Abstract
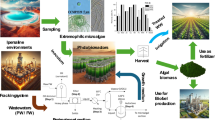
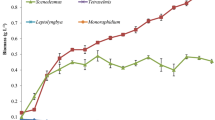
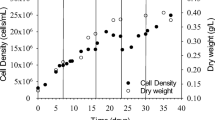
Oilfield-produced wastewater (OPW) provided by the local oil industry was used as a medium (without any pre-treatment) for the outdoor cultivation of microalgae Chlorella pyrenoidosa. The effectiveness of algal growth on the produced water treatment has been investigated. The experimental setups were carried out outdoor, under sunlight radiation, using an open system sited in the desert area. The highest biomass concentration was attaining 1.15 ± 0.07 g/L after 21 culture days. FTIR spectroscopy was used to estimate the lipid content in C. pyrenoidosa grown in BG11 and OPW medium. Daytime temperatures fluctuation was between 26 and 31 °C. The average insolation was no less than 10 h per day with maximum solar irradiation of 1036 ± 30 W/m2, measured between 12 and 1 p.m. C. pyrenoidosa was found highly capable of removing COD, NH4 + –N, TN, and TP by 89.67%, 100%, 57.14%, and 75.51%, respectively, throughout the cultivation period. Biosorption of toxic heavy metal pollutants such as Cu, Pb, and Cd was also achieved at rates of approximately 73.39, 72.80, and 48.42%. Overall, the achieved result of C. pyrenoidosa–based process was compared to the actual process using activated carbon.






Similar content being viewed by others
Data availability
The data used and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Bharte S, Desai K (2019) The enhanced lipid productivity of C. minutissima and C. pyrenoidosa by carbon coupling nitrogen manipulation for biodiesel production. Environ Sci Pollut Res 26(4):3492–500. https://doi.org/10.1007/s11356-018-3757
Bhandari M, Prajapati SK (2022) Use of reverse osmosis reject from drinking water plant for microalgal biomass production. Water Res 210:117989. https://doi.org/10.1016/j.watres.2021.117989
Chen X, Yang Y, Lu Q, Sun X, Wang S, Li Q, Wei X (2021) Wang Y (2021) The influence of light intensity and organic content on cultivation of Chlorella vulgaris in sludge extracts diluted with BG11. Aqua Cult Int 29:2131–2144. https://doi.org/10.1007/s10499-021-00740-3
Ciurli A, Di Baccio D, Scartazza A, Grifoni M, Pezzarossa B, Chiellini C, Mariotti L, Pardossi A (2021) Influence of zinc and manganese enrichments on growth, biosorption and photosynthetic efficiency of Chlorella sp. Environ Sci Pollut Res 7:8539–8555. https://doi.org/10.1007/s11356-020-11033-2
Dahmani S, Zerrouki D, Ramanna L, Rawat I, Bux F (2016) Cultivation of Chlorella pyrenoidosa in outdoor open raceway pond using domestic wastewater as medium in arid desert. Bioresour Technol 219:749–752. https://doi.org/10.1016/j.biortech.2016.08.019
Dean AP, Sigee DC, Estrada B, Pittman JK (2010) Using FTIR spectroscopy for rapid determination of lipid accumulation in response to nitrogen limitation in freshwater microalgae. Bioresour Technol 101:4499–4507. https://doi.org/10.1016/j.biortech.2010.01.065
Guccione A, Biondi N, Sampietro G, Rodolfi L, Bassi N, Tredici MR (2014) Chlorella for protein and biofuels: from strain selection to outdoor cultivation in a Green Wall Panel photobioreactor. Biotechnol Biofuels 7(1):1–12. https://doi.org/10.1186/1754-6834-7-84
Gillard JTF, Hernandez AL, Contreras JA, Francis IM, Cabrales L (2021) Potential for biomass production and remediation by cultivation of the marine model diatom Phaeodactylum tricornutum in oil field produced wastewater media. Water 13:2700. https://doi.org/10.3390/w13192700
Hollanda LR, Santos SB, Faustino JG, Dotto GL, Foletto EL, Chiavone-Filho O (2021) Oil field–produced water treatment: characterization, photochemical systems, and combined processes. Environ Sci Pollut Res 28(38):52744–52763. https://doi.org/10.1007/s11356-021-16222-1
Han F, Huang J, Li Y, Wang W, Wan M, Shen G, Wang J (2014a) Enhanced lipid productivity of Chlorella pyrenoidosa through the culture strategy of semi-continuous cultivation. Bioresour Technol 136:418–424. https://doi.org/10.1016/j.biortech.2013.03.017
Han F, Wang W, Li Y, Shen G, Wan M, Wang J (2014b) Changes of biomass, lipid content and fatty acids composition under a light-dark cyclic culture of Chlorella pyrenoidosa. Bioresour Technol 132:182–189. https://doi.org/10.1016/j.biortech.2012.12.175
Hongyang S, Yalei Z, Chunmin Z, Xuefei Z, Jinpeng L (2011) Cultivation of Chlorella pyrenoidosa in soybean processing wastewater. Bioresour Technol 102:9884–9890. https://doi.org/10.1016/j.biortech.2011.08.016
Igunnu ET, Chen GZ (2012) Produced water treatment technologies. Int J Low-Carbon Technol 9:157–177. https://doi.org/10.1093/ijlct/cts049
Ma X, Zhou W, Fu Z, Cheng Y, Min M, Liu Y, Zhang Y, Chen P, Ruan R (2014) Effect of wastewater-borne bacteria on algal growth and nutrients removal in wastewater-based algae cultivation system. Bioresour Technol 167:8–13. https://doi.org/10.1016/j.biortech.2014.05.087
Madadi R, Pourbabaee AA, Tabatabaei M, Zahed MA, Naghavi MR (2016) Treatment of Petrochemical wastewater by the green algae Chlorella vulgaris. Int J Environ Res 10:555–560. https://doi.org/10.22059/IJER.2016.59684
Markou G, Iconomou D, Muylaert K (2016) Applying raw poultry litter leachate for the cultivation of Arthrospira platensis and Chlorella vulgaris. Algal Res 13:79–84. https://doi.org/10.1016/j.algal.2015.11.018
Mayers J, Flynn KJ, Shields RJ, Mayers JJ, Flynn KJ, Shields RJ (2013) Rapid determination of bulk microalgal biochemical composition by Fourier-transform infrared spectroscopy by Fourier-Transform Infrared spectroscopy. Bioresour Technol 148:215–220. https://doi.org/10.1016/j.biortech.2013.08.133
Meng Y, Yao C, Xue S, Yang H (2014) Application of Fourier transform infrared (FT-IR) spectroscopy in determination of microalgal compositions. Bioresour Technol 151:347–354. https://doi.org/10.1016/j.biortech.2013.10.064
Liu J, Mukherjee J, Hawkes JJ, Wilkinson SJ (2013) Optimization of lipid production for algal biodiesel in nitrogen stressed cells of Dunaliella salina using FTIR analysis. Chem Technol Biotechnol 88(10):1807–1814. https://doi.org/10.1002/jctb.4027
Murdock JN, Wetzel DL (2009) FT-IR microspectroscopy enhances biological and ecological analysis of algae. Appl Spectrosc Rev 44:335–361. https://doi.org/10.1080/05704920902907440
Nelson SG, Kalfayan LJ, Rittenberry WM (2003) The application of a new and unique relative permeability modifier in selectively reducing water production. SPE Annu Tech Conf Exhib SPE-84511-MS. https://doi.org/10.2118/84511-MS
Prajapati SK, Malik A, Vijay VK (2013) Comparative evaluation of biomass production and bioenergy generation potential of Chlorella spp. through anaerobic digestion. Appl Energy 114:790–797
Rana MS, Prajapati SK (2021) Resolving the dilemma of iron bioavailability to microalgae for commercial sustenance. Algal Res 59:102458. https://doi.org/10.1016/j.algal.2021.102458
Rana MS, Bhushan S, Prajapati SK (2020) New insights on improved growth and biogas production potential of Chlorella pyrenoidosa through intermittent iron oxide nanoparticle supplementation. Sci Rep 10(1):1–3. https://doi.org/10.1038/s41598-020-71141-4:790-7.10.1016/j.apenergy.2013.08.021
Qin L, Wang B, Feng P, Cao Y, Wang Z, Zhu S (2022) Treatment and resource utilization of dairy liquid digestate by nitrification of biological aerated filter coupled with assimilation of Chlorella pyrenoidosa. Environ Sci Pollut Res 3:3406–3416. https://doi.org/10.1007/s11356-021-15903-1
Shi W, Jin Z, Hu S, Fang X, Li F (2017) Dissolved organic matter affects the bioaccumulation of copper and lead in Chlorella pyrenoidosa: a case of long-term exposure. Chemosphere 174:447–455. https://doi.org/10.1016/j.chemosphere.2017.01.119
Sullivan EJ, Dean CA, Yoshida TM, Twary SN, Teshima M, Alvarez MA, Zidenga T, Heikoop JM, Perkins GB, Rahn TA, Wagner GL, Laur PM (2017) Oil and gas produced water as a growth medium for microalgae cultivation: a review and feasibility analysis. Algal Res. 1–13. https://doi.org/10.1016/j.algal.2017.01.009
Stephen DP, Ayalur BK (2017) Effect of nutrients on Chlorella pyrenoidosa for treatment of phenolic effluent of coal gasification plant. Environ Sci Pollut Res 15:13594–13603. https://doi.org/10.1007/s11356-017-8891-y)
Torres E, Mera R, Herrero C, Abalde J (2014) Isotherm studies for the determination of Cd (II) ions removal capacity in living biomass of a microalga with high tolerance to cadmium toxicity. Environ Sci Pollut Res 22:12616–12628. https://doi.org/10.1007/s11356-014-3207-y
Talebi A, Dastgheib M, Tirandaz H, Alaie E, Tabatabaei M (2016) Enhanced algal-based treatment of petroleum produced water and biodiesel production. Rsc Adv 6:47001–47009. https://doi.org/10.1039/C6RA06579A
Tan XB, Zhang YL, Yang LB, Chu HQ, Guo J (2016) Outdoor cultures of C. pyrenoidosa in the effluent of anaerobically digested activated sludge: the effects of pH and free ammonia. Bioresour Technol 200:606–615. https://doi.org/10.1016/j.biortech.2015.10.095
Veil J, Clark C (2011) Produced water volume estimates and management practices. Soc Pet Eng Prod Oper 26:234–239. https://doi.org/10.2118/125999-PA
Winckelmann D, Bleeke F, Thomas B, Elle C, Klöck G (2015) Open pond cultures of indigenous algae grown on non-arable land in an arid desert using wastewater. IntAquat Res 221–233. https://doi.org/10.1007/s40071-015-0107-9
Zheng T, Ke C (2017) Treatment of oilfield wastewater using algal–bacterial fluidized bed reactor. Sep Sci Technol 52(13):2090–2097. https://doi.org/10.1080/01496395.2017.1307853
Zhou GJ, Peng FQ, Zhang LJ, Ying GG (2012) Biosorption of zinc and copper from aqueous solutions by two freshwater green microalgae Chlorella pyrenoidosa and Scenedesmus obliquus. Environ Sci Pollut Res 7:2918–2929. https://doi.org/10.1007/s11356-012-0800-9)
Acknowledgements
The author gratefully acknowledges the funding support from the University of Ouargla. The authors would like to thank André Ayral (Professor, University Montpellier) for helpful comments and suggestions that greatly improved the manuscript.
Author information
Authors and Affiliations
Contributions
Djamal Zerrouki, Ahmed Tabchouche, and Lahcène Djafer proposed the original idea, wrote, reviewed, edited the manuscript, and administrated the project, and wrote the original manuscript. Abdellatif Rahmani conducted the experiments and performed laboratory measurements.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Responsible Editor: Ta Yeong Wu
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rahmani, A., Zerrouki, D., Tabchouche, A. et al. Oilfield-produced water as a medium for the growth of Chlorella pyrenoidosa outdoor in an arid region. Environ Sci Pollut Res 29, 87509–87518 (2022). https://doi.org/10.1007/s11356-022-21916-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21916-1