Abstract
The aim of the research work was to explain the possibilities of application of waste activated sludge (WAS) pretreatment processes prior to anaerobic digestion (mesophilic fermentation). Hydrodynamic disintegration and freezing/thawing disintegration methods were used. Based on the microbiological and parasitological analyses, a significant decrease in pathogenic bacteria, coliphages, and parasite eggs was observed. The number of bacteria analyzed (Salmonella sp., Escherichia coli, Clostridium perfringens) and coliphages were reduced from 19.3to 42.3% after hydrodynamic cavitation. A similar effect was achieved for destruction by freezing/thawing with dry ice between 7.8 and 14.9%. The effectiveness of parasite eggs reduction (Ascaris sp., Trichuris sp., Toxocara sp.) for these disintegration methods ranged from 10.7 to 29.3%. The highest results were observed for the hybrid disintegration method (hydrodynamic cavitation + dry ice disintegration) caused by a synergistic effect. Salmonella sp. in 1 gd.w. decrease about 69.7%, E. coli by 70.0%, Clostridium perfringens by 38.4%, and coliphages by 48.2%. Disruption of WAS by a hybrid method led to a reduction in the number of helminth eggs Ascaris sp. (63.8%), Trichuris sp. (64.3%), and Toxocara sp. (66.4%). After anaerobic digestion under mesophilic conditions, an additional reduction of analyzed bacterial pathogens and helminth eggs were observed. The introduction of hybrid disintegrated WAS to the fermentation chamber resulted in higher efficiency in decrease (from 1 to 23%) in comparison to the control sample (70%WAS + 30%DS (inoculum-digested sludge)).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sewage sludge comes into existence in wastewater treatment plants as a peculiar waste of wastewater treatment processes. The legal regulations regarding sewage sludge management have restrictions on the presence of volatile substances, disease-causing pathogenic organisms (viruses, bacteria, protozoa, and parasite eggs), heavy metals, and inorganic ions, along with dangerous chemicals from industrial wastes, household chemicals, and pesticides (López et al. 2019; Li et al. 2021).
However, sewage sludge has intrinsic fertilizer values. Sludges from the biological process of wastewater treatment are abundant in organic substances, nitrogen, phosphorus, calcium, magnesium, sulfur, and microelements essential to the life of plants and the soil fauna. Exploiting the fertilizer value of sewage deposits has great importance in the case of environmental protection, which is a serious problem. Sewage sludge is increasingly used for fertilizing agricultural lands and forests and for the recultivation of devastated areas. However, a very important technical problem is always presented from the point of view of large hydration, mass and sanitary danger. In addition to the content of heavy metals and toxic substances and microbiological contaminations, the usefulness of sewage deposits for use in farming constitutes the basic criterion. Bacteria, viruses, and parasites are the most commonly transmitted fecal–oral pathogens. These microorganisms are transmitted in the sludge in which they accumulate (Gopi Kumar et al. 2012; Lakshmi et al. 2014; Lopes et al. 2017; Jiang et al. 2020).
With regard to pathogens, the requirements are based on the principle of reducing and eliminating pathogens from sewage sludge destined for agricultural use. In the technological process of wastewater treatment plants, elimination of microorganisms is achieved by appropriate treatment (conditioning) of the sludge, e.g., in thermal processes, which ensures the reduction of the amount of pathogens and the appropriate microbiological quality of the sludge (Hu et al. 2011; Lloret et al. 2012; Zhou et al. 2015; Eng et al. 2015; Wójcik et al. 2017; Luukkonen et al. 2020). The sanitary index describes the content of pathogenic bacteria and eggs of intestinal parasites in the sludge on the basis of indirect conclusions. Not so long ago in Polish law, the species that was a sanitary indicator among bacteria was lactose-positive Escherichia coli. Currently, according to the ordinance of the Minister of the Environment (2015), the presence of the lactose-negative bacteria Salmonella sp. is used as a new indicator.
Sewage sludge is often polluted by many types of helminths. The source of parasite eggs in sewage sludge is human and animal feces, which can also cause soil or food contamination. The parasite indicators Ascaris sp., Trichuris sp., and Toxocara sp. are being used in the sanitary estimation of sewage sludge (Pike et al. 1988; Jiang et al. 2020).
The risk of contamination of the environment with pathogenic microorganisms from sewage sludge causes the constant search for methods of their destruction. Sanitary safe sludge can be used for agricultural purposes. Disintegration of activated sludge and fermentation are the most applied processes for the stabilization and hygienization of sewage sludge. The disintegration of sewage sludge can occur by mechanical, chemical, and biological methods and reduces the flock size, disrupts the cell walls, and releases organic material from the cells to the liquid phase (Doğan and Sanin 2009; Liao et al. 2016; Machnicka et al. 2017; Grübel et al. 2018; Khanh Nguyen et al. 2021). The next step in sludge technology is anaerobic digestion conducted under psychrophilic, mesophilic, thermophilic, and two-step processes. In recent years, most wastewater treatment plants have realized this stabilization under mesophilic conditions. The main benefits of fermentation of sewage sludge are its stabilization and reduction of sludge mass, methane production, hygienization, and improvement of the drainage properties of the digested sludge (Zhen et al. 2017; Liu et al. 2019; Khanh Nguyen et al. 2021; Hoang et al. 2022).
Therefore, removal of pathogens by pretreatment of waste activated sludge (WAS) and anaerobic digestion allows applying sludge as a fertilizer. This paper presents results from the hygienization of WAS by pretreatment and anaerobic digestion (mesophilic fermentation).
Materials and methods
Waste-activated sludge (WAS) was collected (from secondary settling tanks) from a wastewater treatment plant (WWTP) in the Silesian region of Poland, working in according to the Enhanced Biological Nutrient Removal (EBNR) processes.
The test material in two stages was collected (first stage—for hygienization by hydrodynamic cavitation and freezing/thawing process, second stage—for hygienization by anaerobic digestion).
Pre-treatment of WAS
Hydrodynamic disintegration
Hydrodynamic disintegration of WAS was done with the use of a pressure pump (12 bar) equipped with a constructed cavitation nozzle (Grübel and Suschka 2015; Machnicka et al. 2015). Pumping 25 l of WAS through the nozzle took 3 min. The process was underway for 30 min, which corresponded to six multiplicity flow by cavitation nozzle.
Disintegration by freezing/thawing
For destruction of WAS, the dry ice was used in the volume ratios of the WAS to dry ice − 1:1. In our previous work, we dealt with the efficiency of sludge disintegration and then anaerobic digestion supported with the use of dry ice (Nowicka et al. 2014; Nowicka and Machnicka 2015).
Disintegration by hybrid methods (WASDH)
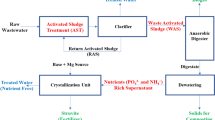
Samples after 30 min of hydrodynamic disintegration and dry ice freezing/thawing process was named as hybrid method (Fig. 1).
The fermentation experiments
The anaerobic digestion experiments were performed in laboratory scale using fermenter chambers (2.5 L). The processes were taking place in thermostatic chamber, with constant temperatures 35 ± 1 °C by 25 days.
The fermentation was performed as follows: fermenter 1 were fed with WAS (70% vol.) and inoculum (DS – digested sludge; 30% vol.). Fermenters 2–8 were fed with (% vol.): 10%WASDH; 20%WASDH, 30%WASDH, 40%WASDH, 50%WASDH, 60%WASDH, and 70%WASDH (Fig. 1).
The dosage of inoculum has been optimized in previous studies in terms of minimizing the lag-phase and decreasing the disturbance of the fermentation process.
Liquid displacement method was used for the daily measurement of produced biogas. Methane concentration in the biogas was measured using GFM416 analyzer (OMC Envag, Poland).
The investigations presented here were repeated 10 times and (mean values, n = 10) arithmetic average and standard deviation were carried out. The standard deviation was determined according to the estimator of the highest credibility in STATISTICA 6.0.
Microbiological analysis
Bacteria and coliphages
The studies related also to the total number of bacteria contained in the WAS before and after the destruction (disintegration by hydrodynamic cavitation and dry ice) and anaerobic digestion (mesophilic fermentation).
Detection and enumeration of Salmonella sp. was according to Holt et al. (1994) with using specific culture media. In order to verify the taxonomic classification of Salmonella sp., the API 20E biochemical test was applied. Detection and enumeration of E. coli and Clostridium perfringens were investigate according to methodology presented in (Grübel and Suschka 2015).
Detection and enumeration of bacteriophages was investigate according to ISO 10705–2:2000.
Helminth parasites
The studies related also to the total number of parasites eggs contained in the WAS before and after each methods of disintegration and fermentation process. In Poland, the parasitological indicators belonging to the evaluation of the sanitary state of soil (i.e., after introduction of WAS) are live eggs of parasites belonging to nematodes of the genera Ascaris spp., Trichuris spp. and Toxocara spp. The methods of their detection and determination in samples have been developed and contained in the Polish Standard No. PN-Z19000-4:2001.
Eggs of Toxocara sp. were being isolated from investigated sample by shaking, centrifugation, filtration of flotation suspension processes, and microscopic observation (Zdybel et al. 2019).
The microscope was used in parasitological studies—Nikon Eclipse TS100 coupled with camera MoticamPro 252A (Conbest, Poland) allowed also for size measurements by a program Images Advanced 3.2. Additionally, counter Scan 500 (Interscience Microbiology, France) were used for counts colonies on Petri dishes.
Results and discussion
The evaluation of a sanitary WAS is based on the inference of the presence of pathogenic bacteria and eggs of intestinal parasites. Its purpose is to detect the so-called sanitary indicator. National and international regulations (European Commission 2000; Council Directive 86/278/EEC; US EPA 2003) dictate that sludge should be stabilized and hygienized before land introduction.
Sewage sludge is characterized by a large number and diversity of microorganisms, including pathogens. The recommended indicators for sanitary assessment of sewage sludge are Salmonella sp., Escherichia coli, fecal coliforms, enteric viruses, sulfate-reducing Clostridia, and parasite eggs. Therefore, it is necessary to look for methods enabling their efficient reduction or total elimination. As a consequence, these techniques allow the possible use of sludge in agriculture. Therefore, the knowledge of selected hygienic indicators in sewage sludge permits making decisions about its use.
The removal of pathogenic bacteria and helminths from waste activated sludge by hydrodynamic destruction and freezing/thawing can help considerably in the reduction of the transfer operation of illnesses. The mechanical process and freezing/thawing can cause the destruction of microorganisms and thus contribute to a partial hygienization of WAS, which was confirmed by the results of the microbiological and parasitological analyses (Figs. 2 and 3).
After hygienization by hydrodynamic cavitation and dry ice, a gradual decrease in the population count of bacteria was observed (Fig. 2). According to Polish law, Salmonella sp. removal is imperative for the agricultural use of sewage sludge (Minister of the Environment 2015). After 30 min of hydrodynamic cavitation of sludge, microorganism disintegration resulted in the elimination of Salmonella sp. from 4.47 log to 4.24 log cfu/gd.w. The number of Salmonella sp. in 1 gd.w. (the volume ratio of WAS to dry ice was 1:1; freezing/thawing) was reduced by approximately 14.5% (Fig. 3).
Depending on disintegration by hydrodynamic destruction and freezing/thawing, a reduction number of E. coli in WAS of approximately 42.3% (from 5.88 log to 5.64 log cfu/gd.w.) and 13.7% (5.88 log to 5.82 log cfu/gd.w.) was observed, respectively (Figs. 2 and 3).
A higher removal efficiency of Escherichia coli during low-frequency ultrasound disintegration was obtained by Hawrylik (2019). She concluded that ultrasound has an effective impact on E. coli, in which a nearly 70% decrease in the amount of bacteria was obtained after 20 min of ultrasound at 40 kHz. Using acidification as a pretreatment method, Fukushi et al. (2003) found an effective reduction of Salmonella spp. in sewage sludge. Methods using chemical oxidants (advanced oxidation processes) have also positive effects in the elimination of microorganisms. After 2 h of reaction of persulfate (0.5 mM) activated by solar energy, a unit reduction of 6.0 log of bacteria in sewage sludge was registered (Ferreira et al. 2020). Luukkonen et al. (2020) used peracetic acid for the conditioning of municipal wastewater sludge. They concluded that with the increase in the dose of peracetic acid to 480 mg/l, a decrease in Salmonella sp. to an amount acceptable for use of such sludge as a fertilizer in Finland occurred. Moreover, using alkalization and ultrasound pretreatment methods on sludge, disintegration deactivations of E. coli and sulfite-reducing Clostridia were obtained (Martín-Díaz et al. 2017). Studies on the amount of energy input with microwaves (up to 7 kWh) into the sludge showed an approximately 5.90 log removal value of the analyzed pathogens (Mawioo et al. 2017).
For an organism to be considered an indicator, it should be characterized by common presence and survival in raw sludge. In addition, it must be easily cultured and easily identified. Therefore, it is difficult to choose one species of bacteria, which is the reason for using several indicator organisms. A potential indicator of the sanitary quality of sewage sludge, as mentioned in the literature (Sidhu and Toze 2009), is Clostridium perfringens. On the basis of microbiological tests, an elimination of Clostridium perfringens bacteria by each method of disintegration was observed. The number of Clostridium perfringens rods in WAS was reduced by 19.6% (after 30 min of disintegration by hydrodynamic cavitation), 7.8% (dry ice), and 38.4% (hybrid disintegration) (Figs. 2 and 3).
Similar effects were obtained for a decrease in the overall number of coliphages (Martín-Díaz et al. 2016). After 30 min of cavitation processes, the number was reduced by approximately 19.3%, while dry ice had a destructive effect of approximately 14.9% (Figs. 2 and 3). After the hybrid process, the number of coliphages changed from 5.77 log to 5.49 log cfu/gd.w. (Figs. 2 and 3).
The indicators of Ascaris sp., Trichuris sp., and Toxocara sp. are also being used in sanitary assessments of sewage sludge. Such indicators allow us to assess the agricultural usefulness of sewage sludge. Unfortunately, there is still a lack of sufficient information on their content in wastewater and sewage sludge and their survival during wastewater treatment processes.
Hydrodynamic disintegration and dry ice destroyed the eggs of helminths. The disintegration of WAS by hydrodynamic cavitation resulted in 26.4%, 22.8%, and 29.3% reductions in Ascaris sp., Trichuris sp., and Toxocara sp., respectively (Figs. 2 and 3). As a result of disintegration by dry ice, the overall egg number decreased. A decrease from 2.08 log to 2.03 log eggs/kgd.w was observed for Ascaris sp., a decrease from 1.96 log to 1.88 log eggs/kgd.w. was observed for Trichuris sp. and a decrease from 3.05 log to 2.34 log eggs/kgd.w. was observed for Toxocara sp. (Figs. 2 and 3). A higher elimination effect was observed after the hybrid process was used.
In connection with the above, it can be clearly stated that the studied methods of disintegration contribute to the partial hygienization of sewage sludge. This is an important aspect because in activated sludge technology, the next stage (in most wastewater treatment plants—WWTPs) is the process of anaerobic stabilization.
The most commonly used process in WWTPs is stabilization under mesophilic conditions, which contributes to the decomposition and degradation of organic matter, biogas production and the diminution of pathogens. This process, unlike thermophilic digestion, has a low efficiency in the scope of sludge hygienization (Ruiz-Hernando et al. 2014; Carrere et al. 2016; Martín-Díaz et al. 2020). A low effectiveness of pathogenic bacterial inactivation in sewage sludge produced in Swedish treatment plants after digestion procedures (both thermophilic/mesophilic processes) was observed by Sahlström et al. (2004). Therefore, it is justified to look for solutions that contribute to the elimination of bacteria and parasite eggs before the fermentation process. One such possibility may be the use of disintegration methods. As we have shown in our earlier works (Grübel and Suschka 2015; Machnicka et al. 2015; Suschka and Grübel 2016; Grübel and Machnicka 2020; Li et al. 2021), the disintegration process effectively releases organic matter into the supernatant liquid of WAS, which contributes to the increased production of biogas. In connection with the above, we have undertaken research to determine the effectiveness of disintegration in removing selected bacterial indicators and parasite eggs.
According to the methodology (Fig. 1), the anaerobic stabilization process was performed on WAS with the addition of sludge after the hybrid disintegration process.
Analyzing the above graph (Fig. 4), it can be noticed that with the increase in the volume of disintegrated WAS in the fermentation mixture, the reduction of the analyzed indicators increases. The reduction efficiency of Salmonella sp., Escherichia coli, Clostridium perfringens, and coliphages was better (from 1 to 23%) in mixtures with WASD compared to the control sample (70% WAS + 30% DS). Obviously, introducing only disintegrated sludge into the digestion chamber (70% WASD + 30% DS) is not economically and energetically justified from the technological point of view. Our previous research shows (Grübel and Suschka 2015) that the optimal dose of WASD is 30–40%, which definitely affects the production of biogas and biogas yield and is still economically justified.
Therefore, taking into account only the limited use of WASD in the fermentation mixture (30% vol.), the hygienization efficiency in relation to bacterial indicators was Salmonella sp. – 11%, Escherichia coli – 13%, Clostridium perfringens – 2%, and coliphages – 12%.
Similar effects were achieved with parasite eggs. Taking into account only 30% of the volume of WASD in the fermentation mixture, a reduction of 10% for Ascaris sp. and Toxocara sp. and 9.5% for Trichuris sp. in comparison to the blank sample (70% WAS + 30% DS) was attained.
Our investigations demonstrated an increase in the elimination of coliphages from 2.8 to 21.6% after the introduction of WASDH to fermentation processes. This result corresponds well to the results after mesophilic fermentation observed by Mandilara et al. (2006), which showed a reduction of 8.5%. Mocé-Llivina et al. (2003) claims that phages were much more resistant to thermal inactivation than bacterial indicators, with the exception of sulfite-reducing Clostridia spores. Somatic coliphages were significantly more resistant than Salmonella choleraesuis, E. coli, F-specific RNA phages, and enteroviruses. The coliphages survived markedly better than Salmonella choleraesuis, and the scope of inactivation indicated that naturally occurring bacteriophages can be used to monitor the deactivation of E. coli and Salmonella sp. Pino-Jelcic et al. (2006) observed the log inactivation of the fecal coliform of approximately 4.2 ± 0.4 for microwaved/digested sludge, whereas for conventionally heated/digested sludge and the control, the reductions were only 2.9 ± 0.5 and 1.5 ± 0.5, respectively. Less log deactivation was noted for Salmonella spp. in microwaved/digested sludge—approximately 2.0 ± 0.3, whereas for conventionally heated/digested sludge and the control, the reductions were 1.9 ± 0.2 and 1.1 ± 0.3, respectively. Similarly, Hong et al. (2006) concluded from bench-scale fermentation that the introduction of microwaved disintegrated sludge to an anaerobic digester was more efficient in the inactivation of fecal coliforms (average of ≥ 2.66 log reduction) in comparison to chambers fed with untreated sludge and externally heated sludge. They observed that Class A sludge can be produced only when the sludge before stabilization is heated using microwaves to 65 °C.
The research by Cella et al. (2016) showed an increase in fecal coliform removal from wastewater sludge depending on microwave intensification. In the fermenter chamber with the addition of microwaved sludge, 73.4% removal of coliforms was observed. Pike et al. (1988) described successful inactivation of Salmonella duesseldorf by mesophilic and thermophilic digestion but incomplete destruction of Ascaris suum ova. They concluded that the retention period influences the effectiveness of inactivation, and an increase in temperature from 36 to 48 °C increases the effectiveness by 3–4 times. Similar conclusions were obtained by Scheinemann et al. (2015). They found that 56 days of fermentation at 37 °C were required for a complete reduction of Ascaris suum ova. Moreover, Maya et al. (2010) noted that temperature plays an essential role in the survival of Ascaris eggs. Mesophilic processes are inefficient at totally eliminating viable nematode eggs (Venglovsky et al. 2006). Similarly, Wagner et al. (2008) and Orzi et al. (2015) found that the temperature and physico-chemical properties of the sludge in the fermenter influence the inhibition of microorganisms.
Very often, after pretreatment and anaerobic processes, there is a regrowth of bacterial indicators. According to Erkan and Sanin (2013) and Lepeuple et al. (2004), factors influencing the regrowth of bacteria are deficiency of nutrients, oxidation and osmotic stress and the presence of toxic substances. They are associated with the entry into the viable state of microorganisms, but they are not suitable for cultivation. This effect has not been found in bacteriophages. Martín-Díaz et al. (2017) concluded that alkali pretreatment and alkali and ultrasound pretreatments followed by anaerobic digestion repeatedly increased the number of Clostridia.
The results presented confirm the effectiveness of destroying microorganisms and therefore WAS flocs. For example, in relation to a standard technological cycle of the wastewater purification process for complex biogenic and organic carbon elimination with the application of activated sludge, this process can be feasibility implemented in several places depending on the expected effects (Fig. 5).
Destruction by disintegration can be implemented in the process of internal (D) or external (E) recirculation together with the part sludge from the secondary settling tank to anaerobic zone (A), anoxic (B), and aerobic (C).
Introducing disintegrated WAS into the anaerobic chamber (A) may help increase phosphorus bacteria activity by increasing the charge of volatile fatty acids (VFA) in the chamber. In the anoxic zone (B) disintegrated WAS containing a high concentration of easily digestible carbon compound will assure to the denitrification bacteria the intensity of the nitrate reduction process in the situation of shortage of organic matter in wastewater flowing into the activated sludge reactor. The aerobic zone (C) through oxygenated metabolisms of heterotrophic microorganisms mainly decreases the concentration of non-settling, soluble and colloid carbon compounds in wastewater. The introduction of additional organic substrate will contribute to the rapid reproduction of bacteria, the increase of zoogleal clusters and the production of WAS flocks. On the other hand, applying WAS disintegration to the recirculation flow (D and E) can be used by additional/external carbon sources for metabolic processes.
Disintegrated WAS introduced with condensed surplus activated sludge (F) to digestion chamber could improve fermentation efficiency. According to the methodology, the impact of the hybrid method of disintegration on the mesophilic fermentation process (biogas production and methane concentration) was determined (Table 1).
The obtained results confirmed that hybrid disintegration pretreatment (hydrodynamic cavitation and dry ice freezing/thawing) of only a part of WAS was substantiated. The volume of WASDH investigated was 10–70% in the digestion mixture. The highest effects of cumulative biogas production were obtained for 30% and 40% WASDH with increases of approximately 61.1% and 62.2%, respectively (Table 1).
The increasing amount of sewage sludge and the legislative regulation of its disposal have caused the need for developing new technologies to effectively process sewage sludge hygienization.
Conclusions
Hybrid pretreatment processes (hydrodynamic disintegration and freezing/thawing) are a method to destroy bacteria and helminth eggs in waste activated sludge. Based on the microbiological and parasitological analyses, a significant reduction in pathogenic bacteria, coliphages and parasite eggs was observed. The number of Salmonella sp. in 1 gd.w. decreased by approximately 69.7%, that of E. coli decreased by 70.0%, that of Clostridium perfringens decreased by 38.4%, and that of coliphages decreased by 48.2% after hybrid disintegration. The disruption of WAS by hybrid pretreatment resulted in a reduction in the number of helminth eggs of Ascaris sp. (63.8%), Trichuris sp. (64.3%), and Toxocara sp. (66.4%).
The application of pretreatment methods and anaerobic digestion for WAS hygienization resulted in a higher reduction efficiency of Salmonella sp., Escherichia coli, Clostridium perfringens, and coliphages, from 1 to 23% in mixtures with WASD compared to the control sample (70% WAS + 30% DS). This indicates a lower transmission of sanitary indicators in the agricultural utilization of sludge after both pretreatments and digestion processes.
Data availability
Not applicable.
References
Carrere H, Antonopoulou G, Affes R et al (2016) Review of feedstock pretreatment strategies for improved anaerobic digestion: from lab-scale research to full-scale application. Biores Technol 199:386–397. https://doi.org/10.1016/J.BIORTECH.2015.09.007
Cella MA, Akgul D, Eskicioglu C (2016) Assessment of microbial viability in municipal sludge following ultrasound and microwave pretreatments and resulting impacts on the efficiency of anaerobic sludge digestion. Appl Microbiol Biotechnol 100:2855–2868. https://doi.org/10.1007/S00253-015-7139-3
Council directive 86/278/EEC on protection of the environment, and in particular of the soil, when sewage sludge is used in agriculture (https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A31986L0278).
Doğan I, Sanin FD (2009) Alkaline solubilization and microwave irradiation as a combined sludge disintegration and minimization method. Water Res 43:2139–2148
Eng SK, Pusparajah P, Ab Mutalib NS et al (2015) Salmonella: a review on pathogenesis, epidemiology and antibiotic resistance. Front Life Sci 8:284–293. https://doi.org/10.1080/21553769.2015.1051243
Erkan M, Sanin FD (2013) Can sludge dewatering reactivate microorganisms in mesophilically digested anaerobic sludge? Case of belt filter versus centrifuge. Water Res 47:428–438. https://doi.org/10.1016/J.WATRES.2012.10.028
European Commission (2000) Working document on sludge, 3rd Official Draft. European Commission, Brussels, Belgium (http://www.ewa-online.eu/comments.html).
Ferreira LC, Castro-Alférez M, Nahim-Granados S et al (2020) Inactivation of water pathogens with solar photo-activated persulfate oxidation. Chem Eng J 381:122275. https://doi.org/10.1016/J.CEJ.2019.122275
Fukushi K, Babel S, Burakrai S (2003) Survival of Salmonella spp. in a simulated acid-phase anaerobic digester treating sewage sludge. Biores Technol 86:177–181. https://doi.org/10.1016/S0960-8524(02)00146-3
Gopi Kumar S, Merrylin J, Kaliappan S et al (2012) Effect of cation binding agents on sludge solubilization potential of bacteria. Biotechnol Bioprocess Eng 17:346–352. https://doi.org/10.1007/s12257-011-0465-0
Grübel K, Suschka J (2015) Hybrid alkali-hydrodynamic disintegration of waste-activated sludge before two-stage anaerobic digestion process. Environ Sci Pollut Res 22:7258–7270. https://doi.org/10.1007/s11356-014-3705-y
Grübel K, Wacławek S, Machnicka A, Nowicka E (2018) Synergetic disintegration of waste activated sludge: improvement of the anaerobic digestion and hygienization of sludge. Journal of Environmental Science and Health, Part A 53:1067–1074. https://doi.org/10.1080/10934529.2018.1474579
Grübel K, Machnicka A (2020) The use of hybrid disintegration of activated sludge to improve anaerobic stabilization process. Inzynieria Ekologiczna 21:. https://doi.org/10.12912/23920629/119104
Hawrylik E (2019) The usage of ultrasounds to disintegrate Escherichia coli bacteria contained in treated wastewater. Architecture Civil Engineering Environment Vol. 12:131–136. https://doi.org/10.21307/ACEE-2019-043
Hoang SA, Bolan N, Madhubashini AMP et al (2022) Treatment processes to eliminate potential environmental hazards and restore agronomic value of sewage sludge: a review. Environ Pollut 293:118564. https://doi.org/10.1016/J.ENVPOL.2021.118564
Holt JG, Krieg NR, Sneath PHA, Staley JT, Williams ST (1994) Bergey’s manual of determinative bacteriology, 9th edn. The Williams & Wilkins Co., Baltimore
Hong SM, Park JK, Teeradej N et al (2006) Pretreatment of sludge with microwaves for pathogen destruction and improved anaerobic digestion performance. Water Environ Res 78:76–83. https://doi.org/10.2175/106143005X84549
Hu K, Jiang J-Q, Zhao Q-L et al (2011) Conditioning of wastewater sludge using freezing and thawing: Role of curing. Water Res 45:5969–5976. https://doi.org/10.1016/j.watres.2011.08.064
ISO 10705–2:2000 - Water quality — Detection and enumeration of bacteriophages — Part 2: Enumeration of somatic coliphages. https://www.iso.org/standard/20127.html. Accessed 25 Mar 2022
Jiang Y, Xie SH, Dennehy C et al (2020) Inactivation of pathogens in anaerobic digestion systems for converting biowastes to bioenergy: a review. Renew Sustain Energy Rev 120.https://doi.org/10.1016/J.RSER.2019.109654
Khanh Nguyen V, Kumar Chaudhary D, Hari Dahal R et al (2021) Review on pretreatment techniques to improve anaerobic digestion of sewage sludge. Fuel 285:119105. https://doi.org/10.1016/J.FUEL.2020.119105
Lakshmi MV, Merrylin J, Kavitha S et al (2014) Solubilization of municipal sewage waste activated sludge by novel lytic bacterial strains. Environ Sci Pollut Res 21:2733–2743. https://doi.org/10.1007/s11356-013-2228-2
Lepeuple AS, Gaval G, Jovic M, Roubin MR De (2004) Horizontal Work Package 3, Task 6: Literature review on levels of pathogens and their abatement in sludges, soil and treated biowaste (https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.531.6070&rep=rep1&type=pdf).
Li M, Song G, Liu R et al (2021) Inactivation and risk control of pathogenic microorganisms in municipal sludge treatment: a review. Front Environ Sci Eng 16(6):1–23. https://doi.org/10.1007/S11783-021-1504-5
Liao X, Li H, Zhang Y et al (2016) Accelerated high-solids anaerobic digestion of sewage sludge using low-temperature thermal pretreatment. Int Biodeterior Biodegradation 106:141–149. https://doi.org/10.1016/j.ibiod.2015.10.023
Liu X, Lendormi T, Lanoisellé JL (2019) Overview of hygienization pretreatment for pasteurization and methane potential enhancement of biowaste: challenges, state of the art and alternative technologies. J Clean Prod 236:117525. https://doi.org/10.1016/J.JCLEPRO.2019.06.356
Lloret E, Pastor L, Martínez-Medina A et al (2012) Evaluation of the removal of pathogens included in the Proposal for a European Directive on spreading of sludge on land during autothermal thermophilic aerobic digestion (ATAD). Chem Eng J 198–199:171–179. https://doi.org/10.1016/J.CEJ.2012.05.068
Lopes TR, Periotto F, Pletsch AL (2017) Bacterial resistance in sanitary sewage sludge in different treatment systems. Management of Environmental Quality: an International Journal 28:32–42. https://doi.org/10.1108/MEQ-07-2015-0146/FULL/HTML
López A, Baguer B, Goñi P et al (2019) Assessment of the methodologies used in microbiological control of sewage sludge. Waste Manage 96:168–174. https://doi.org/10.1016/J.WASMAN.2019.07.024
Luukkonen T, Prokkola H, Pehkonen SO (2020) Peracetic acid for conditioning of municipal wastewater sludge: Hygienization, odor control, and fertilizing properties. Waste Manage 102:371–379. https://doi.org/10.1016/J.WASMAN.2019.11.004
Machnicka A, Grübel K, Mirota K (2015) Considerations of impact of Venturi effect on mesophilic digestion. Ecol Chem Eng S 22:645–658. https://doi.org/10.1515/eces-2015-0039
Machnicka A, Nowicka E, Grübel K (2017) Disintegration as a key-step in pre-treatment of surplus activated sludge. J Water Chem Technol 39:47–55. https://doi.org/10.3103/S1063455X17010088
Mandilara GD, Smeti EM, Mavridou AT et al (2006) Correlation between bacterial indicators and bacteriophages in sewage and sludge. FEMS Microbiol Lett 263:119–126. https://doi.org/10.1111/J.1574-6968.2006.00414.X
Martín-Díaz J, Casas-Mangas R, García-Aljaro C et al (2016) Somatic coliphages as surrogates for enteroviruses in sludge hygienization treatments. Water Sci Technol 73:2182–2188. https://doi.org/10.2166/WST.2016.066
Martín-Díaz J, Ruiz-Hernando M, Astals S, Lucena F (2017) Assessing the usefulness of clostridia spores for evaluating sewage sludge hygienization. Biores Technol 225:286–292. https://doi.org/10.1016/J.BIORTECH.2016.11.075
Martín-Díaz J, Lucena F, Blanch AR, Jofre J (2020) Review: indicator bacteriophages in sludge, biosolids, sediments and soils. Environ Res 182.https://doi.org/10.1016/J.ENVRES.2020.109133
Mawioo PM, Garcia HA, Hooijmans CM et al (2017) A pilot-scale microwave technology for sludge sanitization and drying. Sci Total Environ 601–602:1437–1448. https://doi.org/10.1016/J.SCITOTENV.2017.06.004
Maya C, Ortiz M, Jiménez B (2010) Viability of Ascaris and other helminth genera non larval eggs in different conditions of temperature, lime (pH) and humidity. Water Sci Technol 62:2616–2624. https://doi.org/10.2166/WST.2010.535
Minister of Environment (2015) Journal of Laws, item 257 (in Polish)
Mocé-Llivina L, Muniesa M, Pimenta-Vale H et al (2003) Survival of bacterial indicator species and bacteriophages after thermal treatment of sludge and sewage. Appl Environ Microbiol 69:1452–1456. https://doi.org/10.1128/AEM.69.3.1452-1456.2003
Nowicka E, Machnicka A, Grübel K (2014) Improving of anaerobic digestion by dry ice disintegration of activated sludge. Ecol Chem Eng A 21.https://doi.org/10.2428/ECEA.2014.21(2)17
Nowicka E, Machnicka A (2015) Hygienization of surplus activated sludge by dry ice. Ecol Chem Eng S. https://doi.org/10.1515/eces-2014-0047
Orzi V, Scaglia B, Lonati S et al (2015) The role of biological processes in reducing both odor impact and pathogen content during mesophilic anaerobic digestion. Sci Total Environ 526:116–126. https://doi.org/10.1016/J.SCITOTENV.2015.04.038
Pike EB, Carrington EG, Harman SA (1988) Destruction of salmonellas, enteroviruses and ova of parasites in wastewater sludge by pasteurisation and anaerobic digestion. Water Sci Technol 20:337–343. https://doi.org/10.2166/WST.1988.0304
Pino-Jelcic SA, Hong SM, Park JK (2006) enhanced anaerobic biodegradability and inactivation of fecal coliforms and Salmonella spp. in Wastewater Sludge by Using Microwaves. Water Environ Res 78:209–216
Polish Standard No. PN-Z19000–4:2001
Ruiz-Hernando M, Martín-Díaz J, Labanda J et al (2014) Effect of ultrasound, low-temperature thermal and alkali pre-treatments on waste activated sludge rheology, hygienization and methane potential. Water Res 61:119–129. https://doi.org/10.1016/J.WATRES.2014.05.012
Sahlström L, Aspan A, Bagge E et al (2004) Bacterial pathogen incidences in sludge from Swedish sewage treatment plants. Water Res 38:1989–1994. https://doi.org/10.1016/J.WATRES.2004.01.031
Scheinemann HA, Dittmar K, Stöckel FS et al (2015) Hygienisation and nutrient conservation of sewage sludge or cattle manure by lactic acid fermentation. PLoS ONE 10:e0118230. https://doi.org/10.1371/JOURNAL.PONE.0118230
Sidhu JPS, Toze SG (2009) Human pathogens and their indicators in biosolids: a literature review. Environ Int 35:187–201. https://doi.org/10.1016/J.ENVINT.2008.07.006
Suschka J, Grübel K (2016) Low intensity surplus activated sludge pretreatment before anaerobic digestion. Archives of Environmental Protection 43:50–57. https://doi.org/10.1515/aep-2017-0038
United States Environmental Protection Agency (2003) Environmental regulations and technology control of pathogens and vector attraction in sewage sludge control of pathogens and vector attraction (https://www.epa.gov/biosolids/control-pathogens-and-vector-attraction-sewage-sludge)
Venglovsky J, Martinez J, Placha I (2006) Hygienic and ecological risks connected with utilization of animal manures and biosolids in agriculture. Livest Sci 102:197–203. https://doi.org/10.1016/J.LIVSCI.2006.03.017
Wagner AO, Gstraunthaler G, Illmer P (2008) Survival of bacterial pathogens during the thermophilic anaerobic digestion of biowaste: laboratory experiments and in situ validation. Anaerobe 14:181–183. https://doi.org/10.1016/J.ANAEROBE.2008.03.004
Wójcik M, Stachowicz F, Masłoń A (2017) The possibility of sewage sludge conditioning and dewatering with the use of biomass ashes. Eng Prot Environ 20:153–164. https://doi.org/10.17512/ios.2017.2.1
Zdybel J, Karamon J, Dąbrowska J et al (2019) Parasitological contamination with eggs Ascaris spp., Trichuris spp. and Toxocara spp. of dehydrated municipal sewage sludge in Poland. Environ Pollut 248:621–626. https://doi.org/10.1016/J.ENVPOL.2019.02.003
Zhen G, Lu X, Kato H et al (2017) Overview of pretreatment strategies for enhancing sewage sludge disintegration and subsequent anaerobic digestion: current advances, full-scale application and future perspectives. Renew Sustain Energy Rev 69:559–577. https://doi.org/10.1016/j.rser.2016.11.187
Zhou X, Wang Q, Jiang G et al (2015) A novel conditioning process for enhancing dewaterability of waste activated sludge by combination of zero-valent iron and persulfate. Biores Technol 185:416–420. https://doi.org/10.1016/j.biortech.2015.02.088
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Alicja Machnicka and Klaudiusz Grübel. The first draft of the manuscript was written by Alicja Machnicka and Klaudiusz Grübel and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The manuscript is original and not have been published elsewhere in any form or language.
Consent to participate
Not applicable.
Consent to publish
All authors gave explicit consent to submit the manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Responsible editor: Gerald Thouand
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Machnicka, A., Grübel, K. The effect of pre-treatment and anaerobic digestion for pathogens reduction in agricultural utilization of sewage sludge. Environ Sci Pollut Res 30, 13801–13810 (2023). https://doi.org/10.1007/s11356-022-23164-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23164-9