Abstract
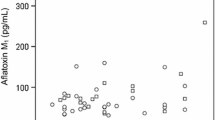
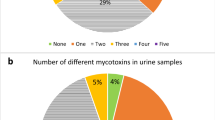
Recently, aflatoxin M1 (AFM1) has emerged as a major health concern owing to its exposure to human being via consumption of milk, dairy products, and food commodities, and this has a strong association with risk factors that may lead to the onset of type 2 diabetes mellitus (T2DM) and various other associated metabolic disorders. This study was conducted to investigate the exposure to AFM1 and its association with sociodemographic features and risk factors of T2DM. Urine and blood samples from 672 participants were collected to investigate the concentration of AFM1 in urine and glucose, glycosylated hemoglobin (HbA1c), insulin, α-amylase, dipeptidyl peptidase-IV (DPP-IV), free fatty acids (FFAs), triglycerides (TGs), high-density lipoprotein cholesterol (HDL-chol), interleukine-6 (IL-6), tumor necrosis factor-α (TNF-α), malondialdehyde (MDA), glutathione (GSH), superoxide dismutase (SOD), creatinine, uric acid, blood urea nitrogen (BUN), aspartate aminotransferase (AST), and alanine transaminase (ALT) from the blood of study participants. Association of exposure to AFM1 with sociodemographic features and risk factors of T2DM was determined using person correlation coefficient (r), coefficient of determination (R2), and 95% confidence interval, and the level of significance (P<0.05) was measured by Student’s unpaired t-test. Among the participants in which AFM1 was detected, 62.91% of participants were found to be diabetic and 37.09% of participants were found to be non-diabetic. Further to this, it was also found that concentration of AFM1 in the urine of diabetic participants was found to be higher (P<0.05) as compared to that in non-diabetic participants. Association of AFM1 exposure with risk factors of T2MD exhibits that exposure to AFM1 was responsible for the induction of inflammatory responses and oxidative stress that may lead to the onset of impaired insulin secretion and metabolism of carbohydrates and ultimately the onset of T2DM and associated metabolic disorders. Hence, it can be summarized that exposure to AFM1 is one of the causative factors that may lead to potentiate the several risk factors notably inflammatory responses and oxidative stress that ultimately induce the pathogenesis of T2DM and associated metabolic disorders. The key findings of this study suggest that human population who are at greater risk of AFM1 exposure can develop T2DM and other associated metabolic risk factors.


Similar content being viewed by others
References
Abdelhiee EY, Elbialy ZI, Saad AH, Dawood MA, Aboubakr M, El-Nagar SH et al (2021) The impact of Moringa oleifera on the health status of Nile tilapia exposed to aflatoxicosis. Aquac Res 533:736110. https://doi.org/10.1016/j.aquaculture.2020.736110
Abubacker N, Abubacker S, Rajendran K, Jayaraman S, Suthakaran P, Kalappan M (2016) A comparative study of spot urine versus 24 hour urine in assessment of proteinuria in varying degree of renal dysfunction. Int J Adv Med 3(1):1–4
Afolabi OK, Wusu AD, Ogunrinola OO, Abam EO, Babayemi DO, Dosumu OA, Onunkwor OB, Balogun EA, Odukoya OO, Ademuyiwa O (2015) Arsenic-induced dyslipidemia in male albino rats: comparison between trivalent and pentavalent inorganic arsenic in drinking water. BMC Pharmacol Toxicol 16(1):1–15. https://doi.org/10.1186/s40360-015-0015-z
Akash MSH, Rehman K, Chen S (2014) Effects of coffee on type 2 diabetes mellitus. Nutr. 30(7-8):755–763. https://doi.org/10.1016/j.nut.2013.11.020
Akash, M. S. H., Rehman, K., Hashmi, M. Z. (2020). Endocrine disrupting chemicals-induced metabolic disorders and treatment strategies: Springer.
Alpsoy L, Yildirim A, Agar G (2009) The antioxidant effects of vitamin A, C, and E on aflatoxin B1-induced oxidative stress in human lymphocytes. Toxicol Ind Health 25(2):121–127. https://doi.org/10.1177/0748233709103413
Carvajal-Moreno M (2017) Do gastroenterologists consider aflatoxins as origin of digestive system cancers. Aust J Pharm 5. https://doi.org/10.4172/2329-6887.1000242
Cheng J, Huang S, Fan C, Zheng N, Zhang Y, Li S, Wang J (2017) Metabolomic analysis of alterations in lipid oxidation, carbohydrate and amino acid metabolism in dairy goats caused by exposure to aflotoxin B1. J dairy res 84(4):401–406. https://doi.org/10.1017/s0022029917000590
Diawara MM, Litt JS, Unis D, Alfonso N, Martinez L, Crock JG, Smith DB, Carsella J (2006) Arsenic, cadmium, lead, and mercury in surface soils, Pueblo, Colorado: implications for population health risk. Environ Geochem Health 28(4):297–315. https://doi.org/10.1007/s10653-005-9000-6
Dohnal V, Wu Q, Kuča K (2014) Metabolism of aflatoxins: key enzymes and interindividual as well as interspecies differences. Arch Toxicol 88(9):1635–1644. https://doi.org/10.1007/s00204-014-1312-9
EL-AGAMY DS (2010) Comparative effects of curcumin and resveratrol on aflatoxin B1-induced liver injury in rats. Arch Toxicol 84(5):389–396. https://doi.org/10.1007/s00204-010-0511-2
Fenichel P, Chevalier N (2017) Environmental endocrine disruptors: new diabetogens? Comptes Rendus Biologies 340(9):446–452. https://doi.org/10.1016/j.crvi.2017.07.003
Goncalves B, Goncalves J, Rosim R, Cappato L, Cruz A, Oliveira C, Corassin C (2017) Effects of different sources of Saccharomyces cerevisiae biomass on milk production, composition, and aflatoxin M1 excretion in milk from dairy cows fed aflatoxin B1. J Dairy Sci 100(7):5701–5708. https://doi.org/10.3168/jds.2016-12215
Haq MEU, Akash MSH, Sabir S, Mahmood MH, Rehman K (2020) Human exposure to bisphenol A through dietary sources and development of diabetes mellitus: a cross-sectional study in Pakistani population. Environ Sci Pollut Res 27(21):26262–26275. https://doi.org/10.1007/s11356-020-09044-0
Howard SG (2018) Developmental exposure to endocrine disrupting chemicals and type 1 diabetes mellitus. Front Endocrinol 9:513–513. https://doi.org/10.3389/fendo.2018.00513
Kaleibar MT, Helan JA (2013) A field outbreak of aflatoxicosis with high fatality rate in feedlot calves in Iran. Comp Clin Pathol 22(6):1155–1163. https://doi.org/10.1007/s00580-012-1543-1
Kamdem LK, Meineke I, Gödtel-Armbrust U, Brockmöller J, Wojnowski L (2006) Dominant contribution of P450 3A4 to the hepatic carcinogenic activation of aflatoxin B1. Chem Res Toxicol 19(4):577–586. https://doi.org/10.1021/tx050358e
Kensler TW, Roebuck BD, Wogan GN, Groopman JD (2010) Aflatoxin: a 50-year odyssey of mechanistic and translational toxicology. Toxicol Sci 120(suppl_1):S28–S48. https://doi.org/10.1093/toxsci/kfq283
Kim K (2012) Blood cadmium concentration and lipid profile in Korean adults. Environ Res 112:225–229
Ledda C, Loreto C, Zammit C, Marconi A, Fago L, Matera S et al (2017) Non-infective occupational risk factors for hepatocellular carcinoma: a review. Mol Med Rep 15(2):511–533. https://doi.org/10.3892/mmr.2016.6046
Lei L-J, Chen L, Jin T-Y, Nordberg M, Chang X-L (2007) Estimation of benchmark dose for pancreatic damage in cadmium-exposed smelters. Toxicol Sci 97(1):189–195. https://doi.org/10.1093/toxsci/kfm016
Leong YH, Latiff AA, Ahmad NI, Rosma A (2012) Exposure measurement of aflatoxins and aflatoxin metabolites in human body fluids. A short review. Mycotoxin Res 28(2):79–87. https://doi.org/10.1007/s12550-012-0129-8
Liu W, Wang L, Zheng C, Liu L, Wang J, Li D, Tan Y, Zhao X, He L, Shu W (2018) Microcystin-LR increases genotoxicity induced by aflatoxin B1 through oxidative stress and DNA base excision repair genes in human hepatic cell lines. Environ Pollut 233:455–463. https://doi.org/10.1016/j.envpol.2017.10.067
Ma Y, Kong Q, Hua H, Luo T, Jiang Y (2012) Aflatoxin B1 up-regulates insulin receptor substrate 2 and stimulates hepatoma cell migration. PLoS One 7(10):e47961–e47961. https://doi.org/10.1371/journal.pone.0047961
Mahato DK, Lee KE, Kamle M, Devi S, Dewangan K, Kumar P, Kang SG (2019) Aflatoxins in food and feed: an overview on prevalence, detection and control strategies. Front Microbiol 10:2266. https://doi.org/10.3389/fmicb.2019.02266
Malekinezhad P, Ellestad LE, Afzali N, Farhangfar SH, Omidi A, Mohammadi A (2021) Evaluation of berberine efficacy in reducing the effects of aflatoxin B1 and ochratoxin A added to male broiler rations. Poult Sci J 100(2):797–809. https://doi.org/10.1016/j.psj.2020.10.040
Marchese S, Sorice A, Ariano A, Florio S, Budillon A, Costantini S, Severino L (2018) Evaluation of aflatoxin M1 effects on the metabolomic and cytokinomic profiling of a hepatoblastoma cell line. Toxins (Basel) 10(11):436. https://doi.org/10.3390/toxins10110436
Mohd Redzwan S, Rosita J, Mohd Sokhini AM, Nurul Aqilah AR (2012) Association between aflatoxin M1 excreted in human urine samples with the consumption of milk and dairy products. Bull Environ Contam Toxicol 89(6):1115–1119. https://doi.org/10.1007/s00266-013-0186-1
Organization, W. H (2011) WHO guidelines on good manufacturing practices for blood establishments. WHO Technical Report Series (961):148–214
Predieri B, Bruzzi P, Bigi E, Ciancia S, Madeo SF, Lucaccioni L, Iughetti L (2020) Endocrine disrupting chemicals and type 1 diabetes. Int J Mol Sci 21(8):2937. https://doi.org/10.3390/ijms21082937
Rehman K, Akash MSH, Liaqat A, Kamal S, Qadir MI, Rasul A (2017) Role of interleukin-6 in development of insulin resistance and type 2 diabetes mellitus. Crit Rev Eukaryot Gene Expr 27(3):229–236. https://doi.org/10.1615/critreveukaryotgeneexpr.2017019712
Rotimi OA, Rotimi SO, Duru CU, Ebebeinwe OJ, Abiodun AO, Oyeniyi BO, Faduyile FA (2017) Acute aflatoxin B1–Induced hepatotoxicity alters gene expression and disrupts lipid and lipoprotein metabolism in rats. Toxicol Rep 4:408–414. https://doi.org/10.1016/j.toxrep.2017.07.006
Rotimi OA, Rotimi SO, Oluwafemi F, Ademuyiwa O, Balogun EA (2018) Oxidative stress in extrahepatic tissues of rats co-exposed to aflatoxin B1 and low protein diet. Toxicol Res 34(3):211–220. https://doi.org/10.5487/TR.2018.34.3.211
Rushing BR, Selim MI (2019) Aflatoxin B1: a review on metabolism, toxicity, occurrence in food, occupational exposure, and detoxification methods. Food Chem Toxicol 124:81–100. https://doi.org/10.1016/j.fct.2018.11.047
Sabir S, Akash MSH, Fiayyaz F, Saleem U, Mehmood MH, Rehman K (2019) Role of cadmium and arsenic as endocrine disruptors in the metabolism of carbohydrates: inserting the association into perspectives. Biomed Pharmacother 114:108802. https://doi.org/10.1016/j.biopha.2019.108802
Sabir S, Rehman K, Fiayyaz F, Kamal S, Akash MSH (2020) Role of aflatoxins as EDCs in metabolic disorders. In: Endocrine Disrupting Chemicals-induced Metabolic Disorders and Treatment Strategies. Springer, pp 381–389
Sabran MR, Jamaluddin R, Abdul Mutalib MS (2012) Screening of aflatoxin M1, a metabolite of aflatoxin B1 in human urine samples in Malaysia: a preliminary study. Food Control 28(1):55–58. https://doi.org/10.1016/j.foodcont.2012.04.048
Saruc M, Karaarslan M, Rasa K, Saygili O, Ince U, Baysal C et al (2009) Pancreatic cancer and glucose metabolism. Turk J Gastroenterol 20(4):257–260. https://doi.org/10.4318/tjg.2009.0022
Tsai F-J, Chen S-Y, Liu Y-C, Liao H-Y, Chen C-J (2017) The comparison of CHCA solvent compositions for improving LC-MALDI performance and its application to study the impact of aflatoxin B1 on the liver proteome of diabetes mellitus type 1 mice. PLoS One 12(7):e0181423. https://doi.org/10.1371/journal.pone.0181423
Ubagai T, Kikuchi T, Fukusato T, Ono Y (2010) Aflatoxin B1 modulates the insulin-like growth factor-2 dependent signaling axis. Toxicol in Vitro 24(3):783–789. https://doi.org/10.1016/j.tiv.2009.12.022
Wild CP, Montesano R (2009) A model of interaction: aflatoxins and hepatitis viruses in liver cancer aetiology and prevention. Cancer Lett 286(1):22–28. https://doi.org/10.1016/j.canlet.2009.02.053
Wu Q, Jezkova A, Yuan Z, Pavlikova L, Dohnal V, Kuca K (2009) Biological degradation of aflatoxins. Drug Metab Rev 41(1):1–7. https://doi.org/10.1080/03602530802563850
Yang X, Shi D, Zhu S, Wang B, Zhang X, Wang G (2018) Portable aptasensor of aflatoxin B1 in bread based on a personal glucose meter and DNA walking machine. ACS Sens 3(7):1368–1375. https://doi.org/10.1021/acssensors.8b00304
Yiannikouris A, Jouany J-P (2002) Mycotoxins in feeds and their fate in animals: a review. Anim Res 51(2):81–99. https://doi.org/10.1051/animres:2002012
Zhang Y, Wang P, Kong Q, Cotty PJ (2021) Biotransformation of aflatoxin B1 by Lactobacillus Helviticus FAM22155 in wheat bran by solid-state fermentation. Food Chem 341:128180. https://doi.org/10.1016/j.foodchem.2020.128180
Zuberi Z, Eeza MN, Matysik J, Berry JP, Alia A (2019) NMR-based metabolic profiles of intact Zebrafish embryos exposed to aflatoxin B1 recapitulates hepatotoxicity and supports possible neurotoxicity. Toxins. 11(5):258. https://doi.org/10.3390/toxins11050258
Availability of data and materials
All data generated and/or analyzed during this study are included in this published article.
Funding
This work has been financially supported by the research grants (8365/Punjab/NRPU/R & D/HEC/2017) received from the Higher Education Commission of Pakistan.
Author information
Authors and Affiliations
Contributions
MSHA: project administration, supervision, conceptualization, methodology, writing—original draft preparation, and editing. MEQ: data curation, experimental analysis, investigation. AQ: writing—original draft, validation. KR: project administration, investigation, conceptualization, writing—final draft, and editing
Corresponding authors
Ethics declarations
Ethical approval
This study was ethically approved by the Institutional Review Board (IRB-GCUF-019928) of Government College University Faisalabad (GCUF).
Consent to participate
Informed written consent was obtained from all the participants before the collection of samples and sociodemographic data, and for those who could not write, verbal consents were recorded and signed by the interviewer on their behalf.
Consent to publish
N/A
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akash, M.S.H., Haq, M.E.u., Qader, A. et al. Biochemical investigation of human exposure to aflatoxin M1 and its association with risk factors of diabetes mellitus. Environ Sci Pollut Res 28, 62907–62918 (2021). https://doi.org/10.1007/s11356-021-14871-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-14871-w