Abstract
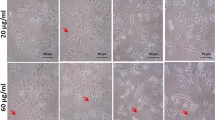
Pluronics, due to its high water-soluble and thermoreversible ability, attracted much in biomedical applications. They are mainly utilized in drug delivery, gene therapy, and tissue remodeling. The study aims to explore the cytocompatibility of Pluronics F-127, which has gained much popularity due to its various properties. The cells were exposed to varying concentrations of Pluronics F-127 in A549 cells for 24 h. According to the MTT and neutral red assay, A549 cells displayed dose-dependent cell viability. The cell’s morphology was preserved after treatment, as seen in phase-contrast and Giemsa staining. When exposed to PF-127, lysosomal, cytoskeletal, and nuclear integrity were maintained. The percentage of live cells in all the treated groups was more significant than 90%, according to the live/dead flow cytometric analyses. The study identified the cytocompatibility of Pluronics F-127 required for the breakthrough in biomedical applications.
Graphical abstract










Similar content being viewed by others
Data availability
The authors declared that the research data was correctly cited in the manuscript’s reference section.
References
Aits S, Jäättelä M (2013) Lysosomal cell death at a glance. J Cell Sci 126:1905–1912. https://doi.org/10.1242/jcs.091181
Akash MSH, Rehman K, Li N, Gao JQ, Sun H, Chen S (2012) Sustained delivery of IL-1Ra from Pluronic F127-based thermosensitive gel prolongs its therapeutic potentials. Pharm Res 29:3475–3485. https://doi.org/10.1007/s11095-012-0843-0
Akash MSH, Rehman K, Sun H, Chen S (2013) Sustained delivery of IL-1Ra from PF127-gel reduces hyperglycemia in diabetic GK-rats. PLoS ONE. https://doi.org/10.1371/journal.pone.0055925
Attama AA, Uzor PF, Nnadi CO, Okafor CG (2011) Evaluation of the wound healing activity of gel formulations of leaf extract of Aspilaafricana Fam. Compositae J Chem Pharm Res 3:718–724
Barry AP, Barry NPE (2014) Barry Pluronic® block-copolymers in medicine: from chemical and biological versatility to rationalization and clinical advances. Polym Chem 5:3291. https://doi.org/10.1039/c4py00039k
Batrakova EV, Kabanov AV (2008) Pluronic block copolymers: evolution of drug delivery concept from inert nanocarriers to biological response modifiers. J Controlled Release 130:98–106. https://doi.org/10.1016/j.jconrel.2008.04.013
Bensaıd W, Triffitt JT, Blanchat C, Oudina K, Sedel L, Petite H (2003) A biodegradable fibrin scaffold for mesenchymal stem cell transplantation. Biomaterials 24:2497–2502. https://doi.org/10.1016/S0142-9612(02)00618-X
Borenfreund E, Puerner JA (1985) Toxicity determined in vitro by morphological alterations and neutral red absorption. Toxicol Lett 24:119–124. https://doi.org/10.1016/0378-4274(85)90046-3
Brunet-Maheu JM, Fernandes JC, De Lacerda CA, Shi Q, Benderdour M, Lavigne P (2009) Pluronic F-127 as a cell carrier for bone tissue engineering. J BiomaterAppl 24:275–287. https://doi.org/10.1177/0885328208096534
Chen L, Sha X, Jiang X, Chen Y, Ren Q, Fang X (2013) Pluronic P105/F127 mixed micelles for the delivery of docetaxel against Taxol-resistant non-small cell lung cancer: optimization and in vitro, in vivo evaluation. Int J Nanomed 8:73–84. https://doi.org/10.2147/IJN.S38221
Cold Spring HarbProtoc; 2011; https://doi.org/10.1101/pdb.prot5556
Danson S, Ferry D, Alakhov V, Margison J, Kerr D, Jowle D, Brampton M, Halbert G, Ranson M (2004) Phase I dose escalation and pharmacokinetic study of pluronic polymer-bound doxorubicin (SP1049C) in patients with advanced cancer. Br J Cancer 90:2085–2091. https://doi.org/10.1038/sj.bjc.6601856
DiBiase MD, Rhodes CT (1996) Formulation and evaluation of epidermal growth factor in pluronic F-127 gel. Drug Dev Ind Pharm 22:823–831. https://doi.org/10.3109/03639049609065912
Escobar-Chávez JJ, López-Cervantes M, Naik A, Kalia Y, Quintanar-Guerrero D, Ganem-Quintanar A (2006) Applications of thermo-reversible pluronic F-127 gels in pharmaceutical formulations. J Pharm Pharm Sci 9:339–358
Fletcher DA, Mullins RD (2010) Cell mechanics and the cytoskeleton. Nature 463:485–492. https://doi.org/10.1038/nature08908
Fowler EB, Cuenin MF, Hokett SD, Peacock ME, McPherson JC III, Dirksen TR, Sharawy M, Billman MA (2002) Evaluation of pluronic polyols as carriers for grafting materials: study in rat calvaria defects. J Periodontol 73:191–197. https://doi.org/10.1902/jop.2002.73.2.191
Hamaguchi T, Kato K, Yasui H, Morizane C, Ikeda M, Ueno H, Muro K, Yamada Y, Okusaka T, Shirao K, Shimada Y (2007) A phase I and pharmacokinetic study of NK105, a paclitaxel-incorporating micellar nanoparticle formulation. Br J Cancer 97:170–176. https://doi.org/10.1038/sj.bjc.6603855
Huang J, Chen W, Liao S, Yang C, Lin S, Wu C (2006) Osteoblastic differentiation of rabbit mesenchymal stem cells loaded in a carrier system of Pluronic F127 and Interpore. Chang Gung Med J 29:363
Huang SJ, Wang TP, Lue SI, Wang LF (2013) Pentablock copolymers of Pluronic F127 and modified poly(2-dimethyl amino)ethyl methacrylate for internalization mechanism and gene transfection studies. Int J Nanomedicine 8:2011–2027. https://doi.org/10.2147/IJN.S44222
Kabanov AV, Batrakova EV, Alakhov VY (2002) Pluronic block copolymers as novel polymer therapeutics for drug and gene delivery. J Controlled Release 82:189–212. https://doi.org/10.1016/s0168-3659(02)00009-3
Kabanov AV, Batrakova EV, Miller DW (2003) Pluronic® block copolymers as modulators of drug efflux transporter activity in the blood–brain barrier. Adv Drug Deliv Rev 55:151–164. https://doi.org/10.1016/S0169-409X(02)00176-X
Kaisang L, Siyu W, Lijun F, Daoyan P, Xian CJ, Jie S (2017) Adipose-derived stem cells seeded in Pluronic F-127 hydrogel promotes diabetic wound healing. J Surg Res 217:63–74. https://doi.org/10.1016/j.jss.2017.04.032
Kim EY, Gao ZG, Park JS, Li H, Han K (2002) rhEGF/HP-β-CD complex in poloxamer gel for ophthalmic delivery. Int J Pharm 233:159–167. https://doi.org/10.1016/s0378-5173(01)00933-4
Lee SY, Tae G (2007) Formulation and in vitro characterization of an in situ gelable, photo-polymerizable pluronic hydrogel suitable for injection. J Control Release 119:313–319. https://doi.org/10.1016/j.jconrel.2007.03.007
Li Z, Xiong X, Peng S, Chen X, Liu W, Liu C (2020) Novel folated Pluronic F127 modified liposomes for delivery of curcumin: preparation, release, and cytotoxicity. J Microencapsul 37:220–229. https://doi.org/10.1080/02652048.2020.1720030
Liu Y, Lu WL, Wang JC, Zhang X, Zhang H, Wang XQ, Zhou TY, Zhang Q (2007) Controlled delivery of recombinant hirudin based on thermo-sensitive Pluronic® F127 hydrogel for subcutaneous administration: in vitro and in vivo characterization. J Controlled Release 117:387–395. https://doi.org/10.1016/j.jconrel.2006.11.024
Matsumura Y (2007) Preclinical and clinical studies of anticancer drug-incorporated polymeric micelles. J Drug Target 15:507–517. https://doi.org/10.1080/10611860701499888
Moebus K, Siepmann J, Bodmeier R (2009) Alginate–poloxamer microparticles for controlled drug delivery to mucosal tissue. Eur J Pharm Biopharm 72:42–53. https://doi.org/10.1016/j.ejpb.2008.12.004
Mosman T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Nie S, Hsiao WW, Pan W, Yang Z (2011) Thermoreversible Pluronic® F127-based hydrogel containing liposomes for the controlled delivery of paclitaxel: in vitro drug release, cell cytotoxicity, and uptake studies. Int J of Nanomedicine 6:151. https://doi.org/10.2147/IJN.S15057
Ottenbrite RM, Javan R (2005) Biological structures. Elsevier Academic Press, USA, Encyclopedia of condensed matter physics, pp 99–108
Rokhade AP, Shelke NB, Patil SA, Aminabhavi TM (2007) Novel hydrogel microspheres of chitosan and Pluronic F-127 for controlled release of 5-fluorouracil. J Microencapsul 24:274–288. https://doi.org/10.1080/02652040701281365
Samal SK, Dash M, Vlierberghe V, Kaplan DL, Chiellini E, Bitterswijk V, Moroni L, Dubruel P (2012) Cationic polymers and their therapeutic potential. Chem Soc Rev 41:7147–7194. https://doi.org/10.1039/C2CS35094G
Shelke NB, Aminabhavi TM (2007) Synthesis and characterization of novel poly (sebacic anhydride-co-Pluronic F68/F127) biopolymeric microspheres for the controlled release of nifedipine. Int J Pharm 345:51–58. https://doi.org/10.1016/j.ijpharm.2007.05.036
Sohaebuddin SK, Thevenot PT, Baker D, Eaton JW, Tang L (2010) Nanomaterial cytotoxicity is composition, size, and cell type dependent. Part Fibre Toxicol 7:1–17. https://doi.org/10.1186/1743-8977-7-22
Valle J, Armstrong A, Newman C, Alakhov V, Pietrzynski G, Brewer J, Campbell S, Corrie P, Rowinsky E, Ranson M (2011) A phase 2 study of SP1049C, doxorubicin in P-glycoprotein-targeting pluronics, in patients with advanced adenocarcinoma of the esophagus and gastroesophageal junction. Invest New Drugs 29:1029–1037. https://doi.org/10.1007/s10637-010-9399-1
Wenzel JG, Balaji KS, Koushik K, Navarre C, Duran SH, Rahe CH, Kompella UB (2002) Pluronic® F127 gel formulations of deslorelin and GnRH reduce drug degradation and sustain drug release and effect in cattle. J Controlled Release 85:51–59. https://doi.org/10.1016/S0168-3659(02)00271-7
Zhang L, Parsons DL, Navarre C, Kompella UB (2002) Development and in-vitro evaluation of sustained release poloxamer 407 (P407) gel formulations of ceftiofur. J Controlled Release 85:73–81. https://doi.org/10.1016/S0168-3659(02)00273-0
Acknowledgements
The authors wish to express their thanks to the Director and Head, Biomedical Technology Wing, Sree Chitra Tirunal Institute for Medical Sciences and Technology (Govt. of India), Trivandrum, Kerala, India, for their support and for providing the infrastructure to carry out this work. Ms Swathi S. thanks the Department of Science and Technology, New Delhi, for DST-SCTIMST Summer Scholarships Programme for students at the UG level.
Author information
Authors and Affiliations
Contributions
Megha: Draft, literature search, data collection, data analysis, and interpretation; Swathi: literature search, data collection, and data analysis; Joseph and Vandana: literature search, data collection, and data analysis; Mohanan: design, data analysis, interpretation, and final approval.
Corresponding author
Ethics declarations
Ethics approval
This is an in vitro study. Appropriate ethical approval is taken for all the assays.
Consent to participate
The consent part from the subject is not applicable for this in vitro study.
Consent for publication
All the authors agreed to submit the manuscript in the journal. The same has approved by the parent Institute.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Sundaresan Swathi attended the DST-SCTIMST Summer Scholarship Program for students at the UG level.
Highlights
• MTT assay for cell viability on exposure to Pluronics F-127.
• Detection of nuclear condensation using DAPI staining.
• Evaluation of cytoskeleton intactness using Rhodamine-phalloidin staining.
• Flow cytometric analysis of the live-dead assay using calcein AM-PI.
• Acridine-orange staining for assessing lysosomal integrity.
Rights and permissions
About this article
Cite this article
Megha, K.B., Swathi, S., Joseph, X. et al. Cytocompatibility of Pluronics F-127 on adenocarcinomic human alveolar basal epithelial cells (A549 cells). Environ Sci Pollut Res 29, 71124–71135 (2022). https://doi.org/10.1007/s11356-022-20925-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-20925-4