem Eng Res Des 144:35–46. https://doi.org/10.1016/j.cherd.2019.01.027
Yaah VBK, Ojala S, Khallok H, Laitinen T, Selent M, Zhao H, Sliz R, de Oliveira SB (2021) Development and characterization of Composite Carbon Adsorbents with Photocatalytic Regeneration Ability: Application to Diclofenac Removal from Water. Catalysts 11:173. https://doi.org/10.3390/catal11020173
Yadav LDS (2005) Organic spectroscopy. Springer Science & Business Media, Dordrecht
Yang SC, Liao Y, Karthikeyan KG, Pan XJ (2021) Mesoporous cellulose-chitosan composite hydrogel fabricated via the co-dissolution-regeneration process as biosorbent of heavy metals. Environ Pollut 286:117324. https://doi.org/10.1016/j.envpol.2021.117324
Yıldız H, Tokalıoğlu Ş, Soykan C (2021) Preparation of polyacrylonitrile/polyindole conducting polymer composite and its use for solid phase extraction of copper in a certified reference material. Spectrochim Acta A Mol Biomol Spectrosc 244:118826. https://doi.org/10.1016/j.saa.2020.118826
Zhang B, Wang X, Li S, Liu Y, An Y, Zheng X (2019a) Preferable adsorption of nitrogen and phosphorus from agricultural wastewater using thermally modified zeolite-diatomite composite adsorbent. Water 11:2053. https://doi.org/10.3390/w11102053
Zhang D, Li N, Cao S, Liu X, Qiao M, Zhang P, Zhao Q, Song L, Huang X (2019b) A layered chitosan/graphene oxide sponge as reusable adsorbent for removal of heavy metal ions. Chem Res Chin Univ 35:463–470. https://doi.org/10.1007/s40242-019-8369-1
Zhang M, Chen J, Zhao F, Zeng B (2020) Determination of fluoroquinolones in foods using ionic liquid modified Fe3O4/MWCNTs as the adsorbent for magnetic solid phase extraction coupled with HPLC. Anal Methods 12:4457–4465. https://doi.org/10.1039/D0AY01045F
Zhao B, Jiang H, Lin Z, Xu S, Xie J, Zhang A (2019) Preparation of acrylamide/acrylic acid cellulose hydrogels for the adsorption of heavy metal ions. Carbohydr Polym 224:115022. https://doi.org/10.1016/j.carbpol.2019.115022
Zhou W, Apkarian R, Wang ZL, Joy D (2007) Fundamentals of scanning electron microscopy (SEM). In: Zhou W, Wang ZL (eds) Scanning Microscopy for Nanotechnology: Techniques and Applications. Springer, New York, pp 1–40. https://doi.org/10.1007/978-0-387-39620-0_1
Zhou Y, Lu J, Zhou Y, Liu Y (2019) Recent advances for dyes removal using novel adsorbents: a review. Environ Pollut 252:352–365
Zhou RY, Yu JX, Li HX, Chi RA (2020) Removal of phosphate from aqueous solution by ferrihydrite/bagasse composite prepared through in situ precipitation method. Colloids and Surf a: Physicochem Eng Asp 603:125144. https://doi.org/10.1016/j.colsurfa.2020.125144
Funding
This study was financed partly by the National Council for Scientific and Technological (CNPq)—Projects Codes 403658/2020–9 and 140541/2021–7 and Coordination for the Improvement of Higher Education Personnel (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Contributions
Conceptualization: M. T. Nazari, J. S. Piccin, G. L. Dotto; methodology: C. V. T. Rigueto, I. Alessandretti; formal analysis and investigation: F. Melara, N. F. da Silva; writing—original draft preparation: M. T. Nazari, J. S. Piccin, L. Crestani, V. Ferrari; writing—review and editing: C. Schnorr, G. L. Dotto, J. Vieillard, L. F. O. Silva; funding acquisition: J. S. Piccin; supervision: J. S. Piccin. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nazari, M.T., Schnorr, C., Rigueto, C.V.T. et al. A review of the main methods for composite adsorbents characterization. Environ Sci Pollut Res 29, 88488–88506 (2022). https://doi.org/10.1007/s11356-022-23883-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23883-z