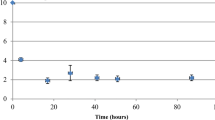
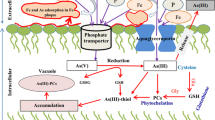
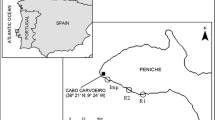
Abstract
Massive Sargassum beachings occurred since 2011 on Caribbean shores. Sargassum inundation events currently involve two species, namely S. fluitans and S. natans circulating and blooming along the North Atlantic subtropical gyre and in the entire Caribbean region up to the Gulf of Mexico. Like other brown seaweeds, Sargassum have been shown to bioaccumulate a large number of heavy metals, alongside with some organic compounds including the contamination by historical chlordecone pollution in French West Indies (FWI), an insecticide used against the banana’s weevil Cosmopolites sordidus. The present study reports, during two successive years, the concentration levels of heavy metals including arsenic in Martinique and Guadeloupe (FWI). We found that Sargassum can also accumulate a high concentration of chlordecone. Sargassum contamination by chlordecone is observed in areas close to contaminated river mouth but can be partly due to chlordecone desorption when secondary drifted on chlordecone-free shore. Our results further demonstrate that algae bleaching raises a number of questions about inorganic and organic pollutant (i) bioaccumulation, at sea for arsenic and close to river plumes for chlordecone, (ii) transport, and (iii) dissemination, depending the shoreline and the speciation for arsenic and/or metabolization for both.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article (and its supplementary information files).
References
Al Mamun MA, Omori Y, Papry RI, Kosugi C, Miki O, Rahman IMM, Mashio AS, Maki T, Hasegawa H (2018) Bioaccumulation and biotransformation of arsenic by the brown macroalga Sargassum patens C. Agardh in seawater: effects of phosphate and iron ions. J Appl Phycol 31(4):2669–2685
Benson AA, Phytoplankton solved the arsenate-phosphate problem. Marine phytoplankton and productivity. Proc. symposium, Taormina 1984; 55-59
Bertilsson S, Widenfalk A (2002) Photochemical degradation of PAHs in freshwaters and their impact on bacterial growth—influence of water chemistry. Hydrobiology 469:23–32
Brown AM. Arsenic speciation in the presence of anoxic mixed valent iron systems. MS (Master of Science) thesis, 2010. University of Iowa.
Brylinsky M (1977) Release of dissolved organic matter by some marine macrophytes. Marine Biol 39:213–220
Bocquené G, Franco A (2005) Pesticide contamination of the coastline of Martinique. Marine Pollut Bull 51(5-7):612–619
Bonanno G, Orlando-Bonaca M (2018) Chemical elements in Mediterranean macroalgae. A review. Ecotoxicol Environ Safety 148:44–71
Bosma TNP, Middeldorp PJM, Schraa G, Zehnder AJB (1997) Mass transfer limitation of biotransformation: quantifying bioavailability. Environ Sci Technol 34:3174–3179
Cavelier N (1980) Contamination of fauna by organochlorine pesticides. In: Actual level of the contamination of biological chains in Guadeloupe by pesticides and heavy metals: 1979-1980. A. Kermarrec (Ed.), Report established for the French Ministry of Environment. pp. 114-128 In French
Chevallier E, Chekri R, Zinck J, Guérin T, Noël L (2015) Simultaneous determination of 31 elements in foodstuffs by ICP-MS after closed-vessel microwave digestion: method validation based on the accuracy profile. J Food Compos Anal 41:35–41
Chiffre A, Degiorgi F, Morin-Crini N, Bolard A, Chanez E, Badot P-M (2015) PAH occurrence in chalk river systems from the Jura region (France). Pertinence of suspended particulate matter and sediment as matrices for river quality monitoring. Environ Sci Pollut Res 22:17486–17498
Coat S, Bocquené G, Godard E (2006) Contamination of some aquatic species with the organochlorine pesticide chlordecone in Martinique. Aqua Living Res 19:181–187
Coat S, Monti D, Legendre P, Bouchon C, Massat F, Lepoint G (2011) Organochlorine pollution in tropical rivers (Guadeloupe): role of ecological factors in food web bioaccumulation. Environ Pollut 159:1692–1701
Crabit A, Cattan P, Colin F, Voltz M (2016) Soil and river contamination patterns of chlordecone in a tropical volcanic catchment in the French West Indies (Guadeloupe). Environ Pollut 212:615–626
Cui X, Mayer P, Gan J (2013) Methods to assess bioavailability of hydrophobic organic contaminants: principles, operations, and limitations. Environ Pollut 172:223–234
Devault DA, Laplanche C, Pascaline H, Mouvet C, Macarie H (2016) Natural transformation of chlordecone into 5b-hydrochlordecone in French West Indies soils: statistical evidence for investigating long-term persistence of organic pollutants. Environ Sci Pollut Res 23(1):81–97
Devault DA, Pierre R, Marfaing H, Dolique F, Lopez P-J Sargassum contamination and consequences for downstream uses: a review. J Appl Phycol 2020a In Press
Devault DA, Amalric L, Bristeau S, Cruz J, Tapie N, Karolak S, Budzinski H, Lévi Y, Removal efficiency of emerging micropollutants in biofilter wastewater treatment plants in tropical areas. Environ Sci Pollut Res 2020b; In Press
Devault DA, Massat F, Baylet A, Dolique F, Lopez P-J. Arsenic and chlordecone contamination and decontamination toxicokinetics in Sargassum. Environ Sci Pollut Res 2021; In Press.
De Weert J, De La Cal A, Van Den Berg H, Murk A, Langenhoff A, Rijnaarts H, Grotenhuis T (2008) Bioavailability and biodegradation of nonylphenol in sediment determined with chemical and bioanalysis. Environ Toxicol Chem 27(4):778–785
Dia M, Zentar R, Abriak N, Nzihou A, Depelsenaire G, Germeau A (2019) Effect of phosphatation and calcination on the environmental behaviour of sediments. Intl J Sediment Res 34(5):486–495
Doleasha D, Simister R, Campbell S, Marstona M, Bose S, McQueen-Mason SJ, Gomez LD, Gallimore WA, Tonon T (2021) Biomass composition of the golden tide pelagic seaweeds Sargassum fluitans and S. natans (morphotypes I and VIII) to inform valorisation pathways. Sci Total Environ 762:25 143134
Dromard CR, Devault DA, Bouchon-Navaro Y, Allénou J-P, Budzinski H, Cordonnier S, Tapie N, Reynal L, Lemoine S, Thomé J-P, Thouard E, Monti D, Bouchon C. Environmental fate of chlordecone in coastal habitats: recent studies conducted in Guadeloupe and Martinique (Lesser Antilles). Environ Sci Pollut Res 2019; In Press.
Dubuisson C, Héraud F, Leblanc J-C, Gallotti S, Flamand C, Blateau A, Quenel P, Volatier J-L (2007) Impact of subsistence production on the management options to reduce the food exposure of the Martinican population to chlordecone. Reg Toxicolicol Pharmacol 49(1):5–16
Ferguson JF, Gavis J (1972) A review of the arsenic cycle in natural waters. Water Res 6(11):1259–1274
García Seoane R, Ángel Fernández J, Boquete T, Aboal J (2018) Application of macroalgae analysis to assess the natural variability in selected pollution concentrations (N and Hg), and to detect sources of it in coastal environments. Sci Total Environ 650(1):1403–1411
Glorennec P, Peyr C, Poupon J, Oulhote Y, Le Bot B (2010) Identifying sources of lead exposure for children, with lead concentrations and isotope ratios. J Occupational Environ Hyg 7(5):253–260
Gourcy L, Baran N, Vittecoq B (2009) Improving the knowledge of pesticide transfer processes using age-dating tools (CFC, SF6, 3H) in a volcanic island (Martinique, French West Indies). J Contamination Hydrol 108:107–117
Guilayn F, Benbrahim M, Rouez M, Crest M, Patureau D, Jimenez J, 2020 Humic-like substances extracted from different digestates: first trials of lettuce biostimulation in hydroponic culture. Waste Manag. 104 (1) 239-245.
Han C, Cao X, Yu J-J, Wang X-R, Shen Y (2009) Arsenic speciation in Sargassum fusiforme by microwave-assisted extraction and LCICP-MS. Chromatography 69:587–591
Héry M, Gault AG, Rowland HAL, Lear G, Polya DA, Lloyd JR (2008) Molecular and cultivation-dependent analysis of metal-reducing bacteria implicated in arsenic mobilisation in South-East Asian aquifers. Appl Geochem 23(11):3215–3223
Howard AG, Comber SDW (1992) Hydride-trapping techniques for the speciation of arsenic. Mikrochimica Acta 109(1-4):27–33
Jondreville C, Lavigne A, Jurjanz S, Dalibard C, Liabeuf JM, Clostre F, Lesueur-Jannoyer M (2014) Contamination of free-range ducks by chlordecone in Martinique (French West Indies): a field study. Sci Total Environ 493:336–341
Kraaij H, Mayer P, Busser F, Van het Bolscher M, Seinen W, Tolls J, Belfroid AC (2003) Measured pore-water concentrations make equilibrium partitioning work—a data analysis. Enviro Sci Technol 37:268–275
Kukkonen JVK, Landrum PF, Mitra S, Gossiaux DC, Gunnarsson J, Weston D (2004) The role of desorption for describing the bioavailability of select polycyclic aromatic hydrocarbon and polychlorinated biphenyl congeners for seven laboratory-spiked sediments. Environ Toxicol Chem 23(8):1842–1851
Lapointe BE, Hanisak MD (1985) Productivity and nutrition of marine biomass systems in Florida. Symposium Papers – Energy from Biomass and Wastes.:111–126
Landrum PF, Robinson SD, Gossiaux DC, You J, Lydy MJ, Mitra S, Hulscher T (2007) TEM predicting bioavailability of sediment-associated organic contaminants for Diporeia spp. and oligochaetes. Enviro. Sci Technol 41(18):6442–6447
Langin K (2018) Seaweed masses assault Caribbean islands. Science 360(6394):1157–1158
Leal-Acosta ML, Shumilin E, Mirlean N, Delgadillo-Hinojosa F, Sánchez-Rodríguez I (2013) The impact of marine shallow-water hydrothermal venting on arsenic and mercury accumulation by seaweed Sargassum sinicola in Concepcion Bay, Gulf of California. Environ Sci Process Impacts 15:470–477
Le Déaut J-Y, Procaccia C, Pesticide use in the Antilles: current situation and perspectives for change. OPECST report n° 487 (2008-2009). French Senat. ISBN: 9782111267688. 2009; 223 pp. In French
Leppänen MT, Kukkonen JVK Effect of sediment-chemical contact time on availability of sediment-associated pyrene and benzo[a]pyrene to oligochaete worms and semi-permeable membrane devices. Aqua Toxicol 2000; 49 (4): 227-241.
Leppänen MT, Landrum PF, Kukkonen JVK, Greenberg MS, Burton GA Jr, Robinson SD, Gossiaux DC (2003) Investigating the role of desorption on the bioavailability of sediment-associated 3,4,3′,4′-tetrachlorobiphenyl in benthic invertebrates. Environ Toxicol Chem 22(12):2861–2871
Leppänen MT, Kukkonen JVK (2006) Evaluating the role of desorption in bioavailability of sediment-associated contaminants using oligochaetes, semipermeable membrane devices and Tenax extraction. Environ Pollut 140(1):150–163
Loffler Z, Hoey AS (2017) Canopy-forming macroalgal beds (Sargassum) on coral reefs are resilient to physical disturbance. J Ecol 106:1156–1164
Magura J, Moodley R, Jonnalagadda SB (2019) Toxic metals (As and Pb) in Sargassum elegans Suhr (1840) and its bioactive compounds. Intl J Environ Health Res 29(3):266–275
Maher W, Foster S, Krikowa F (2009) Arsenic species in Australian temperate marine food chains. Marine Freshw Res 60:885–892
Malea P, Kevrekidis T (2014) Trace element patterns in marine macroalgae. Sci Total Enviro 494-495:144–157
Marmorino GO, Miller WD, Smith GB, Bowles JH (2011) Airborne imagery of a disintegrating Sargassum drift line. Deep Sea Research Part I: Oceanographic Research Papers 58(3):316–321
Méndez-Fernandez P, Kiszka JJ, Heithaus M, Beal A, Vandersarren G, Caurant F, Spitz J, Taniguchia S, Montonea RC (2018) From banana fields to the deep blue: assessment of chlordecone contamination of oceanic cetaceans in the eastern Caribbean. Marine Pollut Bull 137:56–60
Michel P. (1985) Arsenic in marine environment: bibliographic summary. Reu. Trau. Inst. Pèches Marit. 49 (3 and 4), 175-185. In French
Milledge J, Maneein S, Arribas E, Bartlett D (2020) Sargassum inundations in Turks and Caicos: methane potential and proximate, ultimate, lipid, amino acid, metal and metalloid analyses. Energies 13(6):1523–1550
Mink PJ, Alexander DD, Barraj LM, Kelsh MA, Tsuji JS (2008) Low-level arsenic exposure in drinking water and bladder cancer: a review and meta-analysis. Reg Toxicol Pharmacol 52:299–310. https://doi.org/10.1016/j.yrtph.2008.08.010
Modestin E (2020) Consommation de bivalves marins et pollution à l'arsenic : le risque lié à deux espèces comestibles en Martinique. University of French West Indies, pp 372
Morel FMM (1983) Principles of aquatic chemistry. Wiley, New York, pp 300–309
Muse JO, Tudino MB, d'Huicque L, Troccoli OE, Carducci CN (1989) Atomic absorption spectrometric determination of inorganic and organic arsenic in some marine benthic algae of the southern Atlantic coasts. Environ Pollut 58:303–312
Neff JM (1997) Ecotoxicology of arsenic in the marine environment. Environ Toxicol Chem 16(5):917–927
Neff JM (2002) Arsenic in the ocean. In: Bioaccumulation in marine organisms. 468pp. 57-78 Elsevier Ltd ISBN 978-0-08-043716-3.
Niermann U (1986) Distribution of Sargassum natans and some of its epibionts in the Sargasso Sea. Helgoländer Meeresunters 40:343–353
Oxenford HA, Cox S-A, van Tussenbroek BI, Desrochers A (2021) Challenges of turning the Sargassum crisis into gold: current constraints and implications for the Caribbean. Phycology 1-1:27–48
Pell A, Kokkinis G, Malea P, Pergantis SA, Rubio R, López-Sánchez JF (2013) LC–ICP–MS analysis of arsenic compounds in dominant seaweeds from the Thermaikos Gulf (Northern Aegean Sea, Greece). Chemosphere:2187–2194
Pichler T, Amend JP, Garey J, Hallock P, Hsia NP, Karlen DJ, Meyer-Dombard DR, McCloskey BJ, Price RE (2006) A natural laboratory to study arsenic geobiocomplexity. Eos, Trans AGU 87:221–225
Remon E, Bouchardon J-L, Cornier B, Guy B, Leclerc J-C, Faure O (2005) Soil characteristics, heavy metal availability and vegetation recovery at a former metallurgical landfill: implications in risk assessment and site restoration. Environ Pollut 137(2):316–323
Resiere D, Valentino R, Nevière R, Banydeen R, Gueye R, Florentin R, Cabié A, Lebrun T, Mégarbane B, Guerrier G, Mehdaoui H (2018) Sargassum seaweed on Caribbean islands: an international public health concern. The Lancet 392(22-29):2691–2695
Rodríguez-Martínez RE, Roy PD, Torrescano-Valle N, Cabanillas-Terán N, Carrillo-Domínguez S, Collado-Vides L, García-Sánchez M, van Tussenbroek BI (2020) Element concentrations in pelagic Sargassum along the Mexican Caribbean coast in 2018-2019. PeerJ 8:e8667. https://doi.org/10.7717/peerj.8667
Seepersaud MA, Ramkissoon A, Seecharan S, Powder-George YL, Mohammed FK (2018) Environmental monitoring of heavy metals and polycyclic aromatic hydrocarbons (PAHs) in Sargassum filipendula and Sargassum vulgare along the eastern coastal waters of Trinidad and Tobago. West Indies. J Appl Phycol 30:2143–2154
Smedley PL, Kinniburgh DG (2002) A review of the source, behaviour and distribution of arsenic in natural waters. Appl Geochem 17(5):517–568
Stout SA, Litman E, Baker G, Franks JS, Novel biological exposures following the Deepwater Horizon oil spill revealed by chemical fingerprinting. In: Oil spill environmental forensics case studies, Stout S., Wang Z. Eds. 3; 757-784. Butterworth-Heinemann Books, Elsevier, Oxford, UK. 2017; 860 pp.
Tobiszewski M, Namiesnik J (2012) PAH diagnostic ratios for the identification of pollution emission sources. Environ Pollut 162:110–119
Torralba MG, Franks JS, Gomez A, Yooseph S, Nelson KE, Grimes DJ (2017) Effect of Macondo Prospect 252 oil on microbiota associated with pelagic Sargassum in the northern Gulf of Mexico. Microbiol Ecol 73:91–100
van Tussenbroek BI, Hernández Arana HA, Rodríguez-Martínez RE, Espinoza-Avalos J, Canizales-Flores HM, González-Godoy CE, Barba-Santos MG, Vega-Zepeda A, Collado-Vides L, Severe impacts of brown tides caused by Sargassum spp. on near-shore Caribbean seagrass communities. Marine Pollution Bulletin 2017; 122, 1–2 (15): 272-281.
Tsuji JS, Alexander DD, Perez V, Mink PJ (2014) Arsenic exposure and bladder cancer: quantitative assessment of studies in human populations to detect risks at low doses. Toxicology 317:17–30. https://doi.org/10.1016/j.tox.2014.01.004266
Wang M, Hu C, Barnes BB, Mitchum G, Lapointe B, Montoya JP (2019) The great Atlantic Sargassum belt. Science 365(6448):83–87
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York, p 182
Yasmeen A, Qasim M, Ahmed A, Ali MS, Rasheed M (2018) GC-MS and antioxidant studies on botanicals from Sargassum wightii natural product study revealing environmental contaminants. J Chem Soc Pakistan 40(1):2018
Acknowledgements
Authors want to thank the Celia Northam (SIMILITUDE) for English editing. Authors want to thank also Pascal-Jean Lopez and Vincent Hervé for their contribution to conception, sampling analysis of results, and writing of the present article.
Funding
French Energy and Environment Agency (ADEME) granted the study.
Author information
Authors and Affiliations
Contributions
DAD performed the conception, preparation, sampling, experiment, the analysis of results, and the writing of the present article. FM contributed to the preparation, experiment, analysis, and writing. CM and FD contributed to the conception and sampling. JL, LD, and APA contributed to sampling.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix 1. Field contamination: Sargassum content (average, n = 3)
Appendix 1. Field contamination: Sargassum content (average, n = 3)
Table 5
Table 6
Table 7
Rights and permissions
About this article
Cite this article
Devault, D.A., Massat, F., Lambourdière, J. et al. Micropollutant content of Sargassum drifted ashore: arsenic and chlordecone threat assessment and management recommendations for the Caribbean. Environ Sci Pollut Res 29, 66315–66334 (2022). https://doi.org/10.1007/s11356-022-20300-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-20300-3