Abstract
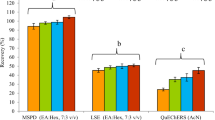
Termites are one of the major pests of sugarcane. Bifenthrin has label claim for use against sugarcane termites and is applied at the time of planting over the cane setts in the furrows. A thorough knowledge on the dissipation kinetics of bifenthrin in the soil and cane setts provides an insight into its dynamic behavior in the soil–plant–environment continuum, and hence, was studied in detail. The recoveries of the method adopted to determine the residues of bifenthrin in the soil, sugarcane setts, stem, and leaf were in the range of 92.78–98.19% at three levels of fortification ranged between 0.01 and 0.1 μg/g of matrix. At the recommended dose (100 g a.i./ha), bifenthrin was found to persist in the soil and cane setts up to 60 and 75 days after treatment (DAT) with the half-lives of 16.4 and 25.0 days, respectively. The dissipation kinetics of bifenthrin followed the biphasic model. At double the recommended dose, the residues persisted up to 75 DAT and reached below the detectable limit (< 0.01 μg/g) on 90 DAT both in the soil and setts. The half-life of bifenthrin was 22.0 days in the sandy clay loam soil and 17.6 days in/on the sugarcane setts. In the present study, dissipation by photolysis has been ruled out, as the treated setts were not exposed to sunlight. This could be one of the reasons for longer persistence of bifenthrin in the sugarcane setts up to 75 DAT with the half-life of more than 2 weeks. The bifenthrin residues were not detected in the leaf and stem of sugarcane plant throughout the experimental period. There could be harmful effects to earthworms (risk quotient > 1.0) due to the presence of bifenthrin residues in the soil of tropical sugarcane ecosystem.


Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anastassiades M, Lehotay SJ, Štajnbaher D, Schenck FJ (2003) Fast and easy multiresidue method employing acetonitrile extraction/partitioning and dispersive solid-phase extraction for the determination of pesticide residues in produce. J AOAC Int 86(2):412–431
Ccanccapa A, Masiá A, Navarro-Ortega A, Picó Y, Barceló D (2016) Pesticides in the Ebro river basin: occurrence and risk assessment. Environ Pollut 211:414–424
Central Insecticide Board & Registration Committee (2020) Major uses of pesticides registered under the Insecticides Act, 1968. http://ppqs.gov.in/sites/default/files/approved_use_of_insecticides.pdf Accessed 01 Apr 2020
Chauhan R, Monga S, Kumari B (2012) Dissipation and decontamination of bifenthrin residues in tomato (Lycopersicon esculentum Mill). Bull Environ Contam Toxicol 89:181–186
David H, Nandagopal V (1986) Pests of sugarcane–distribution, symptomatology of attack and identification. In: David H, Easwaramoorthy S, Jayanthi R (eds) Sugarcane Entomology in India. Sugarcane Breeding Institute, Coimbatore, pp 1–30
European Commission (2010) Guidance document on pesticide residue analytical methods. Document No. SANCO/825/00/rev. 8.1/16/11/2010. http://ec.europa.eu/food/plant/protection/resources/guide_doc_825-00_rev7_en.pdf. Accessed 21 Nov 2015
European Communities (2003) Technical guidance document on risk assessment part II. European Commission Joint Research Centre. EUR 20418 EN/2 Available at <https://echa.europa.eu/documents/10162/16960216/tgdpart2_2ed_en.pdf> Accessed on Apr, 07 2020
European Food Safety Authority (2014) EFSA guidance document for evaluating laboratory and field dissipation studies to obtain DegT50 values of active substances of plant protection products and transformation products of these active substances in soil. EFSA J 12(5):3662
Kumari S, Ramprakash, Kumari B (2014) Persistence of bifenthrin in soil under the cover of okra crop. Pestic Res J 26(1):78–81
Lewis KA, Tzilivakis J, Warner D, Green A (2016) An international database for pesticide risk assessments and management. Hum Ecol Risk Assess 22(4):1050–1064
Mukherjee I, Singh R, Govil JN (2010) Risk assessment of a synthetic pyrethroid, bifenthrin on pulses. Bull Environ Contam Toxicol 84:294–300
NFCSF (National Federation of Cooperative Sugar Factories Limited) (2019) Sugar statistics. Sugar Statistics. Coop Sugar 50(8):46–52
Patra S, Ganguly P, Barik SR, Goon A, Mandal J, Samanta A, Bhattacharyya A (2020) Persistence behavious and safety risk evaluation of pyridalyl in tomato and cabbage. Food Chem 309:125711
Paul B, Khan MA, Paul S, Shankarganesh K, Chakravorty S (2018) Termites and Indian agriculture. In: Khan MA, Ahmad W (eds) Termites and sustainable management, sustainability in plant and crop protection. Springer International Publishing, pp 51–96
PPDB (2020) In: Agriculture & Environment Research Unit, The University of Hertfordshire (ed) Pesticide properties database, Hatfield Available at https://sitem.herts.ac.uk/aeru/ppdb/en/Reports/631.htm. Accessed on Apr 04, 2020
Ramasubramanian T (2013) Persistence and dissipation kinetics of clothianidin in the soil of tropical sugarcane ecosystem. Water Air Soil Pollut 224(3):1468
Ramasubramanian T, Paramasivam M (2016) Dissipation behavior of phorate and its toxic metabolites in the sandy clay loam soil of a tropical sugarcane ecosystem using a single-step sample preparation method and GC–MS. J Sep Sci 39(20):3973–3982
Ramasubramanian T, Paramasivam M (2017) Determination and dissipation of fipronil and its metabolites in/on sugarcane crop. Int J Environ Anal Chem 97(11):1037–1052
Ramasubramanian T, Paramasivam M (2018) Persistence and metabolism of carbofuran in the soil and sugarcane plant. Environ Monit Assess 190(9):538
Ramasubramanian T, Paramasivam M, Jayanthi R (2012) Rapid and sensitive analytical method for simultaneous determination of imidacloprid and thiamethoxam residues in the soils of sugarcane ecosystem. Water Air Soil Pollut 223(9):6045–6050
Ramasubramanian T, Paramasivam M, Nirmala R (2016a) Development, validation and application of a sensitive analytical method for residue determination and dissipation of imidacloprid in sugarcane under tropical field condition. Environ Monit Assess 188(6):375
Ramasubramanian T, Paramasivam M, Jayanthi R, Nirmala R (2016b) Persistence and dissipation kinetics of chlorantraniliprole 0.4G in the soil of tropical sugarcane ecosystem. Environ Monit Assess 188(1):33
Rao NV, Maheswari TU, Prasad PR, Naidu VG, Savithri P (2003) Integrated insect pest management. Agrobios (India), Jodhpur
Sakthiselvi T, Paramasivam M, Vasanthi D, Bhuvaneswari K (2020) Persistence, dietary and ecological risk assessment of indoxacarb residue in/on tomato and soil using GC–MS. Food Chem 328:127134
Shi K, Li L, Yuan L, Li W, Liu F (2016) Residues and risk assessment of bifenthrin and chlorfenapyr in eggplant and soil under open ecosystem conditions. Int J Environ Anal Chem 96:173–184. https://doi.org/10.1080/03067319.2015.1137908
Tariq SR, Ahmed D, Farooq A, Rasheed S, Mansoor M (2017) Photodegradation of bifenthrin and deltamethrin—effect of copper amendment and solvent system. Environ Monit Assess 189:71
Tewary DK, Kumar V, Ravindranath SD, Shanker A (2005) Dissipation behavior of bifenthrin residues in tea and its brew. Food Control 16:231–237
You X, Jiang N, Liu F, Liu C, Wang S (2013) Dissipation and residue of bifenthrin in wheat under field conditions. Bull Environ Contam Toxicol 90:238–241
Acknowledgments
The authors are grateful to Dr. Bakshi Ram, Director, ICAR-Sugarcane Breeding Institute, and Dr. R. Viswanathan, Head, Division of Crop Protection, for their constant encouragement and support in carrying out this research. The technical support rendered by Mrs. C. Yogambal, Technical Assistant (Entomology), in imposing treatments, sampling, and sample preparation is acknowledged.
Funding
This research was funded by the Indian Council of Agricultural Research as Institute’s (ICAR-SBI) project.
Author information
Authors and Affiliations
Contributions
TR conceived the research, conducted the laboratory and field experiments, analyzed the data, and wrote the manuscript. MP standardized the GC-ECD and determined the residues. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical approval and consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Responsible editor: Thomas D. Bucheli
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 37 kb)
Rights and permissions
About this article
Cite this article
Ramasubramanian, T., Paramasivam, M. Bifenthrin in the tropical sugarcane ecosystem: persistence and environmental risk assessment. Environ Sci Pollut Res 28, 3524–3532 (2021). https://doi.org/10.1007/s11356-020-10757-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-10757-5