Abstract
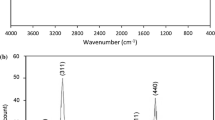
In the present study, a composite was synthesized by the impregnation of activated carbon with magnetite (Fe3O4) using a simple co-precipitation method. Several characterizations were performed, such as X-ray diffraction (XRD), scanning electron microscopy (SEM), point of zero charge (PZC), specific surface area, and Fourier transform infrared spectroscopy (FTIR). The magnetic activated carbon composite (MAC) was used as a new adsorbent for magnetic solid phase extraction (MSPE) of 2,4-dichlorophenol (2,4-DCP) in water samples. The main experimental parameters, such as adsorbent mass, pH, adsorbent/adsorbent contact time, volume and type of desorbent, sample volume, and desorption time were optimized. The method showed linearity in the investigated concentration range of 1 μg mL−1–6 μg mL−1 (R2 = 0.999). The limit of detection (LOD) and the limit of quantification (LOQ) were 0.293 μg mL−1 and 0.890 μg mL−1, respectively. The recoveries for the water samples ranged from 50.0% to 55.0% and the relative standard deviation (RSD) was less than 4.8%. The CAM presented application to MSPE and the magnetic properties inserted in the activated carbon (AC) contributed for the fast and easy removal of the adsorbent from the reaction medium. Thus, the proposed method proved to be easy, efficient, and environmentally friendly due to low solvent consumption.







Similar content being viewed by others
Abbreviations
- Fe3O4 :
-
magnetite
- XRD:
-
X-ray diffraction
- SEM:
-
scanning electron microscopy
- PZC:
-
point of zero charge
- BET method:
-
specific surface area
- FTIR:
-
Fourier transform infrared spectroscopy
- MAC:
-
magnetic activated carbon
- MSPE:
-
magnetic solid phase extraction
- 2,4 DCP:
-
2,4-dichlorophenol
- LOD:
-
limit of detection
- LOQ:
-
limit of quantification
- RSD:
-
relative standard deviation
- AC:
-
activated carbon
- 2,4 D:
-
2,4-dichlorophenoxyacetic acid
- EPA:
-
United States Environmental Protection Agency
- EU:
-
European Union
- MNPs:
-
magnetic nanoparticles
- FeSO4.7H2O:
-
iron sulfate heptahydrate
- KOH:
-
potassium hydroxide
- KNO3 :
-
potassium nitrate
- EMBRAPA:
-
Brazilian Agricultural Research Corporation
- HPLC:
-
High performance liquid chromatograph
- ACN:
-
acetonitrile
- RL :
-
constant dimensionless separation factor
- SPE:
-
solid phase extraction
- SPME:
-
micro-solid phase extraction
References
Alalm, M. G., Samy, M., Ookawara, S., & Ohno, T. (2018). Immobilization of S-TiO2 on reusable aluminum plates by polysiloxane for photocatalytic degradation of 2, 4-dichlorophenol in water. Journal of Water Process Engineering, 26, 329–335.
Aziz, K. H. H., Miessner, H., Mueller, S., Mahyar, A., Kalass, D., Moeller, D., & Rashid, M. A. M. (2018). Comparative study on 2, 4-dichlorophenoxyacetic acid and 2, 4-dichlorophenol removal from aqueous solutions via ozonation, photocatalysis and non-thermal plasma using a planar falling film reactor. Journal of Hazardous Materials, 343, 107–115.
Badruddoza, A. Z. M., Shawon, Z. B. Z., Tay, W. J. D., Hidajat, K., & Uddin, M. S. (2013). Fe3O4/cyclodextrin polymer nanocomposites for selective heavy metals removal from industrial wastewater. Carbohydrate Polymers, 91(1), 322–332.
Castro, C. S., Guerreiro, M. C., Gonçalves, M., Oliveira, L. C., & Anastácio, A. S. (2009). Activated carbon/iron oxide composites for the removal of atrazine from aqueous medium. Journal of Hazardous Materials, 164(2–3), 609–614.
Chahkandi, M., Amiri, A., & Arami, S. R. S. (2019). Extraction and preconcentration of organophosphorus pesticides from water samples and fruit juices utilizing hydroxyapatite/Fe3O4 nanocomposite. Microchemical Journal, 144, 261–269.
Chen, Y., Zhang, W., Zhang, Y., Deng, Z., Zhao, W., Du, H., et al. (2018). In situ preparation of core–shell magnetic porous aromatic framework nanoparticles for mixed–mode solid–phase extraction of trace multitarget analytes. Journal of Chromatography A, 1556, 1–9.
Chu, M., Hu, K., Wang, J., Liu, Y., Ali, S., Qin, C., et al. (2019). Synthesis of g-C3N4-based photocatalysts with recyclable feature for efficient 2, 4-dichlorophenol degradation and mechanisms. Applied Catalysis B: Environmental, 243, 57–65.
Dalida, M. L. P., Mariano, A. F. V., Futalan, C. M., Kan, C. C., Tsai, W. C., & Wan, M. W. (2011). Adsorptive removal of Cu (II) from aqueous solutions using non-crosslinked and crosslinked chitosan-coated bentonite beads. Desalination, 275(1–3), 154–159.
Eurachem/CITAC Guide. (2014). Guide to quality in analytical chemistry: an aid to accreditation. Disponível em. www.eurachem.org.
Filippou, O., Deliyanni, E. A., & Samanidou, V. F. (2017). Fabrication and evaluation of magnetic activated carbon as adsorbent for ultrasonic assisted magnetic solid phase dispersive extraction of bisphenol a from milk prior to high performance liquid chromatographic analysis with ultraviolet detection. Journal of Chromatography A, 1479, 20–31.
Filizola, H. F., Gomes, M. A. F., & De Souza, M. D. (2006). Manual of sampling procedures in agricultural areas for environmental quality analysis: soil, water and sediment. Jaguariúna: Embrapa Environment.
Gao, F., Lu, W., Liu, H., Li, J., & Chen, L. (2018). Dispersive liquid–liquid microextraction of five chlorophenols in water samples followed by determination using capillary electrophoresis. Electrophoresis, 39(19), 2431–2438.
Guilarduci, V. V. D. S., de Mesquita, J. P., Martelli, P. B., & De Fátima, G. H. (2006). Adsorption pf phenol on activated carbono na alcaline médium. Quimica Nova, 29(6), 1226–1232.
Ha, D. D. (2018). Anaerobic degradation of 2, 4-dichlorophenoxyacetic acid by Thauera sp. DKT. Biodegradation, 29(5), 499–510.
Hu, L., Zhou, T., Luo, D., Feng, J., Tao, Y., Zhou, Y., et al. (2019). Bioaccumulation of tetrabromobisphenol A in a laboratory-based fish–water system based on selective magnetic molecularly imprinted solid-phase extraction. Science of the Total Environment, 650, 1356–1362.
Iyengar, S. J., Joy, M., Ghosh, C. K., Dey, S., Kotnala, R. K., & Ghosh, S. (2014). Magnetic, X-ray and Mössbauer studies on magnetite/maghemite core–shell nanostructures fabricated through an aqueous route. RSC Advances, 4(110), 64919–64929.
Jeirani, Z., Niu, C. H., & Soltan, J. (2017). Adsorption of emerging pollutants on activated carbon. Reviews in Chemical Engineering, 33(5), 491–522.
Kwon, J. H., Wilson, L. D., & Sammynaiken, R. (2014). Synthesis and characterization of magnetite and activated carbon binary composites. Synthetic Metals, 197, 8–17.
Kyzas, G. Z., Deliyanni, E. A., & Lazaridis, N. K. (2014). Magnetic modification of microporous carbon for dye adsorption. Journal of Colloid and Interface Science, 430, 166–173.
Li, X. S., Xu, L. D., Shan, Y. B., Yuan, B. F., & Feng, Y. Q. (2012). Preparation of magnetic poly (diethyl vinylphosphonate-co-ethylene glycol dimethacrylate) for the determination of chlorophenols in water samples. Journal of Chromatography A, 1265, 24–30.
Li, N., Chen, J., & Shi, Y. P. (2015). Magnetic graphene solid-phase extraction for the determination of carbamate pesticides in tomatoes coupled with high performance liquid chromatography. Talanta, 141, 212–219.
Li, Q. L., Huang, F., Wang, X. L., Wang, X., & Zhao, R. S. (2017a). Multiple-helix cobalt (II)-based metal-organic nanotubes on stainless steel fibers for solid-phase microextraction of chlorophenol and nitrophenols from water samples. Microchimica Acta, 184(6), 1817–1825.
Li, S., Liu, L., Yu, Y., Wang, G., Zhang, H., & Chen, A. (2017b). Fe3O4 modified mesoporous carbon nanospheres: magnetically separable adsorbent for hexavalent chromium. Journal of Alloys and Compounds, 698, 20–26.
Limchoowong, N., Sricharoen, P., Areerob, Y., Nuengmatcha, P., Sripakdee, T., Techawongstien, S., et al. (2017). Preconcentration and trace determination of copper (II) in Thai food recipes using Fe3O4@ chi–GQDs nanocomposites as a new magnetic adsorbent. Food Chemistry, 230, 388–397.
Liu, F., Zhou, K., Chen, Q., Wang, A., & Chen, W. (2019). Application of magnetic ferrite nanoparticles for removal of cu (II) from copper-ammonia wastewater. Journal of Alloys and Compounds, 773, 140–149.
Lopes, W. A., & Fascio, M. (2004). Scheme for the interpretation of spectra of organic substances in the infrared region. Química nova, 27(4), 670–673.
Mahmoud, M. E., Ahmed, S. B., Osman, M. M., & Abdel-Fattah, T. M. (2015). A novel composite of nanomagnetite-immobilized-baker’s yeast on the surface of activated carbon for magnetic solid phase extraction of Hg (II). Fuel, 139, 614–621.
Meng, J., Shi, C., Wei, B., Yu, W., Deng, C., & Zhang, X. (2011). Preparation of Fe3O4@ C@ PANI magnetic microspheres for the extraction and analysis of phenolic compounds in water samples by gas chromatography–mass spectrometry. Journal of Chromatography A, 1218(20), 2841–2847.
Özdemir, S., Mohamedsaid, S. A., Kılınç, E., & Soylak, M. (2019). Magnetic solid phase extractions of Co (II) and Hg (II) by using magnetized C. micaceus from water and food samples. Food Chemistry, 271, 232–238.
Pashaei, Y., Ghorbani-Bidkorbeh, F., & Shekarchi, M. (2017). Superparamagnetic graphene oxide-based dispersive-solid phase extraction for preconcentration and determination of tamsulosin hydrochloride in human plasma by high performance liquid chromatography-ultraviolet detection. Journal of Chromatography A, 1499, 21–29.
Pokryshkin, S. A., Kosyakov, D. S., Kozhevnikov, A. Y., Lakhmanov, D. E., & Ul’yanovskii, N. V. (2018). Highly sensitive determination of chlorophenols in sea water by gas chromatography−tandem mass spectrometry. Journal of Analytical Chemistry, 73(10), 991–998.
Rey, A., Hungria, A. B., Duran-Valle, C. J., Faraldos, M., Bahamonde, A., Casas, J. A., et al. (2016). On the optimization of activated carbon-supported iron catalysts in catalytic wet peroxide oxidation process. Applied Catalysis B: Environmental, 181, 249–259.
Ribani, M., Bottoli, C. B. G., Collins, C. H., Jardim, I. C. S. F., & Melo, L. F. C. (2004). Validation for chromatographic and electrophoretic methods. Química Nova, 27(5), 771–780.
Silva, A. K., Viana, L. H., Lanças, F. M., & Nazario, C. E. D. (2016a). Extração em fase sólida magnética (MSPE): fundamentos e aplicações. Scientia Chromatographica, 8(4), 239–256.
Silva, M. C., Torres, J. A., Nogueira, F. G., Tavares, T. S., Corrêa, A. D., Oliveira, L. C., & Ramalho, T. C. (2016b). Immobilization of soybean peroxidase on silica-coated magnetic particles: a magnetically recoverable biocatalyst for pollutant removal. RSC Advances, 6(87), 83856–83863.
Simonin, J. P. (2016). On the comparison of pseudo-first order and pseudo-second order rate laws in the modeling of adsorption kinetics. Chemical Engineering Journal, 300, 254–263.
Tian, N., Tian, X., Nie, Y., Yang, C., Zhou, Z., & Li, Y. (2019). Enhanced 2, 4-dichlorophenol degradation at pH 3–11 by peroxymonosulfate via controlling the reactive oxygen species over Ce substituted 3D Mn2O3. Chemical Engineering Journal, 355, 448–456.
Tong, Y., Liu, X., & Zhang, L. (2019). Green construction of Fe3O4@ GC submicrocubes for highly sensitive magnetic dispersive solid-phase extraction of five phthalate esters in beverages and plastic bottles. Food Chemistry, 277, 579–585.
Torres, J. A., Silva, M. C., Lopes, J. H., Nogueira, A. E., Nogueira, F. G. E., & Corrêa, A. D. (2018). Development of a reusable and sustainable biocatalyst by immobilization of soybean peroxidase onto magnetic adsorbent. International Journal of Biological Macromolecules, 114, 1279–1287.
USEPA. 2014. Drinking water contaminants. National primary drinking water regulations. U.S. Environmental Protection Agency, Office of Water, Office of Ground Water and Drinking Water, Washington, DC. Acesso: 02/10/2019. Disponível em: http://water.epa.gov/drink/contaminants/index.cfm.
Wang, C., Liu, L., Zhang, Z., Wu, Q., & Wang, Z. (2016). Magnetic biomass activated carbon-based solid-phase extraction coupled with high performance liquid chromatography for the determination of phenylurea herbicides in bottled rose juice and water samples. Food Analytical Methods, 9(1), 80–87.
Wang, H., Wang, B., Li, J., & Zhu, T. (2019). Adsorption equilibrium and thermodynamics of acetaldehyde/acetone on activated carbon. Separation and Purification Technology, 209, 535–541.
Yu, B. Y., & Kwak, S. Y. (2010). Assembly of magnetite nanocrystals into spherical mesoporous aggregates with a 3-D wormhole-like pore structure. Journal of Materials Chemistry, 20(38), 8320–8328.
Zhang, L., Zhang, B., Wu, T., Sun, D., & Li, Y. (2015). Adsorption behavior and mechanism of chlorophenols onto organoclays in aqueous solution. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 484, 118–129.
Zhang, B., Liu, W., Sun, D., Li, Y., & Wu, T. (2019). Hollow nanoshell of layered double oxides for removal of 2, 4-dichlorophenol from aqueous solution: synthesis, characterization, and adsorption performance study. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 561, 244–253.
Zhou, R., Zhao, J., Shen, N., Ma, T., Su, Y., & Ren, H. (2018). Efficient degradation of 2, 4-dichlorophenol in aqueous solution by peroxymonosulfate activated with magnetic spinel FeCo2O4 nanoparticles. Chemosphere, 197, 670–679.
Acknowledgments
The authors thank the Conselho Nacional de desenvolvimento científico e tecnológico (CNPq), Fundação de amparo a pesquisa de Minas Gerais (FAPEMIG), and the Chemical Analysis and Prospecting Laboratory–CAPQ from the Federal University of Lavras.
Funding
This study was financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior–Brasil (CAPES)–Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rodrigues, S.C., Silva, M.C., Torres, J.A. et al. Use of Magnetic Activated Carbon in a Solid Phase Extraction Procedure for Analysis of 2,4-dichlorophenol in Water Samples. Water Air Soil Pollut 231, 294 (2020). https://doi.org/10.1007/s11270-020-04610-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-020-04610-1