Abstract
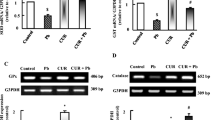
The purpose of this study was to explore the interventional effects of folic acid on the heart damage caused by lead acetate exposure. Twenty-four 60-day-old male Sprague–Dawley (SD) rats were randomly divided into 4 groups with 6 rats in each group. The control group (C group) was normal rats; the lead exposure group (L group) rats drank 0.2% lead acetate solution freely for 14 days. The rats in the intervention group (T group) were given 0.2% lead acetate solution for 14 days, respectively, and 0.4 mg/kg BW folic acid solution was given to the rats by gavage on the 7th day of lead administration. The rats in the folic acid group (group E) were given 0.4 mg/kg BW folic acid solution by gavage. To weigh rat body weight and heart weight, calculate heart index, and observe the expression level of nuclear factor erythroid 2-related factor 2(Nrf2), heme oxygenase 1(HO-1), glucose-regulated protein 78/binding immunoglobulin protein (GRP78), and C/EBP-homologous protein (CHOP) by immunofluorescence method. The results showed that compared with group C, serum lead levels in group L and T were significantly increased (P < 0.05); superoxide dismutase (SOD), glutathione (GSH), and glutathione peroxidase (GSH-PX) levels in group L were significantly decreased (P < 0.05), and malondialdehyde (MDA) content was significantly higher increased (P < 0.05), and the GSH-PX content in group T were significantly increased in group L (P < 0.05), and the MDA content in group T was significantly lower than that in group L (P < 0.05). Compared with group C, the expression of Nrf2, HO-1, GRP78, and CHOP in group L increased significantly, and the difference was statistically significant (P < 0.05). Compared with the L group, the expression of Nrf2, HO-1, GRP78, and CHOP in the T group was reduced. Therefore, folic acid has a certain protective effect on the oxidative damage of lead-exposed rat heart tissue.
Graphical abstract
Lead exposure will increase ROS, NO, MDA, and other oxidizing substances and reduce the level of GSH, SOD, CAT, GPx, and other antioxidant factors, which will lead to cardiac hypertrophy, cardiac index increase, oxidative stress, Nrf2, and HO-1. The expression of stress-related proteins such as GRP78 and CHOP also increased, leading to cardiomyocyte apoptosis. After a folic acid intervention, these changes can be significantly reversed.






Similar content being viewed by others
Data availability
All data generated during this study are available from the corresponding author on request.
Abbreviations
- ROS:
-
Reactive oxygen species
- Nrf2:
-
Nuclear factor erythroid 2-related factor 2
- HO-1:
-
Heme oxygenase 1
- ERS:
-
Endoplasmic reticulum stress
- GRP78:
-
Glucose-regulated protein 78/binding immunoglobulin protein
- ER:
-
Endoplasmic reticulum
- CHOP:
-
C/EBP-homologous protein
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- GSH:
-
Glutathione
- GSH-PX:
-
Glutathione peroxidase
- GSR:
-
Glutathione reductase
- SOD:
-
Superoxide dismutase
- MDA:
-
Malondialdehyde
- DOXO:
-
Doxorubicin
- TNF-α:
-
Tumor necrosis factor-α
- CO2 :
-
Carbon dioxide
- SSA:
-
Specific surface area
- Keap1:
-
Kelch-like erythrocyte-derived protein 1;
- ERK1/2:
-
Extracellular signal-regulated kinase
- AKT:
-
Phosphoinositide3-kinase(PI3K)/protein kinase B
- P38:
-
Protein kinase
- JNK:
-
C-Jun NH2-terminal kinase
- CO:
-
Carbon monoxide
- Hhcy:
-
Hyperhomocysteinemia
- RON:
-
Reactive nitrogen
- Hcy:
-
Homocysteine
References
Abdel Moneim AE (2016) Indigofera oblongifolia prevents lead acetate-induced hepatotoxicity, oxidative stress, fibrosis and apoptosis in rats. PLoS ONE 11:e0158965. https://doi.org/10.1371/journal.pone.0158965
Afridi HI, Kazi TG, Kazi N, Kandhro GA, Baig J A, Jamali MK, . . . Kolachi NF (2011) Association of environmental toxic elements in biological samples of myocardial infarction patients at different stages. Biol Trace Elem Res 141:26–40. https://doi.org/10.1007/s12011-010-8713-2
Ahmad S, Panda BP, Kohli K, Fahim M, Dubey K (2017) Folic acid ameliorates celecoxib cardiotoxicity in a doxorubicin heart failure rat model. Pharm Biol 55:1295–1303. https://doi.org/10.1080/13880209.2017.1299768
Akgun E, Boyacioglu M, Kum S (2021) The potential protective role of folic acid against acetaminophen-induced hepatotoxicity and nephrotoxicity in rats. Exp Anim 70:54–62. https://doi.org/10.1538/expanim.20-0075
Albarakati AJA, Baty RS, Aljoudi AM, Habotta OA, Elmahallawy EK, Kassab RB, Abdel Moneim AE (2020) Luteolin protects against lead acetate-induced nephrotoxicity through antioxidant, anti-inflammatory, anti-apoptotic, and Nrf2/HO-1 signaling pathways. Mol Biol Rep 47:2591–2603. https://doi.org/10.1007/s11033-020-05346-1
Aleksunes LM, Goedken MJ, Rockwell CE, Thomale J, Manautou JE, Klaassen CD (2010) Transcriptional regulation of renal cytoprotective genes by Nrf2 and its potential use as a therapeutic target to mitigate cisplatin-induced nephrotoxicity. J Pharmacol Exp Ther 335:2–12. https://doi.org/10.1124/jpet.110.170084
Azfer A, Niu J, Rogers LM, Adamski FM, Kolattukudy PE (2006) Activation of endoplasmic reticulum stress response during the development of ischemic heart disease. Am J Physiol Heart Circ Physiol 291:H1411-1420. https://doi.org/10.1152/ajpheart.01378.2005
Babiker F, Al-Kouh A, Kilarkaje N (2019) Lead exposure induces oxidative stress, apoptosis, and attenuates protection of cardiac myocytes against ischemia-reperfusion injury. Drug Chem Toxicol 42:147–156. https://doi.org/10.1080/01480545.2018.1429460
Bener A, Obineche E, Gillett M, Pasha MA, Bishawi B (2001) Association between blood levels of lead, blood pressure and risk of diabetes and heart disease in workers. Int Arch Occup Environ Health 74:375–378. https://doi.org/10.1007/s004200100231
Brychtová V, Vojtěšek B (2016) Endoplasmic reticulum chaperones at the tumor cell surface and in the extracellular space. Klin Onkol 29:4S25-24S30. https://doi.org/10.14735/amko20164S25
Budni J, Zomkowski AD, Engel D, Santos DB, dos Santos AA, Moretti M, . . . Rodrigues AL (2013) Folic acid prevents depressive-like behavior and hippocampal antioxidant imbalance induced by restraint stress in mice. Exp Neurol 240:112–121. https://doi.org/10.1016/j.expneurol.2012.10.024
Cao P, Zhang W, Wang G, Zhao X, Gao N, Liu Z, Xu R (2021) Low dose of folic acid can ameliorate hyperhomocysteinemia-induced cardiac fibrosis and diastolic dysfunction in spontaneously hypertensive rats. Int Heart J 62:627–635. https://doi.org/10.1536/ihj.20-593
Chen J, Zhuang T, Chen J, Tian Y, Yi X, Ni Q, . . . Li S (2020) Homocysteine induces melanocytes apoptosis via PERK-eIF2alpha-CHOP pathway in vitiligo. Clin Sci (Lond) 134:1127–1141. https://doi.org/10.1042/CS20200218
Chen Z, Huo X, Chen G, Luo X, Xu X (2021) Lead (Pb) exposure and heart failure risk. Environ Sci Pollut Res Int 28:28833–28847. https://doi.org/10.1007/s11356-021-13725-9
Debski B, Bertrandt J, Klos A, Gralak M (2006) Influence of folic acid, vitamin B2 and B6 supplementation on feed intake, body and organs weight, and liver fatty acids composition in rats subjected to severe protein deprivation. Pol J Vet Sci 9:185–190
Dickhout JG, Carlisle RE, Austin RC (2011) Interrelationship between cardiac hypertrophy, heart failure, and chronic kidney disease: endoplasmic reticulum stress as a mediator of pathogenesis. Circ Res 108:629–642. https://doi.org/10.1161/CIRCRESAHA.110.226803
Djuric D, Vusanovic A, Jakovljevic V (2007) The effects of folic acid and nitric oxide synthase inhibition on coronary flow and oxidative stress markers in isolated rat heart. Mol Cell Biochem 300:177–183. https://doi.org/10.1007/s11010-006-9381-6
Dudka I, Kossowska B, Senhadri H, Latajka R, Hajek J, Andrzejak R, . . . Gancarz R (2014) Metabonomic analysis of serum of workers occupationally exposed to arsenic, cadmium and lead for biomarker research: a preliminary study. Environ Int 68:71-81. https://doi.org/10.1016/j.envint.2014.03.015
Elgharabawy RM, Alhowail AH, Emara AM, Aldubayan MA, Ahmed AS (2021) The impact of chicory (Cichoriumintybus L.) on hemodynamic functions and oxidative stress in cardiac toxicity induced by lead oxide nanoparticles in male rats. Biomed Pharmacother 137:111324. https://doi.org/10.1016/j.biopha.2021.111324
Fedoroff N (2006) Redox regulatory mechanisms in cellular stress responses. Ann Bot 98:289–300. https://doi.org/10.1093/aob/mcl128
Feng L, Yang X, Shi Y, Liang S, Zhao T, Duan J, Sun Z (2018) Co-exposure subacute toxicity of silica nanoparticles and lead acetate on cardiovascular system. Int J Nanomedicine 13:7819–7834. https://doi.org/10.2147/IJN.S185259
Gething MJ (1999) Role and regulation of the ER chaperone BiP. Semin Cell Dev Biol 10:465–472. https://doi.org/10.1006/scdb.1999.0318
Gump BB, Mackenzie JA, Bendinskas K, Morgan R, Dumas AK, Palmer CD, Parsons PJ (2011) Low-level Pb and cardiovascular responses to acute stress in children: the role of cardiac autonomic regulation. Neurotoxicol Teratol 33:212–219. https://doi.org/10.1016/j.ntt.2010.10.001
Hagar HH (2002) Folic acid and vitamin B12 supplementation attenuates isoprenaline-induced myocardial infarction in experimental hyperhomocysteinemic rats. Pharmacol Res 46:213–219. https://doi.org/10.1016/s1043-6618(02)00095-6
Hirsch HVB, Possidente D, Possidente B (2010) Pb2+: An endocrine disruptor in drosophila? Physiol Behav 99:254–259. https://doi.org/10.1016/j.physbeh.2009.09.014
Jain NB, Potula V, Schwartz J, Vokonas PS, Sparrow D, Wright RO, . . . Hu H (2007) Lead levels and ischemic heart disease in a prospective study of middle-aged and elderly men: the VA normative aging study. Environ Health Perspect 115:871–875. https://doi.org/10.1289/ehp.9629
Ji C, Kaplowitz N (2004) Hyperhomocysteinemia, endoplasmic reticulum stress, and alcoholic liver injury. World J Gastroenterol 10:1699–1708. https://doi.org/10.3748/wjg.v10.i12.1699
Jiang X, Yu W, Wu S, Tang L, Zhong G, Wan F, . . . Huang R (2021) Arsenic (III) and/or Antimony (III) induced disruption of calcium homeostasis and endoplasmic reticulum stress resulting in apoptosis in mice heart. Ecotoxicol Environ Saf 220:112394. https://doi.org/10.1016/j.ecoenv.2021.112394
Joseph J, Loscalzo J (2013) Methoxistasis: integrating the roles of homocysteine and folic acid in cardiovascular pathobiology. Nutrients 5:3235–3256. https://doi.org/10.3390/nu5083235
Joshi R, Adhikari S, Patro BS, Chattopadhyay S, Mukherjee T (2001) Free radical scavenging behavior of folic acid: evidence for possible antioxidant activity. Free Radic Biol Med 30:1390–1399. https://doi.org/10.1016/s0891-5849(01)00543-3
Kaufman RJ (1999) Stress signaling from the lumen of the endoplasmic reticulum: coordination of gene transcriptional and translational controls. Genes Dev 13:1211–1233. https://doi.org/10.1101/gad.13.10.1211
Kensler TW, Wakabayashi N, Biswal S (2007) Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu Rev Pharmacol Toxicol 47:89–116. https://doi.org/10.1146/annurev.pharmtox.46.120604.141046
Klinova SV, Katsnelson BA, Minigalieva IA, Gerzen OP, Balakin AA, Lisin R V, . . . Protsenko YL (2021) Cardioinotropic effects in subchronic intoxication of rats with lead and/or cadmium oxide nanoparticles. Int J Mol Sci 22. https://doi.org/10.3390/ijms22073466
Kolling J, Scherer EB, da Cunha AA, da Cunha MJ, Wyse AT (2011) Homocysteine induces oxidative-nitrative stress in heart of rats: prevention by folic acid. Cardiovasc Toxicol 11:67–73. https://doi.org/10.1007/s12012-010-9094-7
Koohpeyma H, Goudarzi I, Elahdadi Salmani M, Lashkarbolouki T, Shabani M (2020) Folic acid protects rat cerebellum against oxidative damage caused by homocysteine: the expression of Bcl-2, Bax, and Caspase-3 apoptotic genes. Neurotox Res 37:564–577. https://doi.org/10.1007/s12640-019-00119-6
Lee AS (2005) The ER chaperone and signaling regulator GRP78/BiP as a monitor of endoplasmic reticulum stress. Methods 35:373–381. https://doi.org/10.1016/j.ymeth.2004.10.010
Lee MG, Chun OK, Song WO (2005) Determinants of the blood lead level of US women of reproductive age. J Am Coll Nutr 24:1–9. https://doi.org/10.1080/07315724.2005.10719436
Lee SE, Yang H, Jeong SI, Jin YH, Park CS, Park YS (2012) Induction of heme oxygenase-1 inhibits cell death in crotonaldehyde-stimulated HepG2 cells via the PKC-delta-p38-Nrf2 pathway. PLoS ONE 7:e41676. https://doi.org/10.1371/journal.pone.0041676
Li N, Zhang D, Cao S, Qiao M, Zhang P, Zhao Q, . . . Song L (2021a) The effects of folic acid on RNA m6A methylation in hippocampus as well as learning and memory ability of rats with acute lead exposure. J Funct Foods 76. https://doi.org/10.1016/j.jff.2020.104276
Li N, Zhao Y, Shen Y, Cheng Y, Qiao M, Song L, Huang X (2021b) Protective effects of folic acid on oxidative damage of rat spleen induced by lead acetate. Ecotoxicol Environ Saf 211:111917. https://doi.org/10.1016/j.ecoenv.2021.111917
LI Ning, CAO Shuai, SHEN Yue, ZHANG Di, SONG Lianjun, HUANG Xianqing, . . . Fan., Z (2019) Effects of folic acid on liver oxidative damage in lead-exposed rats. Anim Nutr 31:5178–5183
Li X, Ye F, Li L, Chang W, Wu X, Chen J (2016) The role of HO-1 in protection against lead-induced neurotoxicity. Neurotoxicology 52:1–11. https://doi.org/10.1016/j.neuro.2015.10.015
Long M, Liu Y, Cao Y, Wang N, Dang M, He J (2016) Proanthocyanidins attenuation of chronic lead-induced liver oxidative damage in Kunming mice via the Nrf2/ARE pathway. Nutrients 8. https://doi.org/10.3390/nu8100656
Lu F, Zhao LY, Zhang ZM, Zou Q, Yu XL, Wei CY (2018) The intervention of enalapril maleate and folic acid tablet on the expressions of the GRP78 and CHOP and vascular remodeling in the vascular smooth muscle cells of H-hypertensive rats with homocysteine. Eur Rev Med Pharmacol Sci 22:2160–2168. https://doi.org/10.26355/eurrev_201804_14750
Ma Q, Liu Y, Chen L (2018) JIP3 deficiency attenuates cardiac hypertrophy by suppression of JNK pathway. Biochem Biophys Res Commun 503:1–7. https://doi.org/10.1016/j.bbrc.2018.03.208
Ma Y, Hendershot LM (2004) The role of the unfolded protein response in tumour development: friend or foe? Nat Rev Cancer 4:966–977. https://doi.org/10.1038/nrc1505
Majumdar S, Maiti A, Karmakar S, Das AS, Mukherjee S, Das D, Mitra C (2012) Antiapoptotic efficacy of folic acid and vitamin B(1)(2) against arsenic-induced toxicity. Environ Toxicol 27:351–363. https://doi.org/10.1002/tox.20648
Manoj Kumar V, Henley AK, Nelson CJ, Indumati O, Prabhakara Rao Y, Rajanna S, Rajanna B (2017) Protective effect of Allium sativum (garlic) aqueous extract against lead-induced oxidative stress in the rat brain, liver, and kidney. Environ Sci Pollut Res Int 24:1544–1552. https://doi.org/10.1007/s11356-016-7923-3
Mao W, Iwai C, Qin F, Liang CS (2005) Norepinephrine induces endoplasmic reticulum stress and downregulation of norepinephrine transporter density in PC12 cells via oxidative stress. Am J Physiol Heart Circ Physiol 288:H2381-2389. https://doi.org/10.1152/ajpheart.00904.2004
Martinez E, Deval C, Jousse C, Mazur A, Brachet P, Comte B (2015) Methyl donor deficiency in H9c2 cardiomyoblasts induces ER stress as an important part of the proteome response. Int J Biochem Cell Biol 59:62–72. https://doi.org/10.1016/j.biocel.2014.11.013
Minamino T, Kitakaze M (2010) ER stress in cardiovascular disease. J Mol Cell Cardiol 48:1105–1110. https://doi.org/10.1016/j.yjmcc.2009.10.026
Moens AL, Vrints CJ, Claeys MJ, Timmermans JP, Champion HC, Kass DA (2008) Mechanisms and potential therapeutic targets for folic acid in cardiovascular disease. Am J Physiol Heart Circ Physiol 294:H1971-1977. https://doi.org/10.1152/ajpheart.91503.2007
Molero L, Carrasco C, Marques M, Vaziri ND, Mateos-Caceres PJ, Casado S, . . . Lopez-Farre AJ (2006) Involvement of endothelium and endothelin-1 in lead-induced smooth muscle cell dysfunction in rats. Kidney Int 69:685–690. https://doi.org/10.1038/sj.ki.5000103
Na L, Bai Q, Xiumei Z, Lingzi Z, Deqin H, Xuanxuan Z, Huanhuan G, Yuan L, Xiujuan C (2020) Research into the intervention effect of folic acid on arsenic-induced heart abnormalities in fetal rats during the periconception period. BMC Cardiovasc Disord 20:139. https://doi.org/10.1186/s12872-020-01418-z
Nam SM, Ahn SC, Go TH, Seo JS, Nahm SS, Chang BJ, Lee JH (2018) Ascorbic acid ameliorates gestational lead exposure-induced developmental alteration in GAD67 and c-Kit expression in the rat cerebellar cortex. Biol Trace Elem Res 182:278–286. https://doi.org/10.1007/s12011-017-1086-z
Navarro-Moreno LG, Quintanar-Escorza MA, Gonzalez S, Mondragon R, Cerbon-Solorzano J, Valdes J, Calderon-Salinas JV (2009) Effects of lead intoxication on intercellular junctions and biochemical alterations of the renal proximal tubule cells. Toxicol in Vitro 23:1298–1304. https://doi.org/10.1016/j.tiv.2009.07.020
Ngwenyama N, Salvador AM, Velazquez F, Nevers T, Levy A, Aronovitz M, . . . Alcaide P (2019) CXCR3 regulates CD4+ T cell cardiotropism in pressure overload-induced cardiac dysfunction. JCI Insight 4. https://doi.org/10.1172/jci.insight.125527
Nolan J, Batin PD, Andrews R, Lindsay SJ, Brooksby P, Mullen M, . . . Fox KA (1998) Prospective study of heart rate variability and mortality in chronic heart failure: results of the United Kingdom heart failure evaluation and assessment of risk trial (UK-heart). Circulation 98:1510–1516. https://doi.org/10.1161/01.cir.98.15.1510
Octavia Y, Kararigas G, de Boer M, Chrifi I, Kietadisorn R, Swinnen M, . . . Moens AL (2017) Folic acid reduces doxorubicin-induced cardiomyopathy by modulating endothelial nitric oxide synthase. J Cell Mol Med 21:3277–3287. https://doi.org/10.1111/jcmm.13231
Okada K, Minamino T, Tsukamoto Y, Liao Y, Tsukamoto O, Takashima S, . . . Kitakaze M (2004) Prolonged endoplasmic reticulum stress in hypertrophic and failing heart after aortic constriction: possible contribution of endoplasmic reticulum stress to cardiac myocyte apoptosis. Circulation 110:705–712. https://doi.org/10.1161/01.CIR.0000137836.95625.D4
Petrovski G, Das S, Juhasz B, Kertesz A, Tosaki A, Das DK (2011) Cardioprotection by endoplasmic reticulum stress-induced autophagy. Antioxid Redox Signal 14:2191–2200. https://doi.org/10.1089/ars.2010.3486
Pinho-Gomes AC, Rahimi K (2019) Management of blood pressure in heart failure. Heart 105:589–595. https://doi.org/10.1136/heartjnl-2018-314438
Piquereau J, Moulin M, Zurlo G, Mateo P, Gressette M, Paul J L, . . . Garnier A (2017) Cobalamin and folate protect mitochondrial and contractile functions in a murine model of cardiac pressure overload. J Mol Cell Cardiol 102: 34–44. https://doi.org/10.1016/j.yjmcc.2016.11.010
Pobre KFR, Poet GJ, Hendershot LM (2019) The endoplasmic reticulum (ER) chaperone BiP is a master regulator of ER functions: getting by with a little help from ERdj friends. J Biol Chem 294:2098–2108. https://doi.org/10.1074/jbc.REV118.002804
Ponce-Canchihuaman JC, Perez-Mendez O, Hernandez-Munoz R, Torres-Duran PV, Juarez-Oropeza MA (2010) Protective effects of Spirulina maxima on hyperlipidemia and oxidative-stress induced by lead acetate in the liver and kidney. Lipids Health Dis 9:35. https://doi.org/10.1186/1476-511X-9-35
Qin L, Wang W, You S, Dong J, Zhou Y, Wang J (2014) In vitro antioxidant activity and in vivo antifatigue effect of layered double hydroxide nanoparticles as delivery vehicles for folic acid. Int J Nanomedicine 9:5701–5710. https://doi.org/10.2147/IJN.S74306
Qu W, Du GL, Feng B, Shao H (2019) Effects of oxidative stress on blood pressure and electrocardiogram findings in workers with occupational exposure to lead. J Int Med Res 47:2461–2470. https://doi.org/10.1177/0300060519842446
Quan FS, Yu XF, Gao Y, Ren WZ (2015) Protective effects of folic acid against central nervous system neurotoxicity induced by lead exposure in rat pups. Genet Mol Res 14:12466–12471. https://doi.org/10.4238/2015.October.16.13
Rahman A, Khan KM, Rao MS (2018) Exposure to low level of lead during preweaning period increases metallothionein-3 expression and dysregulates divalent cation levels in the brain of young rats. Neurotoxicology 65:135–143. https://doi.org/10.1016/j.neuro.2018.02.008
Ricchetti GA, Williams LM, Foxwell BM (2004) Heme oxygenase 1 expression induced by IL-10 requires STAT-3 and phosphoinositol-3 kinase and is inhibited by lipopolysaccharide. J Leukoc Biol 76:719–726. https://doi.org/10.1189/jlb.0104046
Ron D (2002) Translational control in the endoplasmic reticulum stress response. J Clin Investig 110:1383–1388. https://doi.org/10.1172/jci200216784
Shukla PK, Khanna VK, Khan MY, Srimal RC (2003) Protective effect of curcumin against lead neurotoxicity in rat. Hum Exp Toxicol 22:653–658. https://doi.org/10.1191/0960327103ht411oa
Simoes MR, Preti SC, Azevedo BF, Fiorim J, Freire DD Jr, Covre EP, . . . Dos Santos L (2017) Low-level chronic lead exposure impairs neural control of blood pressure and heart rate in rats. Cardiovasc Toxicol 17: 190–199. https://doi.org/10.1007/s12012-016-9374-y
Simoes MR, Ribeiro Junior RF, Vescovi MV, de Jesus HC, Padilha AS, StefanonI, . . . Fioresi M (2011) Acute lead exposure increases arterial pressure: role of the renin-angiotensin system. PLoS One 6:e18730. https://doi.org/10.1371/journal.pone.0018730
Sitia R, Molteni SN (2004) Stress, protein (mis)folding, and signaling: the redox connection. Sci STKE 2004:pe27. https://doi.org/10.1126/stke.2392004pe27
Sun B, Zhang X, Yin Y, Sun H, Ge H, Li W (2017) Effects of sulforaphane and vitamin E on cognitive disorder and oxidative damage in lead-exposed mice hippocampus at lactation. J Trace Elem Med Biol 44:88–92. https://doi.org/10.1016/j.jtemb.2017.06.004
Swaminathan PD, Purohit A, Hund TJ, Anderson ME (2012) Calmodulin-dependent protein kinase II: linking heart failure and arrhythmias. Circ Res 110:1661–1677. https://doi.org/10.1161/CIRCRESAHA.111.243956
Tong S, Von Schirnding YE, Prapamontol T (2000) Environmental lead exposure: a public health problem with global dimensions. Servir 49:35–43
van der Pol A, van Gilst WH, Voors AA, van der Meer P (2019) Treating oxidative stress in heart failure: past, present and future. Eur J Heart Fail 21:425–435. https://doi.org/10.1002/ejhf.1320
Vaziri ND, Lin CY, Farmand F, Sindhu RK (2003) Superoxide dismutase, catalase, glutathione peroxidase and NADPH oxidase in lead-induced hypertension. Kidney Int 63:186–194. https://doi.org/10.1046/j.1523-1755.2003.00711.x
Wang G, DiBari J, Bind E, Steffens AM, Mukherjee J, Azuine R E, . . . Wang X (2019) Association between maternal exposure to lead, maternal folate status, and intergenerational risk of childhood overweight and obesity. JAMA Netw Open 2:e1912343. https://doi.org/10.1001/jamanetworkopen.2019.12343
Waters M, Stasiewicz S, Merrick BA, Tomer K, Bushel P, Paules R, . . . Fostel J (2008) CEBS–chemical effects in biological systems: a public data repository integrating study design and toxicity data with microarray and proteomics data. Nucleic Acids Res 36: D892–900. https://doi.org/10.1093/nar/gkm755
Wu TG, Li WH, Lin ZQ, Wang LX (2008) Effects of folic acid on cardiac myocyte apoptosis in rats with streptozotocin-induced diabetes mellitus. Cardiovasc Drugs Ther 22:299–304. https://doi.org/10.1007/s10557-008-6109-8
Xia S, Duan W, Liu W, Zhang X, Wang Q (2021) GRP78 in lung cancer. J Transl Med 19:118. https://doi.org/10.1186/s12967-021-02786-6
Xu LH, Mu FF, Zhao JH, He Q, Cao CL, Yang H, . . . Sun SJ (2015) Lead induces apoptosis and histone hyperacetylation in rat cardiovascular tissues. PLoS One 10:e0129091. https://doi.org/10.1371/journal.pone.0129091
Yamaji K, Ochiai Y, Ohnishi K, Yawata A, Chikuma T, Hojo H (2008) Up-regulation of hepatic heme oxygenase-1 expression by locally induced interleukin-6 in rats administered carbon tetrachloride intraperitoneally. Toxicol Lett 179:124–129. https://doi.org/10.1016/j.toxlet.2008.04.012
Yang D, Li S, Gao L, Lv Z, Bing Q Lv Q, . . . Zhang Z (2018) Dietary grape seed procyanidin extract protects against lead-induced heart injury in rats involving endoplasmic reticulum stress inhibition and AKT activation. J Nutr Biochem 62:43–49. https://doi.org/10.1016/j.jnutbio.2018.07.013
Ye F, Li X, Li L, Yuan J, Chen J (2016) t-BHQ provides protection against lead neurotoxicity via Nrf2/HO-1 pathway. Oxid Med Cell Longev 2016:2075915. https://doi.org/10.1155/2016/2075915
Yu C, Pan S, Zhang J, Li X, Niu Y (2021) Ferulic acid exerts Nrf2-dependent protection against prenatal lead exposure-induced cognitive impairment in offspring mice. J Nutr Biochem 91:108603. https://doi.org/10.1016/j.jnutbio.2021.108603
Zhang L, Zou L, Jiang X, Cheng S, Zhang J, Qin X, . . . Zou Z (2021) Stabilization of Nrf2 leading to HO-1 activation protects against zinc oxide nanoparticles-induced endothelial cell death. Nanotoxicology 15:779–797. https://doi.org/10.1080/17435390.2021.1919330
Zhang X, Lu L, Dixon C, Wilmer W, Song H, Chen X, Rovin BH (2004) Stress protein activation by the cyclopentenone prostaglandin 15-deoxy-delta12,14-prostaglandin J2 in human mesangial cells. Kidney Int 65:798–810. https://doi.org/10.1111/j.1523-1755.2004.00454.x
Zhou X, Lu B, Fu D, Gui M, Yao L, Li J (2020) Huoxue Qianyang decoction ameliorates cardiac remodeling in obese spontaneously hypertensive rats in association with ATF6-CHOP endoplasmic reticulum stress signaling pathway regulation. Biomed Pharmacother 121:109518. https://doi.org/10.1016/j.biopha.2019.109518
Zhu L, Yu J, Jia B, Zhao F, Tang M, Hu L, Lin F (2015) Effect of losartan with folic acid on plasma homocysteine and vascular ultrastructural changes in spontaneously hypertensive rats. Int J Clin Exp Pathol 8:12908–12914
Acknowledgements
The author thanks all co-authors in the team for participating in this study. Thanks to Dr. Ning Li and Dr. Fangyu Wang. Thanks to the School of Food Science and Technology of Henan Agricultural University and the Key Laboratory of Animal Immunology of Henan Academy of Agricultural Sciences for their great assistance.
Funding
This research was supported by the National Natural Science Foundation of China (NNSFC) by grant NNSFC – 31201878, U1204804, and U1604183. It has also been funded by the Excellent Youth Project of the Natural Science Foundation of Henan Province (202300410193), Henan Engineering Technology Research Center of Food Processing and Circulation Safety Control (20171215) and 2022 Henan Province Postgraduate Joint Training Base Project (YJS2022JD16).
Author information
Authors and Affiliations
Contributions
Ning Li and Yali Zhao participated in the design of this study, statistical analysis, drafting of manuscripts, and literature search. Fangyu Wang, Lianjun Song, and Mingwu Qiao conducted experimental paper design, knowledge content definition, document retrieval, data collection, data analysis, and manuscript preparation. Tianlin Wang and Xianqing Huang provided assistance in literature retrieval, data collection, and manuscript editing. All authors read and approve the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All experimental procedures passed the Animal Protection and Utilization Committee of Henan Academy of Agricultural Sciences-in line with the National Institutional Animal Care Guidelines. Try to protect animal welfare and reduce animal suffering (Zhengzhou, China). All co-authors are known and agreed to be published in this journal.
Consent to participate
Not applicable to this study.
Consent for publication
Not applicable to this study.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, N., Zhao, Y., Wang, F. et al. Folic acid alleviates lead acetate-mediated cardiotoxicity by down-regulating the expression levels of Nrf2, HO-1, GRP78, and CHOP proteins. Environ Sci Pollut Res 29, 55916–55927 (2022). https://doi.org/10.1007/s11356-022-19821-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-19821-8