Abstract
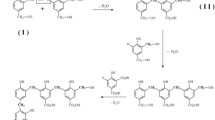
The formation of highly graphitic phenolic resin chars (GPFCs) during catalytic carbonization at relatively low reaction temperature (1200–1600 °C) using novel Ni-Zn-B alloy catalyst with small amount of addition (5–15%) was systematically studied. Only two kinds of graphites (turbostratic graphite and ordered graphite) can be found in GPFCs after catalytic carbonization with Ni-Zn-B and their proportions were changed with reaction conditions. When Ni-Zn-B was involved at 1200–1600 °C, the phenolic resin char was fully transformed to be graphite, and ordered graphite content increased to 28.42% at 1400 °C, which was also almost twice of ordered graphite content in the char catalyzed by pure Ni. But the order graphite content would decrease due to sintering at higher reaction temperature. The addition of Zn and B can promote nickel-based alloy catalytic action by reducing melt point and accelerating graphitization respectively. It was also found that ordered graphite content could be used as a key evaluation parameter to directly reflect the quality of GPFCs based on detailed characteristics analysis. The model between three reaction conditions (reaction temperature, retention time, catalyst content) and ordered graphite content was built with artificial neural network (ANN), and the prediction accuracy of ANN was high up to 91.48%.






Similar content being viewed by others
References
Aci M, Mutlu A (2016) Artificial neural network approach for atomic coordinate prediction of carbon nanotubes. Appl Phys A Mater Sci Process 122:1–14. https://doi.org/10.1007/s00339-016-0153-1
Agar DA, Kwapinska M, Leahy JJ (2018) Pyrolysis of waste water sludge and composted organic fines from municipal solid waste: laboratory reactor characterisation and product distribution. Environ Sci Pollut Res 25:35874–35882. https://doi.org/10.1007/s11356-018-1463-y
Almeida JS (2002) Predictive non-linear modeling of complex data by artificial neural networks. Curr.Opin.Biotech. 13:72–76. https://doi.org/10.1016/S0958-1669(02)00288-4
Bharathy S, Gnanasikamani B, Lawrence KR (2019) Investigation on the use of plastics pyrolysis oil as alternate fuel in a direct injection diesel engine with titanium oxide nanoadditive. Environ Sci Pollut Res 26:10319–10332. https://doi.org/10.1007/s11356-019-04293-0
Bukkarapu K, Siva gangadhar D, Jyothi Y, Kanasani P (2018) Management, conversion and utilization of waste plastic as a source if sustainable energy to run automotive: a review. Energ Sourc A 40:1681–1692. https://doi.org/10.1080/15567036.2018.1486898
Charon E, Rouzaud J-N, Aléon J (2014) Graphitization at low temperature (600-1200 °C) in the presence of iron implications in planetology. Carbon 66:178–190. https://doi.org/10.1016/j.carbon.2013.08.056
Chen Z, Ren W, Gao L, Liu B, Pei S, Cheng H (2011) Three-dimensional flexible and conductive interconnected graphene networks grown by chemical vapour deposition. Nat Mater 10:424–428. https://doi.org/10.1038/nmat3001
Chen R, Li Q, Xu X, Zhang D (2019) Comparative pyrolysis characteristics of representative commercial thermosetting plastic waste in inert and oxygenous atmosphere. Fuel 246:212–221. https://doi.org/10.1016/j.fuel.2019.02.129
Derbyshire FJ, Presland AEB, Trimm DL (1972) The formation of graphite films by precipitation of carbon from nickel foils. Carbon 10:114–115. https://doi.org/10.1016/0008-6223(72)90017-6
Di J, Wang M, Zhu Z (2018) Experiment on the treatment of acid mine drainage with optimized biomedical stone particles by response surface methodology. Environ Sci Pollut Res 25:7978–7990. https://doi.org/10.1007/s11356-017-1135-3
Enemuoh E, EI-Gizawy AS (2003) Optimal neural network model for characterization of process-induced damage in drilling carbon fiber reinforced epoxy composites. Mach Sci Technol 7:389–400. https://doi.org/10.1081/MST-120025285
Feng Z, Li D, Wang L, Li S, Lu Q, Xing P et al (2019) In situ grown nanosheet Ni-Zn alloy on Ni foam for high performance hydrazine electrooxidation. Electrochim Acta 304:275–281. https://doi.org/10.1002/jctb.5943
Gong Y, Li D, Luo C, Fu Q, Pan C (2017) Highly porous graphitic biomass carbons as advanced electrode materials for supercapacitors. Green Chem 19:4132–4410. https://doi.org/10.1039/c7gc01681f
Haward M (2018) Plastic pollution of the world’s seas and oceans as a contemporary challenge in ocean governance. Nat Commun 9:1–3. https://doi.org/10.1038/s41467-018-03104-3
He C, Men G, Xu B, Cui J, Zhao J (2017) Phenolic resin-derived activated carbon-supported divalent metal as efficient adsorbents (M–C, M=Zn, Ni, or Cu) for dibenzothiophene removal. Environ Sci Pollut Res 24:782–794. https://doi.org/10.1007/s11356-016-7795-6
Honada H, Kobayashi K, Sugawara S (1968) X-ray characteristics of non-graphitizing-type carbon. Carbon 6:517–523. https://doi.org/10.1016/0008-6223(68)90091-2
Inomata K, Otake Y (2011) Formation and development of micropores in carbon prepared via catalytic carbonization of phenolic resin containing Fe or Ni compounds. Microporous Mesoporous Mater 143:60–65. https://doi.org/10.1016/j.micromeso.2011.02.005
Jambeck JR, Geyer R, Wilcox C, Siegler TR, Perryman M, Andrady A, Narayan R, Law KL (2015) Plastic waste inputs from land into the ocean. Science 347:768–771. https://doi.org/10.1126/science.1260352
Khokhlova GP, Barnakov CN, Malysheva VY, Popova AN, Ismagilov ZR (2015) Effect of heat treatment conditions on the catalytic graphitization of coal-tar pitch. Solid Fuel Chem 49:66–72. https://doi.org/10.3103/S0361521915020056
Kobayashi K, Sugawara S, Toyoda S, Honda H (1968) An X-ray diffraction study of phenol-formaldehyde resin carbons. Carbon 6:359–363. https://doi.org/10.1016/0008-6223(68)90030-4
Lebreton LCM, Zwet J, Damsteeg J, Slat B, Andrady A, Reisser J (2017) River plastic emissions to the world’s oceans. Nat Commun 8:1–10. https://doi.org/10.1038/ncomms15611
Li H, Yang S, Zhao W, Xu Z, Zhao S, Liu X (2016) Prediction of the physicochemical properties of woody biomass using linear prediction and artificial neural networks. Energy Sources, Part A 38:1569–1573. https://doi.org/10.1080/15567036.2014.934412
Ōtani S, Ōya A, Akagami J (1975) The effects of nickel on structural development in carbons. Carbon 13:353–356. https://doi.org/10.1016/0008-6223(75)90001-9
Ōya A, Marsh H (1982) Phenomena of catalytic graphitization. J Mater Sci 17:309–322. https://doi.org/10.1007/BF00591464
Ōya A, Yamashita R, Otani S (1979) Catalytic graphitization of carbons by borons. Fuel 58:325–330. https://doi.org/10.1016/0016-2361(79)90167-4
Ōya A, Inoue E, Ōtani S, Marsh H (1981) Surface areas of turbostratic graphitic carbons prepared from a resin using nickel particles, 20 nm, as graphitization catalyst. Fuel 60:1099–1100. https://doi.org/10.1016/0016-2361(81)90057-0
Pan Z, Wang R, Chen J (2019) Catalytic deoxygenation of methyl laurate as a model compound to hydrocarbons on hybrid catalysts composed of Ni-Zn alloy and HY zeolite. J Chem Technol Biotechnol 94:1777–1787. https://doi.org/10.1002/jctb.5943
Rico JR, Pardo AG, Fernandez JM, Popov VV, Orlova TS (2016) Thermal conductivity of Fe graphitized wood derived carbon. Mater Des 99:528–534. https://doi.org/10.1016/j.matdes.2016.03.070
Thompson E, Danks AE, Bourgeois L, Schnepp Z (2015) Iron-catalyzed graphitization of biomass. Green Chem 17:551–556. https://doi.org/10.1039/c4gc01673d
Upham DC, Agarwal V, Khechfe A, Snodgrass ZR, Gordan MJ, Metiu H et al (2017) Catalytic molten metals for the direct conversion of methane to hydrogen and separable carbon. Science 358:917–921. https://doi.org/10.1126/science.aao5023
Wahab MAA, Hamada MM, Mohamed A (2011) Artificial neural network and non-linear models for prediction of transformer oil residual operating time. Electr Power Syst Res 81:219–227. https://doi.org/10.1016/j.epsr.2010.08.010
Wang Y, Alsmeyer DC, McCreery RL (1990) Raman spectroscopy of carbon materials: structural basis of observed spectra. Chem Mater 2:557–563. https://doi.org/10.1021/cm00011a018
Williams PT, Williams EA (1999) Interaction of plastics in mixed-plastics pyrolysis. Energy Fuel 13(1):188–196. https://doi.org/10.1016/j.fuel.2010.05.032
Wu L, Dai H, Wen X, Wang P (2017) Ni-Zn alloy nanosheets arrayed on nickel foamas a promising catalyst for electrooxidation of hydrazine. Chem Electrochen 4:1944–1949. https://doi.org/10.1002/celc.201700234
Yan Q, Zhang X, Li J, Hassan E, Wang C, Zhang J et al (2018) Catalytic graphitization of kraft lignin to graphene-based structures with four different transitional metals. J Nanopart Res 20:223–220. https://doi.org/10.1007/s11051-018-4317-0
Yue L, Xia Q, Wang L, Wang L, DaCosta H, Yang J et al (2018) CO2 adsorption at nitrogen-doped carbons prepared by K2CO3 activation of urea-modified coconut shell. J Colloid Interface Sci 511:259–267. https://doi.org/10.1016/j.jcis.2017.09.040
Zhao L, Zhao X, Burke LT, Bennett JC, Dunlap RA, Obrovac MN (2017) Voronoi-tessellated graphite produced by low temperature catalytic graphitization from renewable resources. ChemSusChem 10:3409–3418. https://doi.org/10.1002/cssc.201701211
Zhi M, Yang F, Meng F, Li M, Manivannan A, Wu N (2014) Effects of pore structure on performance of an activated-carbon supercapacitor electrode recycled from scrap waste tires. Acs Sustain Chen Eng 2:1592–1598. https://doi.org/10.1021/sc500336h
Zhou H, Yu Q, Peng Q, Wang H, Chen J, Kuang Y (2008) Catalytic graphitization of carbon fibers with electrodeposited Ni-B alloy coating. Mater Chem Phys 110:434–439. https://doi.org/10.1016/j.matchemphys.2008.02.033
Zhu S, Li J, Ma L, He C, Liu E, He F et al (2018) Artificial neural network enabled capacitance prediction for carbon-based supercapacitors. Mater Lett 233:294–297. https://doi.org/10.1016/j.matlet.2018.09.028
Acknowledgments
The assistance from the Analytical and Testing Center of Huazhong University of Science and Technology is highly acknowledged. Also, thanks to the facility support of the Center for Nanoscale Characterization and Devices (CNCD), WNLO of HUST.
Funding
This research was financially supported by the National Natural Science Foundation of China (NSFC) (No. 51806079 and No. 51976074).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Zhihong Xu
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 272 kb)
Rights and permissions
About this article
Cite this article
Ren, Q., He, L., Hu, S. et al. Formation of highly graphitic char derived from phenolic resin carbonization by Ni-Zn-B alloy. Environ Sci Pollut Res 27, 22639–22647 (2020). https://doi.org/10.1007/s11356-020-08459-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08459-z