Abstract
Nitrated monoaromatic hydrocarbons (NMAHs) are ubiquitous in the environment and an important part of atmospheric humic-like substances (HULIS) and brown carbon. They are ecotoxic and with underresearched toxic potential for humans. NMAHs were determined in size-segregated ambient particulate matter collected at two urban sites in central Europe, Ostrava and Kladno, Czech Republic. The average sums of 12 NMAHs (Σ12NMAH) measured in winter PM10 samples from Ostrava and Kladno were 102 and 93 ng m−3, respectively, and 8.8 ng m−3 in summer PM10 samples from Ostrava. The concentrations in winter corresponded to 6.3–7.3% and 2.6–3.1% of HULIS-C and water-soluble organic carbon (WSOC), respectively. Nitrocatechols represented 67–93%, 61–73% and 28–96% of NMAHs in PM10 samples collected in winter and summer at Ostrava and in winter at Kladno, respectively. The mass size distribution of the targeted substance classes peaked in the submicrometre size fractions (PM1), often in the PM0.5 size fraction especially in summer. The bioaccessible fraction of NMAHs was determined by leaching PM3 samples in two simulated lung fluids, Gamble’s solution and artificial lysosomal fluid (ALF). More than half of NMAH mass is found bioaccessible, almost complete for nitrosalicylic acids. The bioaccessible fraction was generally higher when using ALF (mimics the chemical environment created by macrophage activity, pH 4.5) than Gamble’s solution (pH 7.4). Bioaccessibility may be negligible for lipophilic substances (i.e. log KOW > 4.5).
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
Nitrated monoaromatic hydrocarbons (NMAHs) are an important part of humic-like substances (HULIS), which in turn constitute a large mass fraction of particulate matter (PM) water-soluble organic carbon (WSOC; Graber and Rudich 2006) and brown carbon (Laskin et al. 2015). NMAHs are primarily emitted into the atmosphere or formed by secondary processes. Gas- and aqueous-phase oxidation and nitration of lignin thermal decomposition products (m-cresol, phenols, methoxyphenols, catechols, salicylic acid, etc.) are major formation pathways for 4-nitrocatechol (4-NC), methylnitrocatechols (MNCs), nitroguaiacols (NGs) and nitrosalicylic acids (NSAs; Iinuma et al. 2010; Kelly et al. 2010; Kroflič et al. 2015; Frka et al. 2016; Teich et al. 2017; Xie et al. 2017; Finewax et al. 2018; Wang et al. 2019). Traffic and coal and wood combustion, as well as industry and agricultural use of pesticides, are considered as main primary emission sources of nitrophenols (NPs), which can also be secondarily formed in the atmosphere (Harrison et al. 2005; Iinuma et al. 2007; Kitanovski et al. 2012; Inomata et al. 2015; Wang et al. 2018). 4-NC and MNCs are well-established tracers for biomass burning secondary organic aerosols (Iinuma et al. 2010; Kitanovski et al. 2012; Kahnt et al. 2013; Caumo et al. 2016; Chow et al. 2016). NSAs may also be formed in secondary organic aerosols exposed to NO3 radicals (Ramaswamy et al. 2019). NMAHs may represent up to 1% and 2% of PM10 mass and HULIS, respectively (Claeys et al. 2012; Kitanovski et al. 2012, 2020; Kahnt et al. 2013; Caumo et al. 2016). NPs and NSAs are proven to have adverse effects on human health (estrogenic activity, carcinogenicity, cataract; Karim and Gupta 2001; Brüning et al. 2002; Harrison et al. 2005; Grundlingh et al. 2011; Kovacic and Somanathan 2014), while little is known about the toxicology of NCs. NMAHs may redox cycle in epithelial lung fluid and be a source of reactive oxygen species (ROS) in the lungs.
Organic chemicals in ambient PM contribute significantly to air pollution and its adverse health effects (Lewtas 1993; Jones 1999; Shiraiwa et al. 2017). Extracts of ambient wood burning aerosol induce mutagenicity and intracellular production of ROS more than road traffic aerosol (Velali et al. 2019). Polar fractions of organic PM extracts show higher toxicities than apolar ones (Nováková et al. 2020). The complete pollutant mass in the air may not be bioaccessible upon inhalation as the dissolution of the substance in the epithelial lung lining fluid (LLF) is a prerequisite for biological activity. However, this prerequisite is not needed, when the substances are carried by ultrafine particles which may penetrate membranes completely (Oberdörster et al. 2004; Li et al. 2017). Unlike heavy metals in PM (Wiseman and Zereini 2014; Wiseman 2015; Kastury et al. 2017; Polezer et al. 2019), the organic matter (OM) fraction of PM that is potentially soluble in LLF has hardly been studied. The most common approach for in vitro assessment of the bioaccessibility of PM chemicals in LLF is by determining the fraction of the total concentration of a chemical leached from PM deposited filters immersed in simulated LLFs, under controlled conditions (Wiseman 2015). The two most commonly used simulated LLFs are artificial lysosomal fluid (ALF; Colombo et al. 2008; Wiseman 2015) and Gamble’s solution (Marques et al. 2011; Wiseman 2015). ALF mimics the chemical environment around inhaled particles after being phagocytized by lung alveolar and interstitial macrophages. It is an acidic aqueous electrolyte without lipids, pH 4.5 (Table S1). Gamble’s solution is the most common simulated LLF and represents the interstitial fluid in the lung. It is a neutral aqueous electrolyte without lipids, proteins and antioxidants, pH 7.4 (Table S1). The bioaccessible fraction of a chemical in PM is calculated as fbio_p = cp LLF/cp MeOH × 100 (%), where cp LLF is the leached concentration in LLF and cp MeOH is the total concentration (from extraction in methanol) of the substance in PM samples used for leaching.
The aim of this present study was to determine levels and mass size distributions of NMAHs in the atmospheric PM collected at two urban locations in the Czech Republic. Inhalation bioaccessibility of semivolatile organic compounds so far has been mostly focusing on PAHs (Wei et al. 2018). For the first time, we quantify the inhalation bioaccessibility of NMAHs in PM. PAHs’ and nitro- and oxy-PAHs’ abundances and bioaccessibility in the same PM samples (Lammel et al. 2020a, b), as well as simultaneously in the gas phase (Lammel et al. 2020a), are presented in companion papers. Toxicities of these PM samples, as well as the mixture toxicity of the substance classes addressed (reconstituted mixtures), are published elsewhere (Nováková et al. 2020).
Materials and methods
Sampling sites
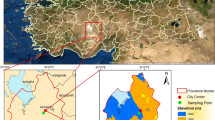
Air samples were collected at two urban and one rural site in the Czech Republic, Kladno-Švermov (50° 10′ 01″ N/14° 06′ 15″E) during 10–14 February 2016 and Ostrava-Přivoz (49°51′23″N/18°16′11″E) during 15–27 February and 5–17 September 2016, respectively (Fig. S1). In Kladno, an industrial town (≈ 70,000 inhabitants), a heat plant but no major industries were working during the campaign. The Ostrava site is located quite central in the industrial area (≈ 500,000 inhabitants). It is a station of the Czech Hydrometeorological Institute (CHMI). A major cokery with 200 furnaces, a major metallurgical plant, a waste burner and other industries are within 3 km from the site. Ostrava is a hot spot of air pollution in Europe (Pokorná et al. 2015, 2016; Kozáková et al. 2019). For example, abundance of polycyclic aromatic hydrocarbons (PAHs) is high in Ostrava and the biological effects of PM are evident, in particular during winter time (Líbalová et al. 2012; Šram et al. 2013; Topinka et al. 2015; Pokorná et al. 2015; Leoni et al. 2016).
Sampling
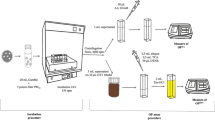
Particulate and gas phase samples were collected side by side by a high-volume air sampler Digitel DH77 (Digitel, Hegnau, Switzerland) and a high-volume 6-stage slot impactor Baghirra HV-100P (Baghirra, Prague). All samplers had PM10 inlets. Only the particulate phase samples were used for this study, i.e. 6 and 12 Digitel samples collected at Kladno and Ostrava winter campaigns, respectively, and 1, 3 and 3 impactor samples collected at Kladno and Ostrava winter and summer campaigns, respectively. The Digitel sampler was equipped with a quartz fibre filter (QFF, Whatman, Little Chalfont, UK), and the Baghirra sampler equipped with a multi-stage cascade impactor (Tisch Environmental Inc., Cleves, USA, series 230, model 235) with five impactor stages, corresponding to 10–7.2, 7.2–3, 3–1.5, 1.5–0.95 and 0.95–0.49 μm of aerodynamic particle size, D, (spaced roughly equal ΔlogD) and a backup filter collecting particles < 0.49 μm. In the impactor, PM was collected on a slotted quartz fibre filter (QFFs, TE-230-QZ, Tisch Environmental Inc., Cleves, USA, 14.3 × 13.7 cm) and the backup filter was a QFF (Whatman). The samplers were operated at constant flow rates of ≈ 29 (Digitel, 24 h sampling) and ≈ 68 m3 h−1 (Baghirra, 96-h sampling).
Filter samples were kept on-site and during transport cool (≈ 0 °C), then stored at temperatures below − 18 °C.
Leaching of NMAHs in simulated lung fluids and chemical analysis
Two LLFs were used, i.e. artificial lysosomal fluid (ALF; Colombo et al. 2008) and Gamble’s solution (Marques et al. 2011). Their compositions are given in the supplementary material (SM) (Table S1). The bioaccessible fractions of NMAHs in PM3 (fbio) were obtained by leaching the slotted and backup PM deposited QFFs with particles < 3 μm in 20 mL of simulated LLF by shaking (60 revolutions min−1) in a 100-mL flask during 24 h in an incubator at 37 °C, in the dark. Dependent on NMAH load, 1.5-cm2 cuts up to one strip (out of 10 strips of length 12 cm) of each slotted QFF were leached, while 1.5–20-cm2 cuts were leached from backup QFFs. The leachates were filtered through 0.45-μm cellulose acetate membrane, acidified with formic acid (1.0 mL 98–100% formic acid per 20 mL leachate), spiked with 4-nitrophenol-d4 (internal standard (IS); spiked mass 100 ng) and loaded on solid-phase extraction disks (SPE disks; BakerBond SPEEDISK DVB H2Ophilic, J.T. Baker). Targeted compounds were eluted from SPE disks sequentially with methanolic solution of EDTA (3.4 nmol mL−1) and a mixture of methanolic solution of EDTA (3.4 nmol mL−1) and acetonitrile (1:1). The obtained extract was concentrated to 0.5 mL using a TurboVap II (bath temperature, 40 °C; nitrogen gas pressure, 15 psi; Biotage, Uppsala, Sweden). The concentrated extract was filtered through a 0.2-μm PTFE syringe filter (4 mm, Whatman; GE Healthcare, Little Chalfont, UK) into a 2-mL vial and was evaporated to near dryness under the gentle stream of nitrogen (99.999%; Westfalen AG, Münster, Germany). Finally, the extract was dissolved in methanol/water mixture (3/7, v/v) containing 5 mM ammonium formate buffer pH 3 and 400 μM EDTA for LC/MS analysis.
The SPE recoveries of NMAHs were 94–101% in methanol extracts, 95–105% and 95–104% from ALF and Gamble’s solution leachates, respectively (Table S3). The results were not recovery corrected.
The determination of NMAHs in the PM filter samples was done using a validated analytical procedure (Kitanovski et al. 2012, 2020) with small modifications. In short, a 1.5-cm2 section of the filter was spiked with 4-nitrophenol-d4 (IS; spiked mass, 100 ng) and extracted three times (5 min each) with 10 mL methanolic solution of EDTA (3.4 nmol mL−1) in an ultrasonic bath. The combined extracts were concentrated, filtered, dried and re-dissolved for LC/MS analysis as described above for SPE extracts.
The targeted NMAHs, i.e. 2 NSAs, 4 NCs and 6 NPs (listed in Table S2 together with main physicochemical properties), were determined using an Agilent 1200 Series HPLC system (Agilent Technologies, Waldbronn, Germany) coupled to an Agilent 6130B single quadrupole mass spectrometer equipped with an electrospray ionisation (ESI) source (Kitanovski et al. 2020). Atlantis T3 column (150 mm × 2.1 mm i.d., 3-μm particles size; Waters, Milford, USA), connected to an Atlantis T3 VanGuard pre-column (5 mm × 2.1 mm i.d., 3-μm particles size; Waters), was used for the separation of the targeted analytes. NMAHs were eluted isocratically using a mobile phase consisted of methanol/tetrahydrofuran/water (30/15/55, v/v/v) mixture containing 5 mM ammonium formate buffer pH 3 at a flow rate of 0.2 mL min−1. The column temperature and injection volume were 30 °C and 10 μL, respectively (Kitanovski et al. 2012). For the detection and quantification of NMAHs, the mass spectrometer was operated in single ion monitoring (SIM) and negative ESI mode. The optimised ESI-MS parameters were as follows: 1000 V for the ESI capillary voltage, 30 psig for the nebulizer pressure and 12 L min−1 and 340 °C for the drying gas flow and temperature, respectively. High-purity nitrogen was used as a nebulizer and drying gas. 3-Methyl-4-nitrocatechol (3-M-4-NC) concentrations were calculated based on the calibration curve of 4-methyl-5-nitrocatechol (4-M-5-NC) due to the lack of a reference standard for 3-M-4-NC and its structural similarity to 4-M-5-NC. LC/MSD ChemStation (Agilent Technologies) was used for data acquisition and analysis.
Field blanks (n = 3) were prepared during sample collection by mounting the pre-baked filters on the sampler without switching it on. These filters were subsequently retrieved and processed along with the rest of the samples. The mean of two or three field blank values was subtracted from the sample values (in both methanol extracts and leachates). Values below the mean + 3 standard deviations of the field blank values were considered to be below the limit of quantification (<LOQ). LOQs for the various campaigns are listed in Table S4. Heavy metal content, aerosol number and mass size distributions (MSDs), meteorological and trace gases were also covered by respective methods, described in the supplementary material (SM) (S1.4).
Results and discussion
Concentration levels and mass size distributions
The levels of the targeted substance classes in PM10 are listed in Table 1, and the time series are shown in Fig. S2. With PM2.5 ranging 15–34 μg m−3 (Table 1), the sites were considerably polluted. The pollution by heavy metals in Ostrava air was found very high, independently of season (Table 1; Fig. S3) and must be seen in the context of the local metallurgical industries and coal production and burning (Pokorná et al. 2015; Vossler et al. 2015). The pollution at the urban sites was less reflected by the levels of the secondary inorganic aerosol (SO42−, NO3−, NH4+), because these are regionally distributed pollutants, exhibiting a low urban-to-rural gradient (Lammel et al. 2003).
The NMAH levels at the Kladno and Ostrava (winter) sites corresponded to 2.6 and 3.1% of the WSOC, respectively, and 6.3 and 7.3% of the HULIS-C, respectively (Voliotis et al. 2017). NMAHs were dominated by 4-NC and MNCs (Fig. S4a). The patterns in PM1 and PM10 are rather similar unlike typical for many other aerosol constituents (Putaud et al. 2010).
Mass size distributions of NMAHs are shown in Fig. 1 and S6. PM1 accounts for 80–90% of NCs, 70–80% of NSAs (as well as the NMAHs in total) and 40–60% of NPs. For all NMAH substance classes, the significance of the smallest size, PM0.49, was higher in summer than that in winter (in Ostrava). In contrast, the significance of a super-μm mode (3–7 μm) of NP and NSA MSDs decreased in summer, completely in the case of NSAs. A high fraction of NPs, 30–50%, was associated with the coarse fraction (PM10–PM3) in winter (Fig. 1). These results are in agreement with previous reports from other urban sites in central and southern Europe (Kitanovski et al. 2020) and China (Li et al. 2016). The aerosol number size distributions (characterised in Fig. S5) indicated close combustion sources and are consistent with the possible influence of wood burning. The MSDs peaking in the sub-micrometre size range highlight the significance of NMAHs’ inhalation exposure of the deep lung (Kitanovski et al. 2020), similar to other aromatic combustion byproducts like the parent PAHs (Ringuet et al. 2012) and polychlorinated dibenzodioxins and -furans (Zhang et al. 2016).
Bioaccessibility
The lowermost 4 impactor stage filters of the campaigns at Kladno (1 winter-time sample set) and Ostrava (3 winter- and 3 summer-time sample sets), encompassing PM3, were leached in ALF. Only one sample set encompassing PM3 per location and season (3 sample sets in total) was leached in Gamble’s solution (GS; Table 2; Table S5; Fig. 2).
Fractions (%) of bioaccessible particulate mass of individual NMAHs and 7 oxygenated polycyclic aromatic hydrocarbons (OPAHs; Lammel et al. 2020b) varying with KOW, as addressed by simulated epithelial lung fluid ALF in PM3 samples collected in Ostrava winter. 3-NSA, 3-nitrosalicylic acid; 3-M-4-NP, 3-methyl-4-nitrophenol; 2-M-4-NP, 2-methyl-4-nitrophenol; 3-M-5-NC, 3-methyl-5-nitrocatechol; 4-M-5-NC, 4-methyl-5-nitrocatechol; 4-NP, 4-nitrophenol; 4-NC, 4-nitrocatechol; OFLN, 9-fluorenone; O2ANT, 9,10-anthraquinone; O2NAP, 1,4-naphthoquinone; O2NAC, 5,12-naphthacenequinone; O2BAA, benzanthracene-7,12-dione; BAN, benzanthrone; BaOFLN, benz(a)fluorenone
Using ALF, more than half of NMAH mass was found bioaccessible in winter, and almost complete, 94%, in summer (Table 2a). This could be related to a higher content of hydrophobic substances in PM in winter. In central Europe, fossil fuel combustion byproducts, in particular PAHs, are much higher concentrated in winter, also in urban air, and also in Ostrava (Lammel et al. 2010; CHMI 2013; Vossler et al. 2015). The difference of fbio_p found when using ALF across the campaigns (Table 2) was not pronounced as compared with when using GS to leach samples (insignificant differences for p < 0.05, t test). Often lower fbio_p was found for all NMAH species when using the neutral GS than when using the acidic ALF (Table 2 and Table S5a; note that due to less samples leached by GS than ALF, directly comparable fbio_p data are given in Table 2, but not in Table S5), but also the opposite was found (Kladno sample, Table 2, Table S5b). NSAs were almost completely bioaccessible, i.e. fbio_p≈100% in both LLFs. Bioaccessible fractions > 100% most likely reflect leaching procedure artefacts. They are more pronounced for NSAs and NCs when leached in ALF (Table S5). Therefore, we investigated the stability of NMAHs during the leaching procedure by spiking the LLFs with NMAH standard mix and carrying out the usual 24-h leaching. The results from stability study (Table S7) showed > 100% recoveries for NSAs in both LLFs, but usually < 100% recoveries for NCs. Neither NCs nor methylnitrophenols (MNPs) or dinitrophenols (DNPs) were found more stable in ALF than in GS (not significant, p < 0.05, t test). With a pKa (acidity constant) of 6.78 at 35 °C (Gelb et al. 1989), the majority of 4-NC (but also MNC) molecules will be deprotonated in GS (pH 7.4) at 37 °C. In deprotonated form, NCs are more susceptible to oxidation (e.g. by the dissolved oxygen in LLFs) and formation of nitrated 1,2-benzoquinones, which could not be measured by the analytical method employed here. In ALF at pH 4.5, NCs are in neutral form and more stable, hence their higher recoveries from ALF. This could also explain their higher fbio_p in ALF (significant at the p < 0.05 level, t test; Table S5). For MNPs and DNPs, however, their lower stability in ALF is unexplained, having in mind their pKa values around 7.3 and 4.0, respectively (Schwarzenbach et al. 1988), as well as their high recoveries during the SPE clean-up after the leaching process (Table S3). Only for summer samples from Ostrava, the NMAHs’ bioaccessible fractions in ALF are much higher than 100% (range, 99–187%; Table S5b), suggesting the possible aqueous-phase formation of NMAHs from their precursors in the PM during the leaching process (positive artefact) under mild acidic conditions (pH 4.5; Kroflič et al. 2018). This hypothesis is supported by the high levels of PM10 (PM2.5), NOx and Fe (Fig. S3) measured during the summer sampling campaign (Table 1a) which could facilitate the oxidation and nitration of NMAH precursors. Interestingly, for the same sample sets, very low bioaccessibility in GS was observed for NCs (range, 9–77%; Table S5b) that cannot be solely explained by the NC stability results (50–99%; Table S7). Due to high Fe content in samples, NCs could partly exist as monocomplexes of Fe3+ and enhance the production of reactive species by Fenton or Fenton-like systems (Salgado et al. 2017). During these processes, NCs can be oxidised or degraded by the formed reactive species, thus diminishing their leached concentrations (negative artefact), as well as their measured bioaccessible fractions.
For both LLFs, fbio_p was found independent of particle size, i.e. do not differ significantly between sub-micrometre particles and the PM3 size fraction (p < 0.05, t test; Table 2). This is also reflected as similar (statistically not different, p < 0.05) values for PM1/PM3 found for PM methanol extracts as for LLFs (Table S6).
The range of physicochemical properties of NMAHs is not large, 2 and 1 order of magnitude for water solubility, s, and KOW, respectively (listed in Table S2). The respective data for Ostrava winter (Table S5) are shown together with data for 7 oxygenated polycyclic aromatic hydrocarbons (OPAHs; Lammel et al. 2020b), hence, s and KOW across the two substance classes ranging 5 and 4 orders of magnitude, respectively (Fig. 2, Fig. S7). The bioaccessible fractions of NMAHs, fbio p, were similar in winter and summer (Fig. S7), reflecting that ambient aerosol chemical composition in source areas (anthropogenic sources) is subject to little seasonal variation (Putaud et al. 2010). It decreased with the compound’s increasing KOW (Fig. 2) and decreasing water solubility (Fig. S7). Bioaccessibility may be negligible for lipophilic substances (i.e. log KOW > 4.5). A lack of a clear trend in Fig. S7 reflects the aqueous electrolyte nature of the LLFs.
The MSDs of the bioaccessible fractions were only slightly shifted against the MSDs of the PM methanol extracts. For example, for GS, the bioaccessible sub-micrometre mass fraction in PM3, i.e. PM1/PM3, deviated typically only within 2% from the total sub-micrometre mass fraction in PM3 (Table S6b), while for ALF these shifts were up to ≈ 10% (Table S6a), in the sense that the sub-micrometre fraction was less bioaccessible than the coarse size fraction. This is possibly related to a higher hydrophobicity of PM1 particles as compared with coarse PM. Hydrophobicity may limit the leachability of particles. Hydrophobicity was not determined, but more than 60% of EC and OC, which often represent hydrophobic constituents, were associated with the PM1 mass fraction, more than in coarse PM (cumulative MSDs, Fig. S5).
Conclusions and suggestions for research
Inhalation bioaccessibility of the nitrated monoaromatic pollutants in PM as operationally defined by leaching filter samples in simulated lung fluids was found very high for both an aqueous acidic (pH 4.5, ALF) and a neutral electrolyte (pH 7.4, Gamble’s solution). This emphasises the human inhalation exposure to polar constituents of particulate organic matter. Bioaccessibility of a given PM constituent will depend on not only the substance properties but also the aerosol matrix (e.g. its hydrophobicity). Here, a limited number of samples have been analysed. Among aerosol types, only urban aerosols, strongly influenced by fossil fuel burning sources (metallurgical industries and coal production and burning, road traffic; Lammel et al. 2020b) were covered. More such data should be gained from other aerosol types and extended to other organic pollutants, abundant in aerosols, such as polycyclic aromatic compounds. The determination of bioaccessibility based on leaching with simulated lung fluids may even be an underestimate, as ultrafine particles may penetrate through the membrane and thus deliver pollutants without dissolution in the lung fluid. On the other hand, the presence of false-positive (fbio p >> 100%) and false-negative artefacts (fbio p < 50%) during the in vitro tests of bioaccessibility should be avoided by (a) optimization of the duration of the tests (allowing less time for unwanted reactions to occur), (b) using degassed LLFs and performing the tests in inert atmosphere for analytes that could be easily oxidised (which is opposite to the real conditions in the lung) and (c) by using more realistic LLF models that contain lipids, proteins and antioxidants (e.g. Boisa et al. 2014). The presence of organic constituents and antioxidants in LLFs would serve as “buffer” for PM and potentially in situ formed ROS during the leaching procedure. Only the bioaccessible fraction of pollutants can become biologically effective, such as ROS active. While the reduction potential as an indicator for redox reactivity is available for a number of NMAHs such as nitrobenzenes (Uchimiya et al. 2010), determination of the oxidative potential (OP) of organic pollutants has so far been limited to quinones (Charrier and Anastasio 2012; Yu et al. 2018; Lammel et al. 2020b) and N-heterocycles (Dou et al. 2015). Finally, the inhalation exposure to the targeted NMAHs is in fact higher, because part of the NMAH mass will be distributed to the gas-phase of ambient aerosols, not considered in this study.
References
Boisa N, Elom N, Dean JR, Deary ME, Bird G, Entwistle JA (2014) Development and application of an inhalation bioaccessibility method (IBM) for lead in the PM10 size fraction of soil. Environ Int 70:132–142. https://doi.org/10.1016/j.envint.2014.05.021
Brüning T, Their R, Bolt H (2002) Nephrotoxicity and nephrocarcinogenicity of dinitrotoluene: new aspects to be considered. Rev Environ Health 17:163–172. https://doi.org/10.1515/REVEH.2002.17.3.163
Caumo SES, Claeys M, Maenhaut W, Vermeylen R, Shabnam B, Shalamzari MS, Vasconcellos PC (2016) Physicochemical characterization of winter PM10 aerosol impacted by sugarcane burning from São Paulo city. Atmos Environ 145:272–279. https://doi.org/10.1016/j.atmosenv.2016.09.046
Charrier JG, Anastasio C (2012) On dithiothreitol (DTT) as a measure of oxidative potential for ambient particles: evidence for the importance of soluble transition metals. Atmos Chem Phys 12:9321–9333
CHMI (2013) Graphic yearbook 2014. Czech Hydrometeorological Institute, Prague. URL: http://portal.chmi.cz/files/portal/docs/uoco/isko/grafroc/13groc/gr13e/V3_OKFM_GB.html. Accessed 30 Sept 2019
Chow KS, Hilda XHH, Yu JZ (2016) Quantification of nitroaromatic compounds in atmospheric fine particulate matter in Hong Kong over 3 years: field measurement evidence for secondary formation derived from biomass burning emissions. Environ Chem 13:665–673. https://doi.org/10.1071/EN15174
Claeys M, Vermeylen R, Yasmeen F, Gómez-González Y, Chi X, Maenhaut W, Mészáros T, Salma I (2012) Chemical characterisation of humic-like substances from urban, rural and tropical biomass burning environments using liquid chromatography with UV/vis photodiode array detection and electrospray ionisation mass spectrometry. Environ Chem 9:273–284. https://doi.org/10.1071/EN11163
Colombo C, Monhemius AJ, Plant JA (2008) Platinum, palladium and rhodium release from vehicle exhaust catalysts and road dust exposed to simulated lung fluids. Ecotox Environ Safety 71:722–730. https://doi.org/10.1016/j.ecoenv.2007.11.011
Dou J, Lin P, Kuang BY, Yu JZ (2015) Reactive oxygen species production mediated by humic-like substances in atmospheric aerosols: enhancement effects by pyridine, imidazole, and their derivatives. Environ Sci Technol 49:6457–6465
Finewax Z, de Gouw JA, Ziemann PJ (2018) Identification and quantification of 4-nitrocatechol formed from OH and NO3 radical-initiated reactions of catechol in air in the presence of NOx: implications for secondary organic aerosol formation from biomass burning. Environ Sci Technol 52:1981–1988. https://doi.org/10.1021/acs.est.7b05864
Frka S, Šala M, Kroflič A, Huš M, Čusak A, Grgić I (2016) Quantum chemical calculations resolved identification of methylnitrocatechols in atmospheric aerosols. Environ Sci Technol 50:5526–5535. https://doi.org/10.1021/acs.est.6b00823
Gelb RI, Laufer DA, Schwartz LM, Wairimu K (1989) Acid dissociation of aqueous 4-nitrocatechol. J Chem Eng Data 34:82–83. https://doi.org/10.1021/je00055a023
Graber ER, Rudich Y (2006) Atmospheric HULIS: How humic-like are they ? A comprehensive and critical review. Atmos Chem Phys 6:729–753. https://doi.org/10.5194/acp-6-729-2006
Grundlingh J, Paul I, el Zanfaly M, Wood D (2011) 2,4-Dinitrophenol (DNP): a weight loss agent with significant acute toxicity and risk of death. J Med Toxicol 7:205–212. https://doi.org/10.1007/s13181-011-0162-6
Harrison MAJ, Barra S, Borghesi D, Vione D, Arsene C, Olariu RI (2005) Nitrated phenols in the atmosphere: a review. Atmos Environ 39:231–248. https://doi.org/10.1016/j.atmosenv.2004.09.044
Iinuma Y, Brüggemann E, Gnauk T, Müller K, Andreae MO, Helas G, Parmar R, Herrmann H (2007) Source characterization of biomass burning particles: the combustion of selected European conifers, African hardwood, savanna grass, and German and Indonesian peat. J Geophys Res 112:D8209. https://doi.org/10.1029/2006JD007120
Iinuma Y, Böge O, Gräfe R, Herrmann H (2010) Methyl-nitrocatechols: atmospheric tracer compounds for biomass burning secondary organic aerosols. Environ Sci Technol 44:8453–8459. https://doi.org/10.1021/es102938a
Inomata S, Fushimi A, Fujitani Y, Yamada H (2015) 4-Nitrophenol, 1-nitropyrene, and 9-nitroanthracene emissions in exhaust particles from diesel vehicles with different exhaust gas treatments. Atmos Environ 110:93–102. https://doi.org/10.1016/j.atmosenv.2015.03.043
Jones AP (1999) Indoor air quality and health. Atmos Environ 33:4535–4564. https://doi.org/10.1016/S1352-2310(99)00272-1
Kahnt A, Behrouzi S, Vermeylen R, Shalamzari MS, Vercauteren J, Roekens E, Claeys M, Maenhaut W (2013) One-year study of nitro-organic compounds and their relation to wood burning in PM10 aerosol from a rural site in Belgium. Atmos Environ 81:561–568. https://doi.org/10.1016/j.atmosenv.2013.09.041
Karim K, Gupta S (2001) Biotransformation of nitrophenols in upflow anaerobic sludge blanket reactors. Bioresource Technol 8:179–186. https://doi.org/10.1016/S0960-8524(01)00092-X
Kastury F, Smith E, Juhasz AL (2017) A critical review of approaches and limitations of inhalation bioaccessibility and bioaccessibility of metal(loid)s from ambient particulate matter or dust. Sci Total Environ 574:1054–1074. https://doi.org/10.1016/j.scitotenv.2016.09.056
Kelly JL, Michelangeli DV, Makar PA, Hastie DR, Mozurkewich M, Auld J (2010) Aerosol speciation and mass prediction from toluene oxidation under high NOx conditions. Atmos Environ 44:361–369. https://doi.org/10.1016/j.atmosenv.2009.10.035
Kitanovski Z, Grgić I, Vermeylen R, Claeys M, Maenhaut W (2012) Liquid chromatography tandem mass spectrometry method for characterization of monoaromatic nitro-compounds in atmospheric particulate matter. J Chromatogr A 1268:35–43. https://doi.org/10.1016/j.chroma.2012.10.021
Kitanovski Z, Shahpoury P, Samara C, Voliotis A, Lammel G (2020) Composition and mass size distribution of nitrated and oxygenated aromatic compounds in ambient particulate matter from southern and central Europe – implications for the origin. Atmos Chem Phys 20:2471–2487. https://doi.org/10.5194/acp-20-2471-2020
Kovacic P, Somanathan R (2014) Nitroaromatic compounds: environmental toxicity, carcinogenicity, mutagenicity, therapy and mechanism. J Appl Toxicol 34:810–824. https://doi.org/10.1002/jat.2980
Kozáková J, Pokorná P, Vodička P, Ondráčková L, Ondráček J, Křůmal K, Mikuška P, Hovorka J, Moravec P, Schwarz J (2019) The influence of local emissions and regional air pollution transport on a European air pollution hot spot. Environ Sci Pollut Res 26:1675–1692. https://doi.org/10.1007/s11356-018-3670-y
Kroflič A, Grilc M, Grgić I (2015) Unraveling pathways of guaiacol nitration in atmospheric waters: Nitrite, a source of reactive nitronium ion in the atmosphere. Environ Sci Technol 49:9150–9158. https://doi.org/10.1021/acs.est.5b01811
Kroflič A, Huš M, Grilc M, Grgić I (2018) Underappreciated and complex role of nitrous acid in aromatic nitration under mild environmental conditions: the case of activated methoxyphenols. Environ Sci Technol 52:13756–13765. https://doi.org/10.1021/acs.est.8b01903
Lammel G, Brüggemann E, Gnauk T, Müller K, Neusüß C, Röhrl A (2003) A new method to study aerosol source contributions along the tracts of air parcels and its application to the near-ground level aerosol chemical composition in central Europe. J Aerosol Sci 34:1–25. https://doi.org/10.1016/S0021-8502(02)00134-9
Lammel G, Novák J, Landlová L, Dvorská A, Klánová J, Čupr P, Kohoutek J, Reimer E, Škrdlíková L (2010) Sources and distributions of polycyclic aromatic hydrocarbons and toxicity of polluted atmosphere aerosols. In: Zereini F, Wiseman CLS (eds) Urban Airborne Particulate Matter: Origins, Chemistry, Fate and Health Impacts. Springer, Berlin, pp 39–62
Lammel G, Shahpoury P, Berkemeier T, Hilscherová K, Kitanovski Z, Kukučka P, Kyprianou M, Novák J, Pöschl U, Příbylová P, Prokeš R, Sáňka O, Stephanou E., Wietzoreck M (2020a) Inhalation exposure to PAHs dominated by the gaseous mass fraction even if minute, submitted manuscript
Lammel G, Kitanovski Z, Kukučka P, Novák J, Arangio A, Codling GP, Filippi A, Hovorka J, Kuta J, Leoni C, Příbylová P, Prokeš R, Sáňka O, Shahpoury P, Tong HJ, Wietzoreck M (2020b) Levels, phase partitioning, mass size distributions and bioaccessibility of oxygenated and nitrated polycyclic aromatic hydrocarbons (OPAHs, NPAHs) in ambient air. Environ Sci Technol 54:2615–2625. https://doi.org/10.1021/acs.est.9b06820
Laskin A, Laskin J, Nizkorodov SA (2015) Chemistry of atmospheric brown carbon. Chem Rev 115:4335–4382. https://doi.org/10.1021/cr5006167
Leoni C, Hovorka J, Dočekalová V, Cajthaml T, Marvanová S (2016) Source impact determination using airborne and ground measurements of industrial plumes. Environ Sci Technol 50:9881–9888. https://doi.org/10.1021/acs.est.6b02304
Lewtas J (1993) Complex mixtures of air pollutants: characterizing the cancer risk of polycyclic organic matter. Environ Health Persp 100:211–218. https://doi.org/10.2307/3431527
Li XN, Jiang L, Hoa LP, Lyu Y, Xu TT, Yang X, Iinuma Y, Chen JM, Herrmann H (2016) Size distribution of particle-phase sugar and nitrophenol tracers during severe urban haze episodes in Shanghai. Atmos Environ 145:115–127. https://doi.org/10.1016/j.atmosenv.2016.09.030
Li XY, Hao L, Liu YH, Chen CY, Pai VJ, Kang JX (2017) Protection against fine particle-induced pulmonary and systemic inflammation by omega-3 polyunsaturated fatty acids. Biochim Biophys Acta 1861:577–584. https://doi.org/10.1016/j.bbagen.2016.12.018
Líbalová H, Uhlířová K, Kléma J, Machala M, Šrám RJ, Ciganek M, Topinka J (2012) Global gene expression changes in human embryonic lung fibroblasts induced by organic extracts from respirable air particles. Part. Fibre Toxicol 9:1–16. https://doi.org/10.1186/1743-8977-9-1
Marques MRC, Loebenberg R, Almukainzi M (2011) Simulated biological fluids with possible application in dissolution testing. Dissolution Technol 15:–28. https://doi.org/10.14227/DT180311P15
Nováková Z, Novák J, Kitanovski Z, Kukučka P, Přibylová P, Prokeš R, Smutná M, Wietzoreck M, Lammel G, Hilscherová K (2020) Toxic potentials of particulate and gaseous air pollutant mixtures and the role of PAHs and their derivatives. Environ Int 139:105634. https://doi.org/10.1016/j.envint.2020.105634
Oberdörster G, Sharp Z, Atudorei V, Elder A, Gelein R, Kreyling W, Cox C (2004) Translocation of inhaled ultrafine particles to the brain. Inhal Toxicol 16:437–445. https://doi.org/10.1080/08958370490439597
Pokorná P, Hovorka J, Klán M, Hopke PK (2015) Source apportionment of size resolved particulate matter at a European air pollution hot spot. Sci Total Environ 502:172–183. https://doi.org/10.1016/j.scitotenv.2014.09.021
Pokorná P, Hovorka J, Hopke PK (2016) Elemental composition and source identification of very fine aerosol particles in a European air pollution hot-spot. Air Pollut Res 7:671–679. https://doi.org/10.1016/j.apr.2016.03.001
Polezer G, Oliveira A, Potgieter-Vermaak SP, Godoi AFL, de Souza RAF, Yamamoto CI, Andreoli RV, Medeiros AS, Machado CMD, dos Santos EO, de André PA, Pauliquevis T, Saldiva PHN, Martin ST, Godoi RHM (2019) The influence that different urban development models has on PM2.5 elemental and bioaccessible profiles. Sci Rep 9:14846. https://doi.org/10.1038/s41598-019-51340-4
Putaud JP, van Dingenen R, Alastuey A, Bauer H, Birmili W, Cyrys J, Flentje H, Fuzzi S, Gehrig R, Hansson HC, Harrison RM, Herrmann H, Hitzenberger R, Hüglin C, Jones AM, Kasper-Giebl A, Kiss G, Kousa A, Kuhlbusch TAJ, Löschau G, Maenhaut W, Molnar A, Moreno T, Pekkanen J, Perrino C, Pitz M, Puxbaum H, Querol X, Rodriguez S, Salma I, Schwarz J, Smolik J, Schneider J, Spindler G, ten Brink H, Tursic J, Viana M, Wiedensohler A, Raes F (2010) A European aerosol phenomenology – 3: physical and chemical characteristics of particulate matter from 60 rural, urban, and kerbside sites across Europe. Atmos Environ 44:1308–1320. https://doi.org/10.1016/j.atmosenv.2009.12.011
Ramaswamy S, Nakayama T, Imamura T, Morino Y, Kajii Y, Sato K (2019) Investigation of dark condition nitrate radical- and ozone-initiated aging of toluene secondary organic aerosol: importance of nitrate radical reactions with phenolic products. Atmos Environ 219:117049. https://doi.org/10.1016/j.atmosenv.2019.117049
Ringuet J, Leoz-Garziandia E, Villenave E, Albinet A (2012) Particle size distribution of nitrated and oxygenated polycyclic aromatic hydrocarbons (NPAHs and OPAHs) on traffic and suburban sites of a European megacity: Paris (France). Atmos Chem Phys 12:8877–8887. https://doi.org/10.5194/acp-12-8877-2012
Salgado P, Melin V, Durán Y, Mansilla H, Contreras D (2017) The reactivity and reaction pathway of Fenton reactions driven by substituted 1,2-dihydroxybenzenes. Environ Sci Technol 51:3687–3693. https://doi.org/10.1021/acs.est.6b05388
Schwarzenbach RP, Stierli R, Folsom BR, Zeyer J (1988) Compound properties relevant for assessing the environmental partitioning of nitrophenols. Environ Sci Technol 22:83–92. https://doi.org/10.1021/es00166a009
Shiraiwa M, Ueda K, Pozzer A, Lammel G, Kampf CJ, Fushimi A, Enami S, Arangio AM, Fröhlich-Nowoisky J, Fujitani Y, Furuyama A, Lakey PSJ, Lelieveld J, Lucas K, Morino Y, Pöschl U, Takahama S, Takami A, Tong HJ, Weber B, Yoshino A, Sato K (2017) Aerosol health effects from molecular to global scales. Environ Sci Technol 51:13545–13567. https://doi.org/10.1021/acs.est.7b04417
Šram RJ, Dostal M, Libalová H, Rossner P, Rossnerová A, Svečová V, Topinka T, Bartonová A (2013) The European hot spot of B[a]P and PM2.5 exposure - the Ostrava region, Czech Republic: Health Research Results. ISRN Publ Health #416071. https://doi.org/10.1155/2013/416701
Teich M, van Pinxteren D, Wang M, Kecorius S, Wang Z, Müller T, Močnik G, Herrmann H (2017) Contributions of nitrated aromatic compounds to the light absorption of water-soluble and particulate brown carbon in different atmospheric environments in Germany and China. Atmos Chem Phys 17:1653–1672. https://doi.org/10.5194/acp-17-1653-2017
Topinka J, Rossner P, Milcová A, Schmuczerová J, Pěnčíková K, Rossnerová A, Ambroz A, Štolcpartová J, Bendl J, Hovorka J, Machala M (2015) Day-to-day variability of toxic events induced by organic compounds bound to size segregated atmospheric aerosol. Environ Pollut 202:135–145. https://doi.org/10.1016/j.envpol.2015.03.024
Uchimiya M, Gorb L, Isayev O, Qasim MM, Leszczynski J (2010) One-electron standard reduction potentials of nitroaromatic and cyclic nitramine explosives. Environ Pollut 158:3048–3053. https://doi.org/10.1016/j.envpol.2010.06.033
Velali E, Pantazaki A, Besis A, Choli-Papadopoulou T, Samara C (2019) Oxidative stress, DNA damage, and mutagenicity induced by the extractable organic matter of airborne particulates on bacterial models. Regul Toxicol Pharmacol 104:59–73. https://doi.org/10.1016/j.yrtph.2019.03.004
Voliotis A, Prokeš R, Lammel G, Samara C (2017) New insights on humic-like substances associated with urban aerosols from central and southern Europe: size-resolved chemical characterization and optical properties. Atmos Environ 166:286–299. https://doi.org/10.1016/j.atmosenv.2017.07.024
Vossler T, Černikovsky L, Novák J, Plachá H, Krejci B, Nikolová I, Chalupníčková E, Williams R (2015) An investigation of local and regional sources of fine particulate matter in Ostrava, the Czech Republic. Atmos Pollut Res 6:454–463. https://doi.org/10.5094/APR.2015.050
Wang L, Wang X, Gu R, Wang H, Yao L, Wen L, Zhu F, Wang W, Xue L, Yang L, Lu K, Chen J, Wang T, Zhang Y, Wang W (2018) Observations of fine particulate nitrated phenols in four sites in northern China: concentrations, source apportionment, and secondary formation. Atmos Chem Phys 18:4349–4359. https://doi.org/10.5194/acp-18-4349-2018
Wang Y, Hu M, Wang Y, Zheng J, Shang D, Yang Y, Liu Y, Li X, Tang R, Zhu W, Du Z, Wu Y, Guo S, Wu Z, Lou S, Hallquist M, Yu J (2019) The formation of nitro-aromatic compounds under high NOx-anthropogenic VOCs dominated atmosphere in summer in Beijing, China. Atmos Chem Phys 19:7649–7665. https://doi.org/10.5194/acp-19-7649-2019
Wei WJ, Bonvallot N, Gustafsson Å, Raffy G, Glorennec P, Krais A, Ramalho O, le Bot B, Mandin C (2018) Bioaccessibility and bioaccessibility of environmental semi-volatile organic compounds via inhalation: a review of methods and models. Environ Int 113:202–213. https://doi.org/10.1016/j.envint.2018.01.024
Wiseman CLS (2015) Analytical methods for assessing metal bioaccessibility in airborne particulate matter: a scoping review. Anal Chim Acta 877:9–18. https://doi.org/10.1016/j.aca.2015.01.024
Wiseman CLS, Zereini F (2014) Characterizing metal(oid) solubility in airborne PM10, PM2.5 and PM1 in Frankfurt, Germany, using simulated lung fluids. Atmos Environ 89:282–289. https://doi.org/10.1016/j.atmosenv.2014.02.055
Xie MJ, Chen X, Hays MD, Lewandowski M, Offenberg J, Kleindienst TE, Holder AL (2017) Light absorption of secondary organic aerosol: composition and contribution of nitroaromatic compounds. Environ Sci Technol 51:11607–11616. https://doi.org/10.1021/acs.est.7b03263
Yu HR, Wie JL, Cheng YL, Subedi K, Verma V (2018) Synergistic and antagonistic interactions among the particulate matter components in generating reactive oxygen species based on the dithiothreitol assay. Environ Sci Technol 52:2261–2270
Zhang X, Zheng MH, Liu GR, Zhu QQ, Dong SJ, Zhang HX, Wang XK, Xiao K, Gao LR, Liu WB (2016) A comparison of the levels and particle size distribution of lower chlorinated dioxin/furans (mono- to tri-chlorinated homologues) with those of tetra- to octa-chlorinated homologues in atmospheric samples. Chemosphere 151:55–58. https://doi.org/10.1016/j.chemosphere.2016.02.059
Acknowledgements
Open access funding provided by Projekt DEAL. We thank Libor Černikovský, Blanka Krejci, Jana Schovánková, Pavel Smolík (Czech Hydrometeorological Institute) and Rostislav Červenka (Masaryk University) for meteorological and trace gas data and carbon fraction analysis. We also thank Sergej Grebenyuk and Anna Honcza for the help during sample preparation. This research was supported by the Max Planck Society, by the Czech Science Foundation (503/16/11537S, 503/12/G147) and by the ACTRIS-CZ (CZ.02.1.01/0.0/0.0/16_013/0001315) and RECETOX (LM2018121) Research Infrastructures funded by the Ministry of Education, Youth and Sports of the Czech Republic.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 860 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kitanovski, Z., Hovorka, J., Kuta, J. et al. Nitrated monoaromatic hydrocarbons (nitrophenols, nitrocatechols, nitrosalicylic acids) in ambient air: levels, mass size distributions and inhalation bioaccessibility. Environ Sci Pollut Res 28, 59131–59140 (2021). https://doi.org/10.1007/s11356-020-09540-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09540-3