Abstract
Many reports have shown a strong association between exposure to neurotoxic air pollutants like heavy metal and particulate matter (PM) as an active participant and neurological disorders. While the effects of these toxic pollutants on cardiopulmonary morbidity have principally been studied, growing evidence has shown that exposure to polluted air is associated with memory impairment, communication deficits, and anxiety/depression among all ages. So, these toxic pollutants in the environment increase the risk of neurodegenerative disease, ischemia, and autism spectrum disorders (ASD). The precise mechanisms in which air pollutants lead to communicative inability, social inability, and declined cognition have remained unknown. Various animal model studies show that amyloid precursor protein (APP), processing, oxidant/antioxidant balance, and inflammation pathways change following the exposure to constituents of polluted air. In the present review study, we collect the probable molecular mechanisms of deleterious CNS effects in response to various air pollutants.
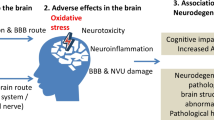
Graphical abstract






Similar content being viewed by others
Data availability
Not applicable.
References
Adams K, Greenbaum DS, Shaikh R, Van Erp AM, Russell AG (2015) Particulate matter components, sources, and health: systematic approaches to testing effects. J Air Waste Manage Assoc 65:544–558. https://doi.org/10.1080/10962247.2014.1001884
Agnihotri A, Aruoma OI (2020) Alzheimer’s disease and Parkinson’s disease: a nutritional toxicology perspective of the impact of oxidative stress, mitochondrial dysfunction, nutrigenomics and environmental chemicals. J Am Coll Nutr 39:16–27. https://doi.org/10.1080/07315724.2019.1683379
Aird WC (2007) Phenotypic heterogeneity of the endothelium: I. Structure, function, and mechanisms. Circ Res 100:158–173
Akhter H, Ballinger C, Liu N, van Groen T, Postlethwait EM, Liu RM (2015) Cyclic ozone exposure induces gender-dependent neuropathology and memory decline in an animal model of Alzheimer’s disease. Toxicol Sci 147(1):222–234. https://doi.org/10.1093/toxsci/kfv124
Allen JL, Liu X, Pelkowski S, Palmer B, Conrad K, Oberdorster G, Weston D, Mayer-Proschel M, Cory-Slechta DA (2014a) Early postnatal exposure to ultrafine particulate matter air pollution: persistent ventriculomegaly, neurochemical disruption, and glial activation preferentially in male mice. Environ Health Perspect 122:939–945. https://doi.org/10.1289/ehp.1307984
Allen JL, Liu X, Weston D, Conrad K, Oberdörster G, Cory-Slechta DA (2014b) Consequences of developmental exposure to concentrated ambient ultrafine particle air pollution combined with the adult paraquat and maneb model of the Parkinson’s disease phenotype in male mice. Neurotoxicol. 41:80–88. https://doi.org/10.1016/j.neuro.2014.01.004
Allen JL, Oberdorster G, Morris-Schaffer K, Wong C, Klocke C, Sobolewski M, Conrad K, Mayer-Proschel M, Cory-Slechta DA (2017) Developmental neurotoxicity of inhaled ambient ultrafine particle air pollution: parallels with neuropathological and behavioral features of autism and other neurodevelopmental disorders. Neurotoxicol 59:140–154. https://doi.org/10.1016/j.neuro.2015.12.014
Anderson FL, Coffey MM, Berwin BL, Havrda MC (2018) Inflammasomes: an emerging mechanism translating environmental toxicant exposure into neuroinflammation in Parkinson’s disease. Toxicol Sci 166:3–15. https://doi.org/10.1093/toxsci/kfy219
Angoa-Perez M, Jiang H, Rodriguez AI, Lemini C, Levine RA, Rivas-Arancibia S (2006) Estrogen counteracts ozone-induced oxidative stress and nigral neuronal death. Neuroreport 17(6):629–633. https://doi.org/10.1097/00001756-200604240-00014
Bagheri G, Rezaee R, Tsarouhas K, Docea AO, Shahraki J, Shahriari M, Wilks MF, Jahantigh H, Tabrizian K, Moghadam AA, Bagheri S (2019) Magnesium sulfate ameliorates carbon monoxide induced cerebral injury in male rats. Mol Med Rep 19:1032–1039. https://doi.org/10.3892/mmr.2018.9771
Barthelemy NR, Li Y, Joseph-Mathurin N, Gordon BA, Hassenstab J, Benzinger TL, Buckles V, Fagan AM, Perrin RJ, Goate AM, Morris JC (2020) A soluble phosphorylated tau signature links tau, amyloid and the evolution of stages of dominantly inherited Alzheimer’s disease. Nat Med 26(3):398–407. https://doi.org/10.1038/s41591-020-0781-z
Bauman MD, Iosif AM, Smith SEP, Bregere C, Amaral DG, Patterson PH (2014) Activation of the maternal immune system during pregnancy alters behavioral development of Rhesus monkey offspring. Biol Psychiatry 75:332–341. https://doi.org/10.1016/j.biopsych.2013.06.025
Beggiato S, Tomasini MC, Cassano T, Ferraro L (2020) Chronic oral palmitoylethanolamide administration rescues cognitive deficit and reduces neuroinflammation, oxidative stress, and glutamate levels in a transgenic murine model of Alzheimer’s disease. J Clin Med 9(2):428. https://doi.org/10.3390/jcm9020428
Beitz JM (2014) Parkinson s disease: a review. Front Biosci (Schol Ed) 6:65–74. https://doi.org/10.2741/s415
Ben-Shachar D (2020) The bimodal mechanism of interaction between dopamine and mitochondria as reflected in Parkinson’s disease and in schizophrenia. J Neural Transm 127:159–168. https://doi.org/10.1007/s00702-019-02120-x
Bermudez E, Mangum JB, Wong BA, Asgharian B, Hext PM, Warheit DB, Everitt JI (2004) Pulmonary responses of mice, rats, and hamsters to subchronic inhalation of ultrafine titanium dioxide particles. Toxicol Sci 77:347–357. https://doi.org/10.1093/toxsci/kfh019
Bernal-Melendez E, Lacroix MC, Bouillaud P, Callebert J, Olivier B, Persuy MA, Durieux D, Rousseau-Ralliard D, Aioun J, Cassee F, Couturier-Tarrade A (2019) Repeated gestational exposure to diesel engine exhaust affects the fetal olfactory system and alters olfactory-based behavior in rabbit offspring. Part Fibre Toxicol 16:5. https://doi.org/10.1186/s12989-018-0288-7
Bhatt DP, Puig KL, Gorr MW, Wold LE, Combs CK (2015) A pilot study to assess effects of long-term inhalation of airborne particulate matter on early Alzheimer-like changes in the mouse brain. PLoS One 10:e0127102. https://doi.org/10.1371/journal.pone.0127102
Block ML, Wu X, Pei Z, Li G, Wang T, Qin L, Wilson B, Yang J, Hong JS, Veronesi B (2004) Nanometer size diesel exhaust particles are selectively toxic to dopaminergic neurons: the role of microglia, phagocytosis, and NADPH oxidase. FASEB J 18:1618–1620
Bolton JL, Auten RL, Bilbo SD (2014) Prenatal air pollution exposure induces sexually dimorphic fetal programming of metabolic and neuroinflammatory outcomes in adult offspring. Brain Behav Immun 37:30–44. https://doi.org/10.1016/j.bbi.2013.10.029
Bolton JL, Marinero S, Hassanzadeh T, Natesan D, Le D, Belliveau C, Mason SN, Auten RL, Bilbo SD (2017) Gestational exposure to air pollution alters cortical volume, microglial morphology, and microglia-neuron interactions in a sex-specific manner. Front Synaptic Neurosci 9:10. https://doi.org/10.3389/fnsyn.2017.00010
Brauer M, Avila-Casado C, Fortoul TI, Vedal S, Stevens B, Churg A (2001) Air pollution and retained particles in the lung. Environ Health Perspect 109:1039–1043. https://doi.org/10.1289/ehp.011091039
Braun JM, Kalkbrenner AE, Just AC, Yolton K, Calafat AM, Sjödin A, Hauser R, Webster GM, Chen A, Lanphear BP (2014) Gestational exposure to endocrine-disrupting chemicals and reciprocal social, repetitive, and stereotypic behaviors in 4-and 5-year-old children: the HOME study. Environ Health Perspect 122(5):513–520. https://doi.org/10.1289/ehp.1307261
Brook RD, Rajagopalan S, Pope CA III, Brook JR, Bhatnagar A, Diez-Roux AV, Holguin F, Hong Y, Luepker RV, Mittleman MA, Peters A (2010) Particulate matter air pollution and cardiovascular disease: an update to the scientific statement from the American Heart Association. Circ 121:2331–2378. https://doi.org/10.1161/CIR.0b013e3181dbece
Cacciottolo M, Morgan TE, Saffari AA, Shirmohammadi F, Forman HJ, Sioutas C, Finch CE (2020) Traffic-related air pollutants (TRAP-PM) promote neuronal Amyloidogenesis through oxidative damage to lipid rafts. Free Radic Biol Med 147:242–251. https://doi.org/10.1016/j.freeradbiomed.2019.12.023
Cacciottolo M, Wang X, Driscoll I, Woodward N, Saffari A, Reyes J, Serre ML, Vizuete W, Sioutas C, Morgan TE, Gatz M (2017) Particulate air pollutants, APOE alleles and their contributions to cognitive impairment in older women and to amyloidogenesis in experimental models. Transl Psychiatry 7:e1022. https://doi.org/10.1016/j.freeradbiomed.2019.12.023
Calderon-Garciduenas L, Azzarelli B, Acuna H, Garcia R, Gambling TM, Osnaya N, Monroy S, Del Rosario Tizapantzi M, Carson JL, Villarreal-Calderon A, Rewcastle B (2012) Air pollution and brain damage. Toxicol Pathol 30:373–389. https://doi.org/10.1080/01926230252929954
Calderon-Garciduenas L, Mora-Tiscareno A, Gomez-Garza G, Carrasco-Portugal MD, Perez-Guille B, Flores-Murrieta FJ, Perez-Guille G, Osnaya N, Juarez-Olguin H, Monroy ME, Monroy S (2009) Effects of a cyclooxygenase-2 preferential inhibitor in young healthy dogs exposed to air pollution: a pilot study. Toxicol Pathol 37:644–660. https://doi.org/10.1177/0192623309340277
Calderon-Garciduenas LI, Reed W, Maronpot RR, Henriquez-Roldan C, Delgado-Chavez R, Calderon-Garciduenas AN, Dragustinovis I, Franco-Lira M, Aragón-Flores M, Solt AC, Altenburg M (2004) Brain inflammation and Alzheimer’s-like pathology in individuals exposed to severe air pollution. Toxicol Pathol 32:650–658. https://doi.org/10.1080/01926230490520232
Calderon-Garciduenas L, Solt AC, Henriquez-Roldan C, Torres-Jardon R, Nuse B, Herritt L, Villarreal-Calderon R, Osnaya N, Stone I, Garcia R, Brooks DM (2008) Long-term air pollution exposure is associated with neuroinflammation, an altered innate immune response, disruption of the blood-brain barrier, ultrafine particulate deposition, and accumulation of amyloid beta-42 and alpha-synuclein in children and young adults. Toxicol Pathol 36:289–310. https://doi.org/10.1177/0192623307313011
Calderon-Garciduenas L, Vojdani A, Blaurock-Busch E, Busch Y, Friedle A, Franco-Lira M, Sarathi-Mukherjee P, Martinez-Aguirre X, Park SB, Torres-Jardon R, D’Angiulli A (2015) Air pollution and children: neural and tight junction antibodies and combustion metals, the role of barrier breakdown and brain immunity in neurodegeneration. J Alzheimers Dis 43(3):1039–1058. https://doi.org/10.3233/JAD-141365
Campbell A (2004) Inflammation, neurodegenerative diseases, and environmental exposures. Ann N Y Acad Sci 1035:117–132. https://doi.org/10.1196/annals.1332.008
Campbell A, Oldham M, Becaria A, Bondy SC, Meacher D, Sioutas C, Misra C, Mendez LB, Kleinman M (2005) Particulate matter in polluted air may increase biomarkers of inflammation in mouse brain. Neurotoxicol 26:133–140. https://doi.org/10.1016/j.neuro.2004.08.003
Carageorgiou H, Tzotzes V, Sideris A, Zarros A, Tsakiris S (2005) Cadmium effects on brain acetylcholinesterase activity and antioxidant status of adult rats: modulation by zinc, calcium and L-cysteine co-administration. Basic Clin Pharmacol Toxicol 97:320–324. https://doi.org/10.1111/j.1742-7843.2005.pto_174.x
Chang YC, Cole TB, Costa LG (2018) Prenatal and early-life diesel exhaust exposure causes autism-like behavioral changes in mice. Part Fibre Toxicol 15(1):18. https://doi.org/10.1186/s12989-018-0254-4
Chang YC, Daza R, Hevner R, Costa LG, Cole TB (2019) Prenatal and early life diesel exhaust exposure disrupts cortical lamina organization: evidence for a reelin-related pathogenic pathway induced by interleukin-6. Brain Behav Immun 78:105–115. https://doi.org/10.1186/s12989-018-0254-4
Cheng H, Saffari A, Sioutas C, Forman HJ, Morgan TE, Finch CE (2016) Nanoscale particulate matter from urban traffic rapidly induces oxidative stress and inflammation in olfactory epithelium with concomitant effects on brain. Environ Health Perspect 124:1537–1546. https://doi.org/10.1289/EHP134
Cheng Y, Thomas A, Mardini F, Bianchi SL, Tang JX, Peng J, Wei H, Eckenhoff MF, Eckenhoff RG, Levy RJ (2012) Neurodevelopmental consequences of sub-clinical carbon monoxide exposure in newborn mice. PLoS One 7:e32029. https://doi.org/10.1371/journal.pone.0032029
Chen P, Shibata M, Zidovetzki R, Fisher M, Zlokovic BV, Hofman FM (2001) Endothelin-1 and monocyte chemoattractant protein-1 modulation in ischemia and human brain-derived endothelial cell cultures. J Neuroimmunol 116:62–73. https://doi.org/10.1016/S0165-5728(01)00280-6
Church JS, Tijerina PB, Emerson FJ, Coburn MA, Blum JL, Zelikoff JT, Schwartzer JJ (2018) Perinatal exposure to concentrated ambient particulates results in autism-like behavioral deficits in adult mice. Neurotoxicol 65:231–240. https://doi.org/10.1016/j.neuro.2017.10.007
Churg A, Brauer M (2000) Ambient atmospheric particles in the airways of human lungs. Ultrastruct Pathol 24:353–361. https://doi.org/10.1080/019131200750060014
Choi S, Lim JW, Kim H (2020) Korean red ginseng inhibits amyloid-β-induced apoptosis and Nucling expression in human neuronal cells. Pharmacol 4:1–2. https://doi.org/10.1159/000505592
Colin-Barenque L, Pedraza-Chaverri J, Medina-Campos O, Jimenez-Martínez R, Bizarro-Nevares P, González-Villalva A, Rojas-Lemus M, Fortoul TI (2015) Functional and morphological olfactory bulb modifications in mice after vanadium inhalation. Toxicol Pathol 43:282–291. https://doi.org/10.1177/0192623314548668
Connolly BS, Lang AE (2014) Pharmacological treatment of Parkinson disease: a review. JAMA. 311:1670–1683. https://doi.org/10.1001/jama.2014.3654
Costa LG, Cole TB, Dao K, Chang YC, Coburn J, Garrick JM (2020) Effects of air pollution on the nervous system and its possible role in neurodevelopmental and neurodegenerative disorders. Pharmacol Therapeut 9:107523. https://doi.org/10.1016/j.pharmthera.2020.107523
Cui Y, Xie X, Jia F, He J, Li Z, Fu M, Hao H, Liu Y, Liu JZ, Cowan PJ, Zhu H (2015) Ambient fine particulate matter induces apoptosis of endothelial progenitor cells through reactive oxygen species formation. Cell Physiol Biochem 35:353–363. https://doi.org/10.1159/000369701
Dantzer R, O Connor JC, Freund GG, Johnson RW, Kelley KW (2008) From inflammation to sickness and depression: when the immune system subjugates the brain. Nat Rev Neurosci 9:46–56. https://doi.org/10.1038/nrn2297
Davis DA, Akopian G, Walsh JP, Sioutas C, Morgan TE, Finch CE (2013) Urban air pollutants reduce synaptic function of CA 1 neurons via an NMDA/NȮ pathway in vitro. J Neurochem 127(4):509–519
De Felice A, Scattoni ML, Ricceri L, Calamandrei G (2015) Prenatal exposure to a common organophosphate insecticide delays motor development in a mouse model of idiopathic autism. PLoS One 10:e0121663. https://doi.org/10.1371/journal.pone.0121663
Del Pino J, Zeballos G, Anadón MJ et al (2016) Cadmium-induced cell death of basal forebrain cholinergic neurons mediated by muscarinic M1 receptor blockade, increase in GSK-3β enzyme, βamyloid and tau protein levels. Arch Toxicol 90:1081–1092. https://doi.org/10.1007/s00204-015-1540-7
Del Zoppo G, Ginis I, Hallenbeck JM, Iadecola C, Wang X, Feurstein GZ (2000) Inflammation and stroke: putative role for cytokines, adhesion molecules and iNOS in brain response to ischemia. Brain Pathol 10:95–112. https://doi.org/10.1111/j.1750-3639.2000.tb00247.x
De Souza Lisboa SF, Gonçalves G, Komatsu F, Salci Queiroz CA, Aparecido Almeida A, Gastaldello Moreira E (2005) Developmental lead exposure induces depressive-like behavior in female rats. Drug Chem Toxicol 28:67–77. https://doi.org/10.1081/DCT-39696
Deng X, Zhang F, Wang L, Rui W, Long F, Zhao Y, Chen D, Ding W (2014) Airborne fine particulate matter induces multiple cell death pathways in human lung epithelial cells. Apoptosis. 19:1099–1112. https://doi.org/10.1007/s10495-014-0980-5
Di Maio R, Barrett P, Hoffman E, Barrett C, Zharikov A, Borah A, Greenamyre T (2016) a-Synuclein binds TOM20 and inhibits mitochondrial protein import in Parkinson’s disease. Sci Transl Med 8:342ra78. https://doi.org/10.1126/scitranslmed.aaf3634
Ding Y, Young CN, Li J, Luan X, McAllister JP II, Clark JD, Diaz FG (2003) Reduced inflammatory mediator expression by prereperfusion infusion into ischemic territory in rats: a real-time polymerase chain reaction analysis. Neurosci Lett 353:173–176. https://doi.org/10.1016/j.neulet.2003.09.055
Dolatshahi M, Farbood Y, Sarkaki A, Mansouri SM, Khodadadi A (2015) Ellagic acid improves hyperalgesia and cognitive deficiency in 6- hydroxidopamine induced rat model of Parkinson’s disease. Iran J Basic Med Sci 18:38–46
Ehsanifar M, Jafari AJ, Nikzad H, Zavareh MS, Atlasi MA, Mohammadi H, Tameh AA (2019) Prenatal exposure to diesel exhaust particles causes anxiety, spatial memory disorders with alters expression of hippocampal pro-inflammatory cytokines and NMDA receptor subunits in adult male mice offspring. Ecotoxicol Environ Saf 176:34–41. https://doi.org/10.1016/j.ecoenv.2019.03.090
Elwood E, Lim Z, Naveed H, Galea I (2017) The effect of systemic inflammation on human brain barrier function. Brain Behav Immun 62:35–40. https://doi.org/10.1016/j.bbi.2016.10.020
Emam B, Shahsavani A, Khodagholi F, Zarandi SM, Hopke PK, Hadei M, Behbahani H, Yarahmadi M (2020) Effects of PM 2.5 and gases exposure during prenatal and early-life on autism–like phenotypes in male rat offspring. Part Fibre Toxicol 17:1–6
Farbood Y, Shabani S, Sarkaki A, Mard SA, Ahangarpour A, Khorsandi L (2017) Peripheral and central administration of T3 improved the histological changes, memory and the dentate gyrus electrophysiological activity in an animal model of Alzheimer’s disease. Metab Brain Dis 32:693–701. https://doi.org/10.1007/s11011-016-9947-2
Fratiglioni L, De Ronchi D, Agüero-Torres H (1999) Worldwide prevalence and incidence of dementia. Drugs Aging 15:365–1375. https://doi.org/10.2165/00002512-199915050-00004
Fonken LK, Xu X, Weil ZM, Chen G, Sun Q, Rajagopalan S, Nelson RJ (2011) Air pollution impairs cognition, provokes depressive-like behaviors and alters hippocampal cytokine expression and morphology. Mol Psychiatry 16(10):987–995. https://doi.org/10.1038/mp.2011.76
Hajipour S, Farbood Y, Gharib-Naseri MK, Goudarzi G, Rashno M, Maleki H, Bakhtiari N, Nesari A, Khoshnam SE, Dianat M, Sarkaki B (2020) Exposure to ambient dusty particulate matter impairs spatial memory and hippocampal LTP by increasing brain inflammation and oxidative stress in rats. Life Sci 242:117210
Hara S, Mukai T, Kurosaki K, Kuriiwa F, Endo T (2002) Modification of the striatal dopaminergic neuron system by carbon monoxide exposure in free-moving rats, as determined by in vivo brain microdialysis. Arch Toxicol 76:596–605. https://doi.org/10.1007/s00204-002-0385-z
Hara S, Mukai T, Kurosaki K, Kuriiwa F, Endo T (2004) Characterization of hydroxyl radical generation in the striatum of free-moving rats due to carbon monoxide poisoning, as determined by in vivo microdialysis. Brain Res 1016:281–284. https://doi.org/10.1016/j.brainres.2004.05.047
Hawkes CH (2008) Parkinson’s disease and aging: same or different process? Mov Disord 23:47–53. https://doi.org/10.1002/mds.21766
He Q, Sawada M, Yamasaki N, Akazawa S, Furuta H, Uenishi H, Meng X, Nakahashi T, Ishigaki Y, Moriya J (2020) Neuroinflammation, oxidative stress, and neurogenesis in a mouse model of chronic fatigue syndrome, and the treatment with Kampo medicine. Biol Pharm Bull 43:110–115. https://doi.org/10.1248/bpb.b19-00616
Hernandez-Zimbron LF, Rivas-Arancibia S (2014) Deciphering an interplay of proteins associated with amyloid 훽 1–42 peptide and molecular mechanisms of Alzheimer’s disease. Rev Neurosci 25:773–783. https://doi.org/10.1515/revneuro-2014-0025
Hernandez-Zimbron LF, Rivas-Arancibia S (2015) Oxidative stress caused by ozone exposure induces β-amyloid 1–42 overproduction and mitochondrial accumulation by activating the amyloidogenic pathway. Neurosci 24:304–340. https://doi.org/10.1016/j.neuroscience.2015.07.011
Heusinkveld HJ, Wahle T, Campbell A, Westerink RH, Tran L, Johnston H, Stone V, Cassee FR, Schins RP (2016) Neurodegenerative and neurological disorders by small inhaled particles. Neurotoxicol 56:94–106. https://doi.org/10.1016/j.neuro.2016.07.007
Hill DS, Cabrera R, Wallis Schultz D, Zhu H, Lu W, Finnell RH, Wlodarczyk BJ (2015) Autism-like behavior and epigenetic changes associated with autism as consequences of in utero exposure to environmental pollutants in a mouse model. Behav Neurol:1–10. https://doi.org/10.1155/2015/426263
Hullmann M, Albrecht C, van Berlo D, Gerlofs-Nijland ME, Wahle T, Boots AW, Krutmann J, Cassee FR, Bayer TA, Schins RP (2017) Diesel engine exhaust accelerates plaque formation in a mouse model of Alzheimer’s disease. Part Fiber Toxicol 14:35. https://doi.org/10.1186/s12989-017-0213-5
Ijomone OM, Miah MR, Akingbade GT, Bucinca H, Aschner M (2020) Nickel-induced developmental neurotoxicity in C. elegans includes cholinergic, dopaminergic and GABAergic degeneration, altered behaviour, and increased SKN-1 activity. Neurotox Res 37:1018–1028. https://doi.org/10.1007/s12640-020-00175-3
Imai F, Suzuki H, Oda J, Ninomiya T, Ono K, Sano H, Sawada M (2007) Neuroprotective effect of exogenous microglia in global brain ischemia. J Cereb Blood Flow Metab 27:488–500. https://doi.org/10.1038/sj.jcbfm.9600362
Islam MT (2017) Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol Res 39:73–82. https://doi.org/10.1080/01616412.2016.1251711
Ghorani-Azam A, Riahi-Zanjani B, Balali-Mood M (2016) Effects of air pollution on human health and practical measures for prevention in Iran. J Res Med Sci 21:65. https://doi.org/10.4103/1735-1995.189646
Gonzalez-Fraguela ME, Hung ML, Vera H, Maragoto C, Noris E, Blanco L, Galvizu R, Robinson M (2013) Oxidative stress markers in children with autism spectrum disorders. J Adv Med Biomed Res. 19:307–317. https://doi.org/10.9734/BJMMR/2013/2335
Guerra R, Vera-Aguilar E, Uribe-Ramirez M, Gookin G, Camacho J, Osornio-Vargas AR, Mugica-Alvarez V, Angulo-Olais R, Campbell A, Froines J, Kleinman TM (2013) Exposure to inhaled particulate matter activates early markers of oxidative stress, inflammation and unfolded protein response in rat striatum. Toxicol Lett 222:146–154. https://doi.org/10.1016/j.toxlet.2013.07.012
Gunnarson E, Axehult G, Baturina G, Zelenin S, Zelenina M, Aperia A (2005) Lead induces increased water permeability in astrocytes expressing aquaporin 4. Neurosci. 136:105–114
Guo L, Li B, Miao JJ, Yun Y, Li GK, Sang N (2015) Seasonal variation in air particulate matter (PM10) exposure-induced ischemia-like injuries in the rat brain. Chem Res Toxicol 28:431–439. https://doi.org/10.1021/tx500392n
Gupta R, Shukla RK, Chandravanshi LP, Srivastava P, Dhuriya YK, Shanker J, Singh MP, Pant AB, Khanna VK (2017) Protective role of quercetin in cadmium-induced cholinergic dysfunctions in rat brain by modulating mitochondrial integrity and MAP kinase signaling. Mol Neurobiol 54:4560–4583. https://doi.org/10.1007/s12035-016-9950-y
Gupta R, Shukla RK, Pant AB, Khanna VK (2018) A dopamine-dependent activity in controlling the motor functions in cadmium induced neurotoxicity: neuroprotective potential of quercetin. Parkinsonism Relat Disord 46:e39. https://doi.org/10.1016/j.parkreldis.2017.11.127
Jahangiri HM, Sarkaki A, Farbood Y, Dianat M, Goudarzi G (2019) Gallic acid affects blood-brain barrier permeability, behaviors, hippocampus local EEG, and brain oxidative stress in ischemic rats exposed to dusty particulate matter. Environ Sci Pollut Res 17:1–2. https://doi.org/10.1007/s11356-019-07076-9
Jang S, Kim EW, Zhang Y, Lee J, Cho SY, Ha J, Kim H, Kim E (2018) Particulate matter increases beta-amyloid and activated glial cells in hippocampal tissues of transgenic Alzheimer's mouse: involvement of PARP-1. Biochem Biophys Res Commun 500(2):333–338. https://doi.org/10.1016/j.bbrc.2018.04.068
Jankovic J, Stacy M (2007) Medical management of levodopa-associated motor complications in patients with Parkinson’s disease. CNS Drugs 21:677–692. https://doi.org/10.2165/00023210-200721080-00005
Jeynes B, Provias J (2008) Evidence for altered LRP/RAGE expression in Alzheimer lesion pathogenesis. Curr Alzheimer Res 5:432–437. https://doi.org/10.2174/156720508785908937
Jiang LF, Yao TM, Zhu ZL, Wang C, Ji LN (2007) Impacts of Cd (II) on the conformation and self-aggregation of Alzheimer’s tau fragment corresponding to the third repeat of microtubule-binding domain. Biochim Biophys Acta, Proteins Proteomics 774(11):1414–1421. https://doi.org/10.1016/j.bbapap.2007.08.014
Johnson JM (2003) Genome-wide survey of human alternative pre-mRNA splicing with exon junction microarrays. Sci 302 (5653):2141-2144. https://doi.org/10.1126/science.1090100
Jones KL, Croen LA, Yoshida CK, Heuer L, Hansen R, Zerbo O, DeLorenze GN, Kharrazi M, Yolken R, Ashwood P, Van de Water J (2017) Autism with intellectual disability is associated with increased levels of maternal cytokines and chemokines during gestation. Mol Psychiatry 22(2):273. https://doi.org/10.1038/mp.2016.77
Jung YS, Jeong EM, Park EK, Kim YM, Sohn S, Lee SH, Baik EJ, Moon CH (2008) Cadmium induces apoptotic cell death through p38 MAPK in brain microvessel endothelial cells. Eur J Pharmacol 578:11–18. https://doi.org/10.1016/j.ejphar.2007.08.049
Kalkbrenner AE, Windham GC, Serre ML, Akita Y, Wang X, Hoffman K, Thayer BP, Daniels JL (2015) Particulate matter exposure, prenatal and postnatal windows of susceptibility, and autism spectrum disorders. Epidemiol 26:30–42. https://doi.org/10.1097/EDE.0000000000000173
Kam W, Liacos JW, Schauer JJ, Delfino RJ, Sioutas C (2012) Size segregated composition of particulate matter (PM) in major roadways and surface streets. Atmos Environ 55:90–97. https://doi.org/10.1016/j.atmosenv.2012.03.028
Kanninen KM, Lampinen R, Rantanen LM, Odendaal L, Jalava P, Chew S, White AR (2020) Olfactory cell cultures to investigate health effects of air pollution exposure: implications for neurodegeneration. Neurochem Int 19:104729. https://doi.org/10.1016/j.neuint.2020.104729
Karri V, Schuhmacher M, Kumar V (2020) A systems toxicology approach to compare the heavy metal mixtures (Pb, As, MeHg) impact in neurodegenerative diseases. Food Chem Toxicol 111257. https://doi.org/10.1016/j.fct.2020.111257
Keikhaei F, Mirshekar MA, Shahraki MR, Dashipour A (2020) Antiepileptogenic effect of myricitrin on spatial memory and learning in a kainate-induced model of temporal lobe epilepsy. Learn Motiv 69:101610. https://doi.org/10.1016/j.lmot.2019.101610
Kim SH, Fraser PE, Westaway D, George-Hyslop PH, Ehrlich ME, Gandy S (2010) Group II metabotropic glutamate receptor stimulation triggers production and release of Alzheimer’s amyloid β42 from isolated intact nerve terminals. J Neurosci 30:3870–3875
Kim SH, Knight EM, Saunders EL, Cuevas AK, Popovech M, Chen LC, Gandy S (2012) Rapid doubling of Alzheimer’s amyloid-β40 and 42 levels in brains of mice exposed to a nickel nanoparticle model of air pollution. F1000Res 1:70. https://doi.org/10.12688/f1000research.1-70.v1
Kleinman MT, Araujo JA, Nel A, Sioutas C, Campbell A, Cong PQ, Li H, Bondy SC (2008) Inhaled ultrafine particulate matter affects CNS inflammatory processes and may act via MAP kinase signaling pathways. Toxicol Lett 178:127–130. https://doi.org/10.1016/j.toxlet.2008.03.001
Lam KS, Aman MG, Arnold LE (2006) Neurochemical correlates of autistic disorder. A review of the literature. Rev Dev Disab 27:254–289. https://doi.org/10.1016/j.ridd.2005.03.003
Lee E, Hwang I, Park S, Hong S, Hwang B, Cho Y, Son J, Yu JW (2018) MPTP-driven NLRP3 inflammasome activation in microglia plays a central role in dopaminergic neurodegeneration. Cell Death Differ. https://doi.org/10.1016/j.toxlet.2008.03.001
Leung JW, Chung SS, Chung SK (2009) Endothelial endothelin-1 over-expression using receptor tyrosine kinase tie-1 promoter leads to more severe vascular permeability and blood brain barrier breakdown after transient middle cerebral artery occlusion. Brain Res 1266:121–129. https://doi.org/10.1016/j.brainres.2009.01070
Levesque S, Surace MJ, McDonald J, Block ML (2011a) Air pollution & the brain: subchronic diesel exhaust exposure causes neuroinflammation and elevates early markers of neurodegenerative disease. J Neuroinflammation 8(1):105. https://doi.org/10.1186/1742-2094-8-105
Levesque S, Taetzsch T, Lull ME, Kodavanti U, Stadler K, Wagner A, Johnson JA, Duke L, Kodavanti P, Surace MJ, Block ML (2011b) Diesel exhaust activates and primes microglia: air pollution, neuroinflammation, and regulation of dopaminergic neurotoxicity. Environ Health Perspect 119:1149–1155. https://doi.org/10.1289/ehp.1002986
Li K, Li L, Cui B, Gai Z, Li Q, Wang S, Yan J, Lin B, Tian L, Liu H, Liu X (2018) Early postnatal exposure to airborne fine particulate matter induces autism-like phenotypes in male rats. Toxicol Sci 162:189–199. https://doi.org/10.1093/toxsci/kfx240
Li N, Liu F, Song L, Zhang P, Qiao M, Zhao Q, Li W (2014) The effects of early life Pb exposure on the expression of IL1-beta, TNF-alpha and Abeta in cerebral cortex of mouse pups. J Trace Elem Med Biol 28:100–104. https://doi.org/10.1016/j.jtemb.2013.07.003
Liu Q, Babadjouni R, Radwanski R, Cheng H, Patel A, Hodis DM, He S, Baumbacher P, Russin JJ, Morgan TE, Sioutas C (2016) Stroke damage is exacerbated by nano-size particulate matter in a mouse model. PLoS One 11:4. https://doi.org/10.1371/journal.pone.0153376
Liu X, Zhang Y, Luo C, Kang J, Li J, Wang K, Ma P, Yang X (2017) At seeming safe concentrations, synergistic effects of PM2.5 and formaldehyde co-exposure induces Alzheimer-like changes in mouse brain. Oncotarget 8:98567–98579. https://doi.org/10.18632/oncotarget.21637
Lo EH, Dalkara T, Moskowitz MA (2003) Mechanisms, challenges and opportunities in stroke. Nat Rev Neurosci 4:399–415. https://doi.org/10.1038/nrn1106.Return
Lucero J, Suwannasual U, Herbert LM, McDonald JD, Lund AK (2017) The role of the lectin-like ox LDL receptor (LOX-1) in traffic-generated air pollution exposure-mediated alteration of the brain microvasculature in apolipoprotein (Apo) E knockout mice. Inhal Toxicol 29:266–281. https://doi.org/10.1080/08958378.2017.1357774
Luo J, Gurung S, Lee L, Henley JM, Wilkinson KA, Guo C (2017) Increased SUMO-2/3-ylation mediated by SENP3 degradation is protective against cadmium-induced caspase 3–dependent cytotoxicity. J Toxicol Sci 42:529–538. https://doi.org/10.2131/jts.42.529
Marshall HE, Stamler JS (2001) Inhibition of NF-kappaB by S-nitrosylation. Biochem 13:1688–1693. https://doi.org/10.1021/bi002239y
Martinez EM, Young AL, Patankar YR, Berwin BL, Wang L, von Herrmann KM, Weier JM, Havrda MC (2017) Editor’s highlight: Nlrp3 is required for inflammatory changes and nigral cell loss resulting from chronic intragastric rotenone exposure in mice. Toxicol Sci 159:64–75. https://doi.org/10.1093/toxsci/kfx117
Mason LH, Harp JP, Han DY (2014) Pb neurotoxicity: neuropsychological effects of lead toxicity. Biomed Res Int. https://doi.org/10.1155/2014/840547
Migliore L, Coppedè F (2009) Environmental-induced oxidative stress in neurodegenerative disorders and aging. Mutat Res Genet Toxicol Environ Mutagen 674:73–84. https://doi.org/10.1016/j.mrgentox.2008.09.013
Mills NL, Donaldson K, Hadoke PW, Boon NA, MacNee W, Cassee FR, Sandström T, Blomberg A, Newby DE (2009) Adverse cardiovascular effects of air pollution. Nat Clin Pract Cardiovasc Med 6:36–44. https://doi.org/10.1038/ncpcardio1399
Misonou H, Morishima-Kawashima M, Ihara Y (2000) Oxidative stress induces intracellular accumulation of amyloid β-protein (Aβ) in human neuroblastoma cells. Biochem 39(23):6951–6959. https://doi.org/10.1021/bi000169p
Modabbernia A, Velthorst E, Reichenberg A (2017) Environmental risk factors for autism: an evidence-based review of systematic reviews and meta-analyses. Molecular autism 8(1):13. https://doi.org/10.1186/s13229-017-0121-4
Mokoena ML, Harvey BH, Viljoen F, Ellis SM, Brink CB (2015) Ozone exposure of Flinders Sensitive Line rats is a rodent translational model of neurobiological oxidative stress with relevance for depression and antidepressant response. Psychopharmacol 232:2921–2938. https://doi.org/10.1007/s00213-015-3928-8
Moreno-Gonzalez I, Estrada LD, Sanchez-Mejias E, Soto C (2013) Smoking exacerbates amyloid pathology in a mouse model of Alzheimer’s disease. Nat Commun 4:1. https://doi.org/10.1038/ncomms2494
Morgan MJ, Liu ZG (2011) Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res 21(1):103–115. https://doi.org/10.1038/cr.2010.178
Mousavi SE, Heydarpour P, Reis J, Amiri M, Sahraian MA (2017) Multiple sclerosis and air pollution exposure: mechanisms toward brain autoimmunity. Med Hypotheses 100:23–30. https://doi.org/10.1016/j.mehy.2017.01.003
Muoth C, Aengenheister L, Kucki M, Wick P, Buerki-Thurnherr T (2016) Nanoparticle transport across the placental barrier: pushing the field forward! Nanomedicine (London, England) 11(8):941–957. https://doi.org/10.2217/nnm-2015-0012
Nephew BC, Nemeth A, Hudda N, Beamer G, Mann P, Petitto J, Cali R, Febo M, Kulkarni P, Poirier G, King J (2020) Traffic-related particulate matter affects behavior, inflammation, and neural integrity in a developmental rodent model. Environ Res 183:109242
Oberdorster G, Sharp Z, Atudorei V, Elder A, Gelein R, Kreyling W, Cox C (2004) Translocation of inhaled ultrafine particles to the brain. Inhal Toxicol 16:437–445. https://doi.org/10.1080/08958370490439597
Oppenheim HA, Lucero J, Guyot A-C, Herbert LM, McDonald JD, Mabondzo A, Lund AK (2013) Exposure to vehicle emissions results in altered blood brain barrier permeability and expression of matrix metalloproteinases and tight junction proteins in mice. Part Fibre Toxicol 10:1–14. https://doi.org/10.1186/1743-8977-10-62
Orre M, Kamphuis W, Osborn LM, Jansen AH, Kooijman L, Bossers K, Hol EM (2014) Isolation of glia from Alzheimer’s mice reveals inflammation and dysfunction. Neurobiol Aging 35:2746–2760. https://doi.org/10.1016/j.neurobiolaging.2014.06.004
Pardo CA, Vargas DL, Zimmerman AW (2005) Immunity, neuroglia and neuroinflammation in autism. Int Rev Psychiatry 17:485–495. https://doi.org/10.1080/02646830500381930
Park HJ, Zhao TT, Kim SH, Lee CK, Hwang BY, Lee KE, Lee MK (2020) Ethanol extract from Gynostemma pentaphyllum ameliorates dopaminergic neuronal cell death in transgenic mice expressing mutant A53T human alpha-synuclein. Neural Regen Res 15:361. https://doi.org/10.4103/1673-5374.265557
Paval D (2017) A dopamine hypothesis of autism spectrum disorder. Dev Neurosci 39(5):355–360
Pereyra-Munoz N, Rugerio-Vargas C, Angoa-Pérez M, Borgonio-Pérez G, Rivas-Arancibia S (2006) Oxidative damage in substantia nigra and striatum of rats chronically exposed to ozone. J Chem Neuroanat 31:114–123. https://doi.org/10.1016/j.jchemneu.2005.09.006
Petersone EW, Cardoso ER (1983) The blood-brain barrier following experimental subarachnoid hemorrhage. Part 2: response to mercuric chloride infusion. J Neurosurg 58:345–351. https://doi.org/10.3171/jns.1983.58.3.0345
Pinheiro CR, Moura EG, Manhaes AC, Fraga MC, Claudio-Neto S, Abreu-Villaça Y, Oliveira E, Lisboa PC (2015) Concurrent maternal and pup postnatal tobacco smoke exposure in Wistar rats changes food preference and dopaminergic reward system parameters in the adult male offspring. Neurosci 301:178–192. https://doi.org/10.1016/j.neuroscience.2015.06.001
Rahman MA, Rahman MS, Uddin MJ, Mamum-Or-Rashid AN, Pang MG, Rhim H (2020) Emerging risk of environmental factors: insight mechanisms of Alzheimer’s diseases. Environ Sci Pollut Res 23:1–4. https://doi.org/10.1007/s11356-020-08243-z
Rao KV, Jayakumar AR, Reddy PV, Tong X, Curtis KM, Norenberg MD (2010) Aquaporin-4 in manganese-treated cultured astrocytes. Glia. 58:1490–1499
Rempe RG, Hartz AMS, Soldner ELB, Sokola BS, Alluri SR, Abner EL, Kryscio RJ, Pekcec A, Schlichtiger J, Bauer B (2018) Matrix metalloproteinase-mediated blood-brain barrier dysfunction in epilepsy. J Neurosci 38:4301–4315
Rescorla LA, Winder-Patel BM, Paterson SJ, Pandey J, Wolff JJ, Schultz RT, Piven J (2019) Autism spectrum disorder screening with the CBCL/1½–5: findings for young children at high risk for autism spectrum disorder. Autism 23:29–38. https://doi.org/10.1177/1362361317718482
Rivas-Arancibia S, Dorado-Martínez C, Colin-Barenque L, Kendrick KM, de la Riva C, Guevara-Guzmán R (2003) Effect of acute ozone exposure on locomotor behavior and striatal function. Pharmacol Biochem Behav 74:891–900. https://doi.org/10.1016/S0091-3057(03)00011-X
Rivas-Arancibia S, Guevara-Guzmán R, Lopez-Vidal Y, Rodríguez-Martínez E, Zanardo-Gomes M, Angoa-Pérez M, Raisman-Vozari R (2009) Oxidative stress caused by ozone exposure induces loss of brain repair in the hippocampus of adult rats. Toxicol Sci 113:187–197. https://doi.org/10.1093/toxsci/kfp252
Rivas-Arancibia S, Hernández-Zimbrón LF, Rodríguez-Martínez E, Borgonio-Pérez G, Velumani V, Durán-Bedolla J (2013) Chronic exposure to low doses of ozone produces a state of oxidative stress and blood-brain barrier damage in the hippocampus of rat. Adv Biosci Biotechnol 04 (11):24–29. https://doi.org/10.4236/abb.2013.411A2004
Rivas-Arancibia S, Rodríguez-Martínez E, Badillo-Ramírez I, López-González U, Saniger JM (2017) Structural changes of amyloid beta in hippocampus of rats exposed to ozone: a Raman spectroscopy study. Front Mol Neurosci 22(10):137. https://doi.org/10.3389/fnmol.2017.00137
Rivas-Arancibia S, Zimbron LF, Rodriguez-Martinez E, Maldonado PD, Borgonio Perez G, Sepulveda-Parada M (2015) Oxidative stress-dependent changes in immune responses and cell death in the substantia nigra after ozone exposure in rat. Front Aging Neurosci 7:65. https://doi.org/10.3389/fnagi.2015.00065
Rocha E, Miranda B, Sanders L (2018) Alpha-synuclein: pathology, mitochondrial dysfunction and neuroinflammation in Parkinson’s disease. Neurobiol Dis 109:249–257. https://doi.org/10.1016/j.nbd.2017.04.004
Rose S, Melnyk S, Pavliv O, Bai S, Nick TG, Frye RE, James SJ (2012) Evidence of oxidative damage and inflammation associated with low glutathione redox status in the autism brain. Transl Psychiatry 2:e134. https://doi.org/10.1038/tp.2012.61
Sacino AN, Brooks M, Thomas MA, McKinney AB, Lee S, Regenhardt RW, McGarvey NH, Ayers JI, Notterpek L, Borchelt DR, Golde TE (2014) Intramuscular injection of α-synuclein induces CNS α-synuclein pathology and a rapid-onset motor phenotype in transgenic mice. Proc Natl Acad Sci USA 111(29):10732–10737. https://doi.org/10.1073/pnas.1321785111
Sadek KM, Lebda MA, Abouzed TK (2019) The possible neuroprotective effects of melatonin in aluminum chloride-induced neurotoxicity via antioxidant pathway and Nrf2 signaling apart from metal chelation. Environ Sci Pollut Res 26:9174–9183. https://doi.org/10.1007/s11356-019-04430-9
Salvi A, Liu H, Salim S (2020) Involvement of oxidative stress and mitochondrial mechanisms in air pollution-related neurobiological impairments. Neurobiol Stress 12:100205. https://doi.org/10.1016/j.ynstr.2019.100205
Sang N, Yun Y, Li H, Hou L, Han M, Li G (2010) SO2 inhalation contributes to the development and progression of ischemic stroke in the brain. Toxicol Sci 114(2):226–236. https://doi.org/10.1093/toxsci/kfq010
Santiago-Lopez D, Bautista-Martinez JA, Reyes-Hernandez CI, Aguilar-Martinez M, Rivas-Arancibia S (2010) Oxidative stress, progressive damage in the substantia nigra and plasma dopamine oxidation, in rats chronically exposed to ozone. Toxicol Lett 197:193–200. https://doi.org/10.1016/j.toxlet.2010.05.020
Sardar SB, Fine PM, Sioutas C (2005) Seasonal and spatial variability of the size-resolved chemical composition of particulate matter (PM10) in the Los Angeles Basin. J Geophys Res Oceans 16:110 (D7). https://doi.org/10.1029/2004JD004627
Sarkaki A, Farbood Y, Badavi M, Ghadiri A, Ghasemi Dehcheshmeh M, Mansouri E, Navabi SP (2018) The protective effect of betulinic acid on microvascular responsivity and protein expression in Alzheimer disease induced by cerebral micro-injection of beta-amyloid and streptozotocin. Microcirc 25:e12503. https://doi.org/10.1111/micc.12503
Sedaghat G, Mirshekar MA, Amirpour M, Montazerifar F, Miri S, Shourestani S (2019) Sub-chronic administration of brewed coffee on rat behavior and cognition and oxidative stress Alzheimer’s disease model. Clin Nutr Exp 28:62–73. https://doi.org/10.1016/j.yclnex.2019.10.005
Seelen M, Toro Campos RA, Veldink JH, Visser AE, Hoek G, Brunekreef B, van der Kooi AJ, de Visser M, Raaphorst J, van den Berg LH, Vermeulen RC (2017) Long-term air pollution exposure and amyotrophic lateral sclerosis in Netherlands: a population-based case–control study. Environ Health Perspect 125:097023. https://doi.org/10.1289/EHP1115
Shabani S, Farbood Y, Sarkaki A, Mard SA, Ahangarpour A, Khorsandi L (2019) The effect of triiodothyronine on the hippocampal long-term potentiation in an animal model of the Alzheimer’s disease: the role of BDNF and reelin. Neurol Psychol Brain Res 33:82–88. https://doi.org/10.1016/j.npbr.2019.07.004
Shabani S, Sarkaki A, Mard SA, Ahangarpour A, Khorsandi L, Farbood Y (2016) Central and peripheral administrations of levothyroxine improved memory performance and amplified brain electrical activity in the rat model of Alzheimer’s disease. Neuropeptides 59:111–116. https://doi.org/10.1016/j.npep.2016.09.003
Shabani S, Mirshekar MA (2018) Diosmin is neuroprotective in a rat model of scopolamine-induced cognitive impairment. Biomed Pharmacother 108:1376–1383. https://doi.org/10.1016/j.biopha.2018.09.127
Shati AA, Alfaifi MY (2019) Trans-resveratrol inhibits tau phosphorylation in the brains of control and cadmium chloride-treated rats by activating PP2A and PI3K/Akt induced-inhibition of GSK3β. Neurochem Res 44:357–373. https://doi.org/10.1007/s11064-018-2683-8
Shaw CA, Li Y, Tomljenovic L (2013) Administration of aluminium to neonatal mice in vaccine-relevant amounts is associated with adverse long term neurological outcomes. J Inorg Biochem 128:237–244. https://doi.org/10.1016/j.jinorgbio.2013.07.022
Sobolewski M, Anderson T, Conrad K, Marvin E, Klocke C, Morris-Schaffer K, Allen JL, Cory-Slechta DA (2018) Developmental exposures to ultrafine particle air pollution reduces early testosterone levels and adult male social novelty preference: risk for Children’s sex-biased neurobehavioral disorders. Neurotoxicol 68:203–211
Sprowles JL, Amos-Kroohs RM, Braun AA, Sugimoto C, Vorhees CV, Williams MT (2018) Developmental manganese, lead, and barren cage exposure have adverse long-term neurocognitive, behavioral and monoamine effects in Sprague-Dawley rats. Neurotoxicol Teratol 67:50–64. https://doi.org/10.1016/j.ntt.2018.04.001
Suades-Gonzalez E, Gascon M, Guxens M, Sunyer J (2015) Air pollution and neuropsychological development: a review of the latest evidence. Endocrinol 156(10):3473–3482. https://doi.org/10.1210/en.2015-1403
Suwannasual U, Lucero J, McDonald JD, Lund AK (2018) Exposure to traffic-generated air pollutants mediates alterations in brain microvascular integrity in wildtype mice on a high-fat diet. Environ Res 160:449–461. https://doi.org/10.1016/j.envres.2017.10.029
Takashima A (2006) GSK-3 is essential in the pathogenesis of Alzheimer’s disease. J Alzheimers Dis 9:309–317
Tamegart L, Abbaoui A, Makbal R, Zroudi M, Bouizgarne B, Bouyatas MM, Gamrani H (2019) Crocus sativus restores dopaminergic and noradrenergic damages induced by lead in Meriones shawi: a possible link with Parkinson’s disease. Acta Histochem 121:171–181. https://doi.org/10.1016/j.acthis.2018.12.003
Tamagawa E, Van Eeden SF (2006) Impaired lung function and risk for stroke: role of the systemic inflammation response? Chest 130:1631–1633. https://doi.org/10.1378/chest.130.6.1631
Tan DW, Foo YZ, Downs J, Finlay-Jones A, Leonard H, Licari MK, Mullan N, Symons M, Varcin KJ, Whitehouse AJ, Alvares GA (2020) A preliminary investigation of the effects of prenatal alcohol exposure on facial morphology in children with autism spectrum disorder. Alcohol 121:171–181. https://doi.org/10.1016/j.alcohol.2020.03.010
Theoharides TC, Asadi S, Patel AB (2013) Focal brain inflammation and autism. J Neuroinflammation 10:46
Thirtamara Rajamani K, Doherty-Lyons S, Bolden C, Willis D, Hoffman C, Zelikoff J, Chen LC, Gu H (2013) Prenatal and early-life exposure to high-level diesel exhaust particles leads to increased locomotor activity and repetitive behaviors in mice. Autism Res 6(4):248–257. https://doi.org/10.1002/aur.1287
Thomson EM, Kumarathasan P, Calderon-Garciduenas L, Vincent R (2007) Air pollution alters brain and pituitary endothelin-1 and inducible nitric oxide synthase gene expression. Environ Res 105(2):224–233. https://doi.org/10.1016/j.envres.2007.06.005
Tracey KJ (2009) Reflex control of immunity. Nat Rev Immunol 9:418–428. https://doi.org/10.1038/nri2566
Uchino S, Waga C (2013) SHANK3 as an autism spectrum disorder-associated gene. Brain Dev 35:106–110
Vadillo-Ortega F, Osornio-Vargas A, Buxton MA, Sánchez BN, Rojas-Bracho L, Viveros-Alcaráz M, Castillo-Castrejón M, Beltrán-Montoya J, Brown DG, O’Neill MS (2014) Air pollution, inflammation and preterm birth: apotential mechanistic link. Med Hypotheses 82:219–224. https://doi.org/10.1016/j.mehy.2013.11.042
Valentino SA, Tarrade A, Aioun J, Mourier E, Richard C, Dahirel M, Rousseau-Ralliard D, Fournier N, Aubrière MC, Lallemand MS, Camous S (2016) Maternal exposure to diluted diesel engine exhaust alters placental function and induces intergenerational effects in rabbits. Part Fibre Toxicol 13:39. https://doi.org/10.1186/s12989-016-0151-7
Wang BR, Shi JQ, Ge NN, Ou Z, Tian YY, Jiang T, Zhou JS, Xu J, Zhang YD (2018a) PM2.5 exposure aggravates oligomeric amyloid beta-induced neuronal injury and promotes NLRP3 inflammasome activation in an in vitro model of Alzheimer’s disease. J Neuroinflammation 15:132. https://doi.org/10.1186/s12974-018-1178-5
Wang H, Zhang L, Abel GM, Storm DR, Xia Z (2018b) Cadmium exposure impairs cognition and olfactory memory in male C57BL/6 mice. Toxicol Sci 161:87–102. https://doi.org/10.1093/toxsci/kfx202
Wang Y, Liu D, Zhang H, Wang Y, Wei L, Liu Y, Liao J, Gao HM, Zhou H (2017) Ultrafine carbon particles promote rotenone-induced dopamine neuronal loss through activating microglial NADPH oxidase. Toxicol Appl Pharmacol 322:51–59. https://doi.org/10.1016/j.taap.2017.03.005
Wei H, Liang F, Meng G, Nie Z, Zhou R, Cheng W, Wu X, Feng Y, Wang Y (2016) Redox/methylation mediated abnormal DNA methylation as regulators of ambient fine particulate matter-induced neurodevelopment related impairment in human neuronal cells. Sci Rep 6:33402. https://doi.org/10.1038/srep33402
Wingate M, Mulvihill B, Kirby RS, Pettygrove S, Cunniff C, Meaney F et al (2012) Prevalence of autism spectrum disorders-autism and developmental disabilities monitoring network, 14 sites, United States, 2008. MMWR Surveill Summ 61:1–19
Win-Shwe TT, Kyi-Tha-Thu C, Moe Y, Fujitani Y, Tsukahara S, Hirano S (2016) Exposure of BALB/c mice to diesel engine exhaust origin secondary organic aerosol (DE-SOA) during the developmental stages impairs the social behavior in adult life of the males. Front Neurosci 9:524. https://doi.org/10.3389/fnins.2015.00524
Win-Shwe TT, Mitsushima D, Yamamoto S, Fujitani Y, Funabashi T, Hirano S, Fujimaki H (2009) Extracellular glutamate level and NMDA receptor subunit expression in mouse olfactory bulb following nanoparticle-rich diesel exhaust exposure. Inhal Toxicol 21:828–836. https://doi.org/10.1080/08958370802538068
Woodruff TM, Thundyil J, Tang SC, Sobey CG, Taylor SM, Arumugam TV (2011) Pathophysiology, treatment, and animal and cellular models of human ischemic stroke. Mol Neurodegener 6:11. https://doi.org/10.1038/tp.2012.61
Xu MX, Ge CX, Li Q, Lou DS, Hu LF, Sun Y, Xiong MX, Lai LL, Zhong SY, Yi C, Wang BC (2020) Fisetin nanoparticles protect against PM2. 5 exposure-induced neuroinflammation by down-regulation of astrocytes activation related NF-κB signaling pathway. J Funct Foods 65:103716. https://doi.org/10.1016/j.jff.2019.103716
Yang M, Bozdagi O, Scattoni ML, Wöhr M, Roullet FI, Katz AM, Abrams DN, Kalikhman D, Simon H, Woldeyohannes L, Zhang JY (2012) Reduced excitatory neurotransmission and mild autism-relevant phenotypes in adolescent Shank3 null mutant mice. J Neurosci 32:6525–6541. https://doi.org/10.1523/JNEUROSCI.6107-11.2012
Yang Z, Chen Y, Zhang Y, Li R, Dong C (2017) The role of pro-/anti-inflammation imbalance in Aβ42 accumulation of rat brain co-exposed to fine particle matter and sulfur dioxide. Toxicol Mech Methods 27:568–574. https://doi.org/10.1080/15376516.2017.1337256
Yan W, Zhao X, Chen H, Zhong D, Jin J, Qin Q, Zhang H, Ma S, Li G (2016) β-Dystroglycan cleavage by matrix metalloproteinase-2/-9 disturbs aquaporin-4 polarization and influences brain edema in acute cerebral ischemia. Neurosci. 326:141–157
Yavuz Y, Mollaoglu H, Yürümez Y, Ucok K, Duran L, Tünay K, Akgün L (2013) Therapeutic effect of magnesium sulphate on carbon monoxide toxicity-mediated brain lipid peroxidation. Eur Rev Med Pharmacol Sci 1:28–33
Yokota S, Oshio S, Takeda K (2016) In utero exposure to diesel exhaust particles induces anxiogenic effects on male offspring via chronic activation of serotonergic neuron in dorsal raphe nucleus. J Toxicol Sci 41:583–593. https://doi.org/10.2131/jts.41.583
Zarandi MS, Shahsavani A, Khodagholi F, Fakhri Y (2019) Co-exposure to ambient PM2.5 plus gaseous pollutants increases amyloid β1–42 accumulation in the hippocampus of male and female rats. Toxin Rev 7:1. https://doi.org/10.1080/15569543.2019.1611604
Zhang C, Meng Q, Zhang X, Wu S, Wang S, Chen R, Li X (2016a) Role of astrocyte activation in fine particulate matter-enhancement of existing ischemic stroke in Sprague-Dawley male rats. J Toxicol Environ Heal A 79:393–401. https://doi.org/10.1080/15287394.2016.1176615
Zhang L, Wang H, Abel GM, Storm DR, Xia Z (2020) The effects of gene-environment interactions between cadmium exposure and apolipoprotein E4 on memory in a mouse model of Alzheimer’s disease. Toxicol Sci 173:189–201
Zhang W, Wang T, Pei Z, Mille D, Wu X, Block M, Zhang J (2016b) Aggregated a-synuclein activates microglia: a process leading to disease progression in Parkinson’s disease. FASEB J 19:533–542. https://doi.org/10.1096/fj.04-2751com
Zhang X, Gu Y, Li P, Jiang A, Sheng X, Jin X, Shi Y, Li G (2019) Matrix metalloproteases-mediated cleavage on beta-dystroglycan may play a key role in the blood-brain barrier after intracerebral hemorrhage in rats. Med Sci Monit 25:794–800
Zhao Y, Dang M, Zhang W, Lei Y, Ramesh T, Veeraraghavan VP, Hou X (2020) Neuroprotective effects of Syringic acid against aluminium chloride induced oxidative stress mediated neuroinflammation in rat model of Alzheimer’s disease. J Funct Foods 71:104009. https://doi.org/10.1016/j.jff.2020.104009
Acknowledgments
The author is thankful for the Medical Plants Research Center, Shahrekord University of Medical Sciences, Shahrekord, Iran, for supporting this study.
Author information
Authors and Affiliations
Contributions
Project administration, writing—original draft, writing—review and editing, approval of manuscript for publication: Sahreh Shabani
Corresponding author
Ethics declarations
Ethics approval
All procedures were carried out in accordance with the regulations of the Shahrekord University of Medical Sciences.
Consent for publication
Not applicable.
Competing interests
The author declares that there is no conflict of interest.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shabani, S. A mechanistic view on the neurotoxic effects of air pollution on central nervous system: risk for autism and neurodegenerative diseases. Environ Sci Pollut Res 28, 6349–6373 (2021). https://doi.org/10.1007/s11356-020-11620-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-11620-3