Abstract
Backgrounds
The effect of folic acid on cardiac myocyte apoptosis secondary to diabetes is unknown.
Methods
Diabetic rats were divided into diabetic control (DC, n = 11), low-dose (LDF, 0.4 mg/kg/day, n = 12) and high-dose (HDF, 1.2 mg/kg/day, n = 12) folic acid groups. Non-diabetic rats (n = 11) were used as the normal control (NC).
Results
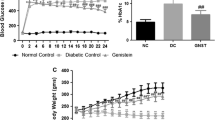
After 11 weeks of treatment, compared with the NC group, the DC group showed a reduced blood levels of reactive oxygen species (ROS, P < 0.01). The rate of cardiac myocyte apoptosis in the diabetic control group was also greater than in the non-diabetic control group (P < 0.01). In folic acid-treated rats, the blood levels of ROS was higher than in the diabetic control group (P < 0.05). There was a dose-dependent reduction in the rate of cardiac myocyte apoptosis in the folic acid groups (P < 0.01), and this was accompanied by an increased level of anti-apoptotic protein Bcl-2 and decreased level of pro-apoptotic protein Bax and Fas (P < 0.01).
Conclusions
Dietary folic acid supplementation diminishes the cardiac myocyte apoptosis in streptozotocin-induced diabetes. The apoptosis suppression is accompanied by an increase in the expression of Bcl-2 and a decrease in Bax and Fas.

Similar content being viewed by others
References
Galderisi M, Anderson KM, Wilson PW, Levy D. Echocardiographic evidence for the existence of a distinct diabetic cardiomyopathy (the Framingham Heart Study). Am J Cardiol. 1991;68:85–9.
An D, Rodrigues B. Role of changes in cardiac metabolism in development of diabetic cardiomyopathy. Am J Physiol—Heart Circ Physiol. 2006;291:H1489–506.
Fiordaliso F, Li B, Latini R, Sonnenblick EH, Anversa P, Leri A, Kajstura J. Myocytes death in streptozotocin-induced diabetes in rats is angiotensin II-dependent. Lab Invest. 2000;80:513–27.
Kajstura J, Fiordaliso F, Andreoli AM, Li B, Chimenti S, Medow MS, Limana F, Nadal-Ginard B, Leri A, Anversa P. IGF-1 overexpression inhibits the development of diabetic cardiomyopathy and angiotensin II-mediated oxidative stress. Diabetes 2001;50:1414–24.
Frustaci A, Kajstura J, Chimenti C, Jakoniuk I, Len A, Maseri A, Nadal-Ginard B, Anversa P. Myocardial cell death in human diabetes. Circ Res. 2000;87:1123–32.
Mangoni AA, Sherwood RA, Asonganyi B, Swift CG, Thomas S, Jackson SH. Short-term oral folic acid supplementation enhances endothelial function in patients with type 2 diabetes. Am J Hyperten. 2005;18:220–6.
Gareskog M, Eriksson UJ, Wetzel P. Combined supplementation of folic acid and vitamin E diminishes diabetes-induced embryotoxicity in rats. Birth Defects Res. 2006;76:483–90.
Craciunescu CN, Brown EC, Mar MH, Albright CD, Nadeau MR, Zeisel SH. Folic acid deficiency during late gestation decreases progenitor cell proliferation and increases apoptosis in fetal mouse brain. J Nutr. 2004;134:162–6.
Lin HL, Chen CJ, Tsai WC, Yen JH, Liu HW. In vitro folate deficiency induces apoptosis by a p53, Fas (Apo-1, CD95) independent, bcl-2 related mechanism in phytohaemagglutinin-stimulated human peripheral blood lymphocytes. Br J Nutr. 2006;95:870–8.
Chern CL, Huang RF, Chen YH, Cheng JT, Liu TZ. Folate deficiency-induced oxidative stress and apoptosis are mediated via homocysteine-dependent overproduction of hydrogen peroxide and enhanced activation of NF-kappaB in human Hep G2 cells. Biomed Pharmacother. 2001;55:434–42.
Berkels R, Purol-Schnabel S, Roesen R. A new method to measure nitrate/nitrite with a NO-sensitive electrode. J Appl Physiol. 2001;90:317–20.
Durak I, Yurtarslanl Z, Canbolat O, Akyol O. A methodological approach to superoxide dismutase (SOD) activity assay based on inhibition of nitroblue tetrazolium (NBT) reduction. Clin Chim Acta. 1993;214:103–4.
Draper HH, Hadley M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 1990;186:421–31.
Dandona P, Thusu K, Cook S, Snyder B, Makowski J, Armstrong D, Nicotera T. Oxidative damage to DNA in diabetes mellitus. Lancet 1996;347:444–5.
Tanaka Y, Gleason CE, Tran PO, Harmon JS, Robertson RP. Prevention of glucose toxicity in HIT-T15 cells and Zucker diabetic fatty rats by antioxidants. Proc Natl Acad Sci. 1999;96:10857–62.
Berry C, Hamilton CA, Brosnan MJ, Magill FG, Berg GA, McMurray JJ, Dominiczak AF. Investigation into the sources of superoxide in human blood vessels: angiotensin II increases superoxide production in human internal mammary arteries. Circulation 2000;101:2206–12.
Fiordaliso F, De Angelis N, Bai A, Cuccovillo I, Salio M, Maria Serra D, Bianchi R, Razzetti R, Latini R, Masson S. Effect of beta-adrenergic and renin–angiotensin system blockade on myocyte apoptosis and oxidative stress in diabetic hypertensive rats. Life Sci. 2007;81:951–9.
Jones WK, Flaherty MP, Tang XL, Takano H, Qiu Y, Banerjee S, Smith T, Bolli R. Ischemic preconditioning increases iNOS transcript levels in conscious rabbits via a nitric oxide-dependent mechanism. J Mol Cell Cardiol. 1999;31:1469–81.
Adeghate E. Molecular and cellular basis of the aetiology and management of diabetic cardiomyopathy: a short review. Mol Cell Biochem. 2004;261:187–91.
Del Poeta G, Venditti A, Del Principe MI, Maurillo L, Buccisano F, Tamburini A, Cox MC, Franchi A, Bruno A, Mazzone C, Panetta P, Suppo G, Masi M, Amadori S. Amount of spontaneous apoptosis detected by bax/bcl-2 ratio predicts outcome in acute myeloid leukemia (AML). Blood 2003;101:2125–31.
Chinnaiyan AM, O’Rourke K, Tewari M, Dixit VM. FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis. Cell 1995;81:505–12.
Huang RF, Yaong HC, Chen SC, Lu YF. In vitro folate supplementation alleviates oxidative stress, mitochondria-associated death signaling and apoptosis induced by 7-ketocholesterol. Br J Nutr. 2004;92:887–94.
Paolisso G, Tataranni PA, Foley JE, Bogardus C, Howard BV, Ravussin E. A high concentration of fasting plasma non-esterified fatty acids is a risk factor for the development of NIDDM. Diabetologia 1995;38:1213–7.
Cnop M, Hannaert JC, Hoorens A, Eizirik DL, Pipeleers DG. Inverse relationship between cytotoxicity of free fatty acids in pancreatic islet cells and cellular triglyceride accumulation. Diabetes 2001;50:1771–7.
Artwohl M, Roden M, Waldhausl W, Freudenthaler A, Baumgartner-Parzer SM. Free fatty acids trigger apoptosis and inhibit cell cycle progression in human vascular endothelial cells. FASEB J. 2004;18:146–8.
Bitar MS. Co-administration of etomoxir and RU-486 mitigates insulin resistance in hepatic and muscular tissues of STZ-induced diabetic rats. Horm Metab Res. 2001;33:577–84.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wu, TG., Li, WH., Lin, ZQ. et al. Effects of Folic Acid on Cardiac Myocyte Apoptosis in Rats with Streptozotocin-induced Diabetes Mellitus. Cardiovasc Drugs Ther 22, 299–304 (2008). https://doi.org/10.1007/s10557-008-6109-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-008-6109-8