Abstract
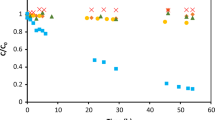
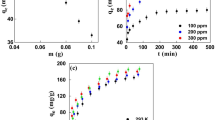
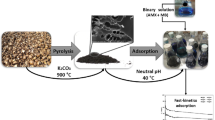
Activated carbon (AC) can be used for the removal of emerging contaminants (e.g., drugs) in water and wastewater treatment plants. In the present study, we investigated the performance of two ACs (from coconut shell and Pinnus sp.) in the adsorption of caffeine, carbamazepine, and ricobendazole considering the compounds separately and in combination in batch-scale experiments. The concentrations of the drugs were determined by a validated method using solid-phase extraction with on-line ultra-high performance liquid chromatography-tandem mass spectrometry. The most mesoporous AC provided higher drug removal. The kinetic data were described by the pseudo-second-order, Elovich, and Weber-Morris models, while the adsorption isotherms showed a better fit to the Freundlich model, indicative of multilayer adsorption. The Dubinin-Radushkevich model was used as a first approach to estimate the mean adsorption energy (E) and the results indicate that chemisorption governed the adsorption process, with E higher than 8 kJ mol−1. In the multicomponent assays, the adsorption of caffeine showed the greatest hindrance caused by the presence of other drugs. Multicomponent assays are fundamental to evaluate the potential adsorption capacity in real water treatment plants. Our study suggests that drugs with different structures and physicochemical properties may interact differently with ACs, especially in multicomponent solutions, with important implications for the design (e.g., volumes and areas of treatment plants) and operation (e.g., water residence time) of the treatment plants.




Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its supplementary information files. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ahmed MJ (2017) Adsorption of non-steroidal anti-inflammatory drugs from aqueous solution using activated carbons: review. J Environ Manag 191:274–282. https://doi.org/10.1016/j.jenvman.2016.12.073
Araújo LS, Coutinho AP, Alvarez-Mendez MO, Moruzzi RB, Calijuri MC, Cunha DGF (2018) Caracterização e avaliação de fatores que determinam a remoção de microcistina-LR em carvão ativado granular produzido a partir de diferentes matérias-primas. Engenharia Sanitária e Ambiental 23:1131–1142. https://doi.org/10.1016/10.1590/S1413-41522018177756
ASTM. D 3860. Standard Practice for Determination of Adsorptive Capacity of Activated Carbon by Aqueous Phase Isotherm Technique. 2014.
Baccar R, Sarrà M, Bouzid J, Feki M, Blánquez P (2012) Removal of pharmaceutical compounds by activated carbon prepared from agricultural by-product. Chem Eng J 211–212:310–317. https://doi.org/10.1016/j.cej.2012.09.099
Baçaoui A, Yaacoubi A, Dahbi A, Bennouna C, Luu RPT, Maldonado-Hodar FJ, Rivera-Utilla J, Moreno-Castilla C (2001) Optimization of conditions for the preparation of activated carbons from olive-waste cakes. Carbon. 39(3):425–432. https://doi.org/10.1016/S0008-6223(00)00135-4
Beltrame KK, Cazetta AL, Souza PSC, Spessato L, Silva TL, Almeida VC (2018) Adsorption of caffeine on mesoporous activated carbon fibers prepared from pineapple plant leaves. Ecotoxicol Environ Saf 147:64–71. https://doi.org/10.1016/j.ecoenv.2017.08.034
Blanchard, G., Maunaye, M., Maritin, G. 1984. Removal of heavy metals from waters by means of natural zeolites. Water Res 18(12), 1501-1507. https://doi.org/10.1016/0043-1354(84)90124-6
Brazilian Association of Technical Standards (ABNT) (1991a) EB 2133 (NBR 11.834). Pulverized activated carbon. Rio de Janeiro
Brazilian Association of Technical Standards (ABNT) (1991b) MB 3410 (NBR 12.073): carvão ativado pulverizado: determinação no número de iodo. Rio de Janeiro
Boehm HP (2002) Surface oxides on carbon and their analysis: a critical assessment. Carbon. 40(2):145–149
de Aquino SF, Brandt EMF, Chernicharo CAL (2013) Remoção de fármacos e desreguladores endócrinos em estações de tratamento de esgoto: Revisão da literatura. Engenharia Sanitária e Ambiental 18(3):187–204. https://doi.org/10.1590/S1413-41522013000300002
Cabrera-Lafaurie WA, Román FR, Hernández-Maldonado AJ (2015) Single and multi-component adsorption of salicylic acid, clofibric acid, carbamazepine and caffeine from water onto transition metal modified and partially calcined inorganic–organic pillared clay fixed beds. J Hazard Mater 282:174–182. https://doi.org/10.1016/j.jhazmat.2014.03.009
Cheng N, Wang B, Wu P, Lee X, Xing Y, Chen M, Gao B (2021) Adsorption of emerging contaminants from water and wastewater by modified biochar: a review. Environ Pollut 273:273. https://doi.org/10.1016/j.envpol.2021.116448
Chien SH, Clayton WR (1980) Application of elovich equation to the kinetics of phosphate release and sorption in soils. Soil Sci Soc Am J 44(2):265–268. https://doi.org/10.2136/sssaj1980.03615995004400020013x
Di Bernardo L, Dantas ADB, Voltan PEN (2017) Métodos e Técnicas de Tratamento de Águas, 3ª edn. LDiBe, São Carlos
De Witte B, Dewulf J, Demeestere K, Van Langenhove H (2009) Ozonation and advanced oxidation by the peroxone process of ciprofloxacin in water. J Hazard Mater:701–708. https://doi.org/10.1016/j.jhazmat.2008.04.021161
Dominguez JR, González T, Palo P, Cuerda-Correa EM (2011) Removal of common pharmaceuticals present in surface waters by Amberlite XAD-7 acrulic-ester-resin: Influence of pH and presence of other drugs. Desalination. 269:231–238. https://doi.org/10.1016/j.desal.2010.10.065
Donati C, Drikas M, Hayes R, Newcombe G (1994) Microcystin-LR adsorption by powdered activated carbon. Water Res 28(8):1735–1742. https://doi.org/10.1016/0043-1354(94)90245-3
Dubinin M., Radushkevich, L. 1947. Equation of the characteristic curve of activated charcoal. Chem Zentr, 1 (1) (1947), p. 875
François, L.L., Haro, N.K., Souza, F.S., Féris, L.A., 2016. Remoção de Cafeína por Adsorção em Carvão Ativado. Scientia cum Industria. 4(2), 64–8. 10.18226/23185279.v4iss2p64
Freundlich H (1906) Über die adsorption in Lösungen. Z Phys Chem 57U(1):385–470. https://doi.org/10.1515/zpch-1907-5723
Galhetas M, Mestre AS, Pinto ML, Gulyurtlu I, Lopes H, Carvalho AP (2014) Chars from gasification of coal and pine activated with K2CO3: acetaminophen and caffeine adsorption from aqueous solutions. J Colloid Interface Sci 443:94–103. https://doi.org/10.1016/j.jcis.2014.06.043
Giles, C.H., MacEwan, T.H., Nakhwa, S.N., Smith, D. 1960. 786. Studies in adsorption. Part XI. A system of classification of solution adsorption isothermis, and its use in diagnosis of adsorption mechanisms and in measurement of specific surface areas of solids. J Chem Soc. 3973-3993. https://doi.org/10.1039/JR9600003973
González-García P (2018) Activated carbon from lignocellulosics precursors: a review of the synthesis methods, characterization techniques and applications. Renew Sust Energ Rev 82(1):1393–1414. https://doi.org/10.1016/j.rser.2017.04.117
Hu Q, Zhang Z (2019) Application of Dubinin–Radushkevich isotherm model at the solid/solution interface: a theoretical analysis. J Mol Liq 277:646–648. https://doi.org/10.1016/j.molliq.2019.01.005
Jain JS, Snoeyink VL (1973) Adsorption from bisolute systems on active carbon. J Water Pollut Control Fed 45(12):2463–2479
JIS. K1474: Test methods for activated carbon. Tokyo; 2014.
Jaguaribe EF, Medeireos LL, Barreto MCS, Araújo LP (2005) The performance of activated carbon from sugarcane bagasse, babassu, and coconut shells in removing residual chlorine. Braz J Chem Eng 22(1):41–47. https://doi.org/10.1590/S0104-66322005000100005
Juang R, Cheng M (1997) Application of the Elovich equation to the kinetics of metal sorption with solvent-impregnated resins. Ind Eng Chem Res 36(3):813–820. https://doi.org/10.1021/ie960351f
Kuroda EK, Júnio ECA, Di Bernardo L, Trofino L (2005) Caracterização e escolha do tipo de carvão ativado a ser empregado no tratamento de águas contendo microcistinas. In: Congresso Brasileiro de Engenharia Sanitária e Ambiental. 23.
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40(9):1361–1403. https://doi.org/10.1021/ja02242a004
Li W, Peng J, Zhang L, Yang K, Xia H, Zhang S, Guo S (2009) Preparation of activated carbon from coconut shell chars in pilot-scale microwave heating equipment at 60 kW. Waste Manag 29(2):756–760. https://doi.org/10.1016/j.wasman.2008.03.004
Lima ÉC, Adebayo MA, Machado FM (2015) Kinetic and equilibrium models of adsorption. In: Bergmann C, Machado F (eds) Carbon nanomaterials as adsorbents for environmental and biological applications. Carbon Nanostructures. Springer, Cham. https://doi.org/10.1007/978-3-319-18875-1_3
Li Y, Gan Z, Liu Y, Chen S, Su S, Ding S, Tran NH, Chen X, Long Z (2020) Determination of 19 anthelmintics in environmental water and sediment using an optimized PLE and SPE method coupled with UHPLC-MS/MS. Sci Total Environ 719:137516. https://doi.org/10.1016/j.scitotenv.2020.137516
Liu H, Zhang J, Nho HH, Gio W, Wu H, Guo Z, Cheng C, Zhang C (2015) Effect on physical and chemical characteristics of activated carbon on adsorption of trimethoprim: mechanisms study. Royal Soc Chem Adv 5:85187–85195. https://doi.org/10.1039/c5ra17968h
Liu J, Zhou B, Zhang H, Ma J, Mu B, Zhang W (2019) A novel biochar modified by chitosan-Fe/S for tetracycline adsorption and studies on site energy distribution. Bioresour Technol 294:294. https://doi.org/10.1016/j.biortech.2019.122152
Mansouri H, Carmona RJ, Gomis-Berenguer A, Souissi-Najar S, Ouederni A, Ania CO (2015) Competitive adsorption of ibuprofen and amoxicillin combination from aqueous solution on activated carbons. J Colloid Interface Sci 449:252–260
Matsui Y, Yoshida T, Nakao S, Knappe DRU, Matsuhita T (2012) Characteristics of competitive adsorption between 2-methylisoborneol and natural organic matter on superfine and conventionally sized powdered activated carbons. Water Res 46(15):4741–4749. https://doi.org/10.1016/j.watres.2012.06.002
Mclintock IS (1967) The Elovich equation in chemisorption kinetics. Nature 216:1204–1205. https://doi.org/10.1038/2161204a0
Medina FMO, Aguiar MB, Parolo ME, Avena MJ (2021) Insights of competitive adsorption on activated carbon of binary caffeine and diclofenac solutions. J Environ Manag 272(2):111523. https://doi.org/10.1016/j.jenvman.2020.111523
Mestre AS, Pires J, Nogueira JMF, Carvalho AP (2007) Activated carbons for the adsorption of ibuprofen. Carbon. 45:1979–1988. https://doi.org/10.1016/j.carbon.2007.06.005
Miazek K, Brozek-Pluska B (2019) Effect of PHRs and PCPs on microalgal growth, metabolism and microalgae-based bioremediation processes: a review. Int J Mol Sci 20(10):2492. https://doi.org/10.3390/ijms20102492
Müller CC, Raya-Rodriguez MT, Cybis LF (2009) Adsorção em carvão ativado em pó para remoção de microcistina de água de abastecimento público. Engenharia Sanitária e Ambiental 14(1):29–38
Mourão PAM, Laginhas C, Custódio F, Nabais JMV, Carrott PJM, Carrott MML (2011) Influence of oxidation process on the adsorption capacity of activated carbons from lignocellulosic precursors. Fuel Process Technol 92(2):241–246. https://doi.org/10.1016/j.fuproc.2010.04.013
Nam SW, Choi DJ, Kim SK, Her N, Zoh KD (2014) Adsorption characteristics of selected hydrophilic and hydrophobic micropollutants in water using activated carbon. J Hazard Mater 270:144–152. https://doi.org/10.1016/j.jhazmat.2014.01.037
Nascimento FR, Lima ACA,Vidal BC, Melo QD, Raulino CSG, (2014) Adsorção: aspectos teóricos e aplicações ambientais. 2014. Avaliabe: http://www.repositorio.ufc.br/handle/riufc/10267
Nielsen L, Bandosz TJ (2016) Analysis of the competitive adsorption of pharmaceuticals on waste derived materials. Chem Eng J:287
Nwabue FI, Itumoh EJ (2020) Adsorption isotherm and kinetic modeling of a novel procedure for physical modification of silica gel using aqueous solutions of 4,4′-(1,2-ethanediyldinitrilo)bis-(2-pentanone) for preconcentration of Ni(II) ion. Sep Sci Technol 55(16):2919–2932. https://doi.org/10.1080/01496395.2019.1659821
Pendleton P, Shumann R, Wong SH (2001) Microcystin-LR adsorption by activated carbon. J Colloid Interface Sci 240(1):1–8. https://doi.org/10.1006/jcis.2001.7616
Pérez-Marín AB, Zapata VM, Ortuño JF, Aguilar M, Sáez J, Lloréns M (2007) Removal of cadmium from aqueous solutions by adsorption onto orange waste. J Hazard Mater 139(1):122–131. https://doi.org/10.1016/j.jhazmat.2006.06.008
Peteffi GP, Fleck JD, Kael IM, Girardi V, Bündchen R, Krajeski DM, Demoliner M, Silva FP, da Rosa DC, Antunes M, Linden V, R. (2018) Caffeine levels as a predictor of human mastadenovirus presence in surface waters—a case study in the Sinos River basin—Brazil. Environ Sci Pollut Res 25(16):15774–15784. https://doi.org/10.1007/s11356-018-1649-3
Paunovic O, Pap S, Maletic S, Taggart MA, Boskovic N, Sekulic MT (2019) Ionisable emerging pharmaceutical adsorption onto microwave functionalised biochar derived from novel lignocellulosic waste biomass. J Colloid Interface Sci 547:350–360. https://doi.org/10.1016/j.jcis.2019.04.011
Pomati F, Orlandi C, Clerici M, Luciani F, Zuccato E (2008) Effects and interactions in an environmentally relevant mixture of pharmaceuticals. Toxicol Sci 102(1):129–137. https://doi.org/10.1093/toxsci/kfm291
Porto RS, Rodrigues-Silva C, Schneider J, Rath S (2019) Benzimidazoles in wastewater: analytical method development, monitoring and degradation by photolysis and ozonation. J Environ Manag 232:729–737. https://doi.org/10.1016/j.jenvman.2018.11.121
Pivetta, RC., Rodrigues-Silva, C., Ribeiro, A.R., Rath, S. 2020. Tracking the occurrence of psychotropic pharmaceuticals in Brazilian wastewater treatment plants and surface water, with assessment of environmental risks. Sci Total Environ 727. https://doi.org/10.1016/j.scitotenv.2020.138661, 138661
Qiu H, Lv L, Pan BC, Zhang QJ, Zhang WM, Zhang QX (2009) Critical review in adsorption kinetic models. J Zhejiang Univ Sci A 10(5):716–724
Roginsky S, Zeldivich YB (1934) The catalytic oxidation of carbon monoxide on manganese dioxide. Acta Phys Chem USSR 1:554
Sotelo JL, Ovejero G, Rodríguez A, Álvarez S, Galán J, García J (2014) Competitive adsorption studies of caffeine and diclofenac aqueous solutions by activated carbon. Chem Eng J 240:443–453. https://doi.org/10.1016/j.cej.2013.11.094
Spina M, Venâncio W, Rodrigues-Silva C, Pivetta RC, Diniz V, Rath S, Guimarães JR (2021) Degradation of antidepressant pharmaceuticals by photoperoxidation in diverse water matrices: a highlight in the evaluation of acute and chronic toxicity. Environ Sci Pollut Res 28:24034–24045. https://doi.org/10.1007/s11356-020-11657-4
Sui Q, Cao X, Lu S, Zhao W, Qiu Z, Yu G (2015) Occurrence, sources and fate of pharmaceuticals and personal care products in the groundwater: A review. Emerging Contaminants 1:14–24. https://doi.org/10.1016/j.emcon.2015.07.001
Suriyanon N, Punyapalakul P, Ngamcharussrivichai C (2013) Mechanistic study of diclofenac and carbamazepine adsorption on functionalized silica-based porous materials. Chem Eng J 214:208–218. https://doi.org/10.1016/j.cej.2012.10.052
Tanada S, Kawasaki N, Nakamura T, Araki M, Isomura M (1999) Removal of formaldehyde by activated carbons containing amino group. J Colloid Interface Sci 214:106–108. https://doi.org/10.1006/jcis.1999.6176
Tetzner NF, Maniero MG, Rodrigues-Silva C, Rath S (2016) On-line solid phase extraction-ultra high performance liquid chromatography-tandem mass spectrometry as a powerful technique for the determination of sulfonamide residues in soils. J Chromatogr A 1452:89–97. https://doi.org/10.1016/j.chroma.2016.05.034
Tran HN, You S, Chao HP (2016) Thermodynamic parameters of cadmium adsorption onto orange peel calculated from various methods: A comparison study. J Environ Chem Eng 4(3):2671–2682. https://doi.org/10.1016/j.jece.2016.05.009
Tran HN, You S, Hosseini-Nadegharaei A, Chao H (2017) Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: a critical review. Water Res 120:88–116. https://doi.org/10.1016/j.watres.2017.04.014
Umemura Y, Shinohara E, Schoonheydt RA (2009) Preparation of Langmuir–Blodgett films of aligned sepiolite fibers and orientation of methylene blue molecules adsorbed on the film. Phys Chem Chem Phys 11:9804–9810. https://doi.org/10.1039/b817635c
Valderrama C, Gamisans X, de las Heras X, Farrán A, Cortina JL (2008) Sorption kinetics of polycyclic aromatic hydrocarbons removal using granular activated carbon: Intraparticle diffusion coefficients. J Hazard Mater 157(2-3):386–396. https://doi.org/10.1016/j.jhazmat.2007.12.119
Varga M, ELAbadsa M, Tatár E, Mihucz VG (2019) Removal of selected pharmaceuticals from aqueous matrices with activated carbon under batch conditions. Microchem J 148:661–672. https://doi.org/10.1016/j.microc.2019.05.038
Venancio WAL, Rodrigues-Silva C, Spina M, Diniz V, Guimarães JR (2020) Degradation of benzimidazoles by photoperoxidation: metabolites detection and ecotoxicity assessment using Raphidocelis subcapitata microalgae and Vibrio fischeri. Environ Sci Pollut Res 28:23742–23752. https://doi.org/10.1007/s11356-020-11294-x
Vimonses V, Lei S, Jin B, Chow CWK, Saint C (2009) Kinetic study and equilibrium isotherm analysis of Congo Red adsorption by clay materials. Chem Eng J 148(2-3):354–364. https://doi.org/10.1016/j.cej.2008.09.009
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div 89(2):31–60
Weber WJ, Smith EH (1987) Simulation and design models for adsorption processes. Environ Sci Technol 21(11):1040–1050. https://doi.org/10.1021/es00164a002
Wu F, Tseng R, Juang R (2009) Characteristics of Elovich equation used for the analysis of adsorption kinetics in dye-chitosan systems. Chem Eng J 150(2-3):366–373. https://doi.org/10.1016/j.cej.2009.01.014
Yu Z, Peldszus S, Huck PM (2008) Adsorption characteristics of selected pharmaceuticals and an endocrine disrupting compound-Naproxen, carbamazepine and nonylphenol-on activated carbon. Water Res 42(12):2873–2882. https://doi.org/10.1016/j.watres.2008.02.020
Acknowledgements
The authors are grateful for the financial support provided by the Brazilian agencies Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, grant #2018/03571-2), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, grants #301737/2017-7, 406855/2016-1, and 310844/2020-7), and Instituto Nacional de Ciências e Tecnologias Analíticas Avançadas (INCTAA: FAPESP, grant #2014/50951-4; CNPq, grant #465768/2014-8). Scholarships were awarded to V.D. (CNPq, grant #131091/2018-2) and L.S.A (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior). The authors also thank CAQI/IQSC/USP for the use of the SEM facilities.
Funding
The financial support was provided by FAPESP, CAPES, CNPq, and INCTAA. The detailed funding grant number is highlighted in the Acknowledgements section.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Vinicius Diniz: method, validation, formal analysis, and investigation, visualization, writing—original draft, and writing—review and editing. Gabriela Rath: analytical method validation, investigation, visualization, and writing—review and editing. Susanne Rath: analytical method validation, funding acquisition, visualization, writing—original draft, and writing—review and editing. Larissa Sene Araújo: method, validation, formal analysis, and investigation. Davi Gasparini Fernandes Cunha: supervision, project administration, funding acquisition, writing—original draft, writing—review and editing and visualization. The first draft of the manuscript was written by Vinicius Diniz, Davi Gasparini Fernandes Cunha, and Susanne Rath, and all authors commented on the versions of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 927 kb)
Rights and permissions
About this article
Cite this article
Diniz, ., Rath, G., Rath, S. et al. Competitive kinetics of adsorption onto activated carbon for emerging contaminants with contrasting physicochemical properties. Environ Sci Pollut Res 29, 42185–42200 (2022). https://doi.org/10.1007/s11356-021-16043-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-16043-2