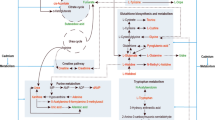
Abstract
Cadmium (Cd) has become one of the most important environmental pollutants in the world, derived from natural and industrial sources, which is known to be accumulated in the human body, producing serious health effects. On the other hand, Selenium (Se) is an essential element for mammals, which is well known for its antagonistic interaction against Cd toxicity, such as the prevention of oxidative stress induced by this element. For this reason, the use of complementary analytical methods to study the homeostasis of metals, “traffic” between different organs and massive information about metabolites altered by the exposure, is of great interest. To this end, a metabolomic workflow based on the use of direct infusion mass spectrometry (DIMS) and gas chromatography mass spectrometry (GC–MS) was applied in mice serum. On the other hand, metal homeostasis and traffic between different organs and serum of mice exposed to Cd and Se have been evaluated by determining the concentration of metals by inductively coupled plasma mass spectrometry. This work demonstrates for the first time that Cd exposure causes a decrease of all the elements studied in the lung except itself. On the other hand, Se provokes As trafficking from metabolically less active organs (brain, lung, and testes) to others with greater metabolic activity (kidney), which also facilitates its excretion. Moreover, when mice are only exposed to Se, it provokes the accumulation of almost all the elements in the kidney, except Cd that increases also in the liver and brain. However, when both elements are simultaneously administered, Se increases Cd concentration in all the organs except in the serum and especially in the testis. On the other hand, important metabolic alterations have been detected in the energy and amino acid metabolism, as well as degradation of phospholipidic membranes, and in free fatty acids. In summary, the results show the high potential of the combined use of organic and inorganic mass spectrometry to establish Cd and Se interaction and the biological impairments caused and to provide information about metal traffic and metabolomic changes in exposure experiments.



Similar content being viewed by others
References
Afolabi OK, Oyewo EB, Adekunle AS et al (2012) Impaired lipid levels and inflammatory response in rats exposed to cadmium. EXCLI J 11:677–687
Bashir S, Sharma Y, Irshad M et al (2006) Arsenic-induced cell death in liver and brain of experimental rats. Basic Clin Pharmacol Toxicol 98:38–43. https://doi.org/10.1111/j.1742-7843.2006.pto_170.x
Begley P, Francis-McIntyre S, Dunn WB et al (2009) Development and performance of a gas chromatography-time-of-flight mass spectrometry analysis for large-scale nontargeted metabolomic studies of human serum. Anal Chem 81:7038–7046. https://doi.org/10.1021/ac9011599
Bell RR, Soliman MRI, Early JL II (1990) Acute effects of cadmium and selenium on glucose output from rat liver hepatocytes using various gluconeogenic precursors. Toxicology 65:161–168. https://doi.org/10.1016/0300-483X(90)90086-V
Benavides MP, Gallego SM, Tomaro ML (2005) Cadmium toxicity in plants. Braz J Plant Physiol 17:21–34. https://doi.org/10.1590/S1677-04202005000100003
Bernard A (2008) Cadmium & its adverse effects on human health. Indian J Med Res 128:557–564
Bowen BP, Northen TR (2010) Dealing with the unknown: metabolomics and metabolite atlases. J Am Soc Mass Spectrom 21:1471–1476. https://doi.org/10.1016/j.jasms.2010.04.003
Chang K-C, Hsu C-C, Liu S-H et al (2013) Cadmium induces apoptosis in pancreatic β-cells through a mitochondria-dependent pathway: the role of oxidative stress-mediated c-Jun N-terminal kinase activation. PLoS One:8–e54374. https://doi.org/10.1371/journal.pone.0054374
Dakubo GD (2010) The Warburg phenomenon and other metabolic alterations of cancer cells. In: Mitochondrial genetics and cancer. Springer Berlin Heidelberg, Berlin, Heidelberg
Dettmer K, Aronov PA, Hammock BD (2007) Mass spectrometry-based metabolomics. Mass Spectrom Rev 26:51–78. https://doi.org/10.1002/mas.20108
Draper J, Lloyd AJ, Goodacre R, Beckmann M (2013) Flow infusion electrospray ionisation mass spectrometry for high throughput, non-targeted metabolite fingerprinting: A review. Metabolomics 9. https://doi.org/10.1007/s11306-012-0449-x
Early JL Jr, Schnell RC (1981) Selenium antagonism of cadmium-induced inhibition of hepatic drug metabolism in the male rat. Toxicol Appl Pharmacol 58:57–66. https://doi.org/10.1016/0041-008X(81)90115-0
El-Maraghy SA, Gad MZ, Fahim AT, Hamdy MA (2001) Effect of cadmium and aluminum intake on the antioxidant status and lipid peroxidation in rat tissues. J Biochem Mol Toxicol 15:207–214. https://doi.org/10.1002/jbt.18
FAO/WHO (Expert Committee on food additives) (2004) Summary of evaluations performed by the joint FAO/WHO expert committee on food additives (JECFA) 1956–2003
Favretto D, Cosmi E, Ragazzi E et al (2012) Cord blood metabolomic profiling in intrauterine growth restriction. Anal Bioanal Chem 402:1109–1121. https://doi.org/10.1007/s00216-011-5540-z
Fernández-Caballero A, Wróbel JS, Mummery PM, Nguyen-Manh D (2017) Short-range order in high entropy alloys: theoretical formulation and application to Mo-Nb-Ta-V-W system. J Phase Equilib Diffus 38:391–403. https://doi.org/10.1007/s11669-017-0582-3
Flora SJS, Behari JR, Ashquin M, Tandon SK (1982) Time-dependent protective effect of selenium against cadmium-induced nephrotoxicity and hepatotoxicity. Chem Biol Interact 42:345–351. https://doi.org/10.1016/0009-2797(82)90078-3
Frisk P, Yaqob A, Lindh U (2002) Indications of selenium protection against cadmium toxicity in cultured K-562 cells. Sci Total Environ 296:189–197. https://doi.org/10.1016/S0048-9697(02)00080-3
García-Barrera T, Gómez-Ariza JL, González-Fernández M, Moreno F, García-Sevillano MA, Gómez-Jacinto V (2012) Biological responses related to agonistic, antagonistic and synergistic interactions of chemical species. Anal Bioanal Chem 403:2237–2253. https://doi.org/10.1007/s00216-012-5776-2
García-Barrera T, Rodríguez-Moro G, Callejón-Leblic B et al (2018) Mass spectrometry based analytical approaches and pitfalls for toxicometabolomics of arsenic in mammals: a tutorial review. Anal Chim Acta 1000:41–66. https://doi.org/10.1016/j.aca.2017.10.019
García-Sevillano MA, García-Barrera T, Navarro F, Gómez-Ariza JL (2013a) Analysis of the biological response of mouse liver (Mus musculus) exposed to As2O3 based on integrated -omics approaches. Metallomics 5:1644–1655. https://doi.org/10.1039/c3mt00186e
García-Sevillano MA, Jara-Biedma R, González-Fernández M, García-Barrera T, Gómez-Ariza JL (2013b) Metal interactions in mice under environmental stress. Biometals 26:651–666. https://doi.org/10.1007/s10534-013-9642-2
García-Sevillano MA, Contreras-Acuña M, García-Barrera T, Navarro F, Gómez-Ariza JL (2014a) Metabolomic study in plasma, liver and kidney of mice exposed to inorganic arsenic based on mass spectrometry. Anal Bioanal Chem 406:1455–1469. https://doi.org/10.1007/s00216-013-7564-z
García-Sevillano MA, García-Barrera T, Navarro F, Gómez-Ariza JL (2014b) Cadmium toxicity in Mus musculus mice based on a metallomic study. Antagonistic interaction between Se and Cd in the bloodstream. Metallomics 6:672–681. https://doi.org/10.1039/c3mt00350g
García-Sevillano MT, García-Barrera T, Navarro-Roldán F et al (2014c) A combination of metallomics and metabolomics studies to evaluate the effects of metal interactions in mammals. Application to Mus musculus mice under arsenic/cadmium exposure. J Proteomics 104. https://doi.org/10.1016/j.jprot.2014.02.011
García-Sevillano MA, Abril N, Fernández-Cisnal R, García-Barrera T, Pueyo C, López-Barea J, Gómez-Ariza JL (2015a) Functional genomics and metabolomics reveal the toxicological effects of cadmium in Mus musculus mice. Metabolomics 11:1432–1450. https://doi.org/10.1007/s11306-015-0801-z
García-Sevillano MA, García-Barrera T, Navarro F et al (2015b) Combination of direct infusion mass spectrometry and gas chromatography mass spectrometry for toxicometabolomic study of red blood cells and serum of mice Mus musculus after mercury exposure. J Chromatogr B Anal Technol Biomed Life Sci 985:75–84. https://doi.org/10.1016/j.jchromb.2015.01.029
García-Sevillano MA, Rodríguez-Moro G, García-Barrera T et al (2015c) Biological interactions between mercury and selenium in distribution and detoxification processes in mice under controlled exposure. Effects on selenoprotein. Chem Biol Interact 229:82–90. https://doi.org/10.1016/j.cbi.2015.02.001
Glantzounis G, Tsimoyiannis E, Kappas A, Galaris D (2005) Uric acid and oxidative stress. Curr Pharm Des 11:4145–4151. https://doi.org/10.2174/138161205774913255
Goldsmith P, Fenton H, Morris-Stiff G et al (2010) Metabonomics: a useful tool for the future surgeon. J Surg Res 160:122–132. https://doi.org/10.1016/j.jss.2009.03.003
González-Domínguez R, García-Barrera T, Gómez-Ariza J-L (2012) Metabolomic approach to Alzheimer’s disease diagnosis based on mass spectrometry. Chem Pap 66:829–835. https://doi.org/10.2478/s11696-012-0184-9
González-Domínguez R, García-Barrera T, Vitorica J, Gómez-Ariza JL (2015) Metabolomics reveals significant impairments in the immune system of the APP/PS1 transgenic mice of Alzheimer’s disease. Electrophoresis 36:577–587. https://doi.org/10.1002/elps.201400450
Griffin JL, Walker L, Shore RF, Nicholson JK (2001) High-resolution magic angle spinning 1H-NMR spectroscopy studies on the renal biochemistry in the bank vole (Clethrionomys glareolus) and the effects of arsenic (As3+) toxicity. Xenobiotica 31:377–385. https://doi.org/10.1080/00498250110055938
Gürer H, Ozgünes H, Saygin E, Ercal N (2001) Antioxidant effect of taurine against lead-induced oxidative stress. Arch Environ Contam Toxicol 41:397–402. https://doi.org/10.1007/s002440010265
Hems DA (1972) Metabolism of glutamine and glutamic acid by isolated perfused kidneys of normal and acidotic rats. Biochem J 130:671–680
Holmberg RE, Ferm VH (1969) Interrelationships of selenium, cadmium, and arsenic in mammalian teratogenesis. Arch Environ Health 18:873–877. https://doi.org/10.1080/00039896.1969.10665508
Jin Y, Zhao F, Zhong Y et al (2010) Effects of exogenous GSH and methionine on methylation of inorganic arsenic in mice exposed to arsenite through drinking water. Environ Toxicol 25:361–366. https://doi.org/10.1002/tox.20509
Katajamaa M, Oresic M (2007) Data processing for mass spectrometry-based metabolomics. J Chromatogr A 1158:318–328. https://doi.org/10.1016/j.chroma.2007.04.021
Koivusalo M, Haimi P, Heikinheimo L et al (2001) Quantitative determination of phospholipid compositions by esi-ms: Effects of acyl chain length, unsaturation, and lipid concentration on instrument response. J Lipid Res 42:663–672
Koyama H, Kitoh H, Satoh M, Tohyama C (2002) Low dose exposure to cadmium and its health effects (1). Genotoxicity and carcinogenicity Japanese. J Hyg 57:547–555
Larregle EV, Varas SM, Oliveros LB et al (2008) Lipid metabolism in liver of rat exposed to cadmium. Food Chem Toxicol 46:1786–1792. https://doi.org/10.1016/j.fct.2008.01.018
Lin L, Yu Q, Yan X et al (2010) Direct infusion mass spectrometry or liquid chromatography mass spectrometry for human metabonomics? A serum metabonomic study of kidney cancer. Analyst 135:2970–2978. https://doi.org/10.1039/c0an00265h
Liu Y, Zhang S-P, Cai Y-Q (2007) Cytoprotective effects of selenium on cadmium-induced LLC-PK<inf>1</inf>cells apoptosis by activating JNK pathway. Toxicol in Vitro 21:677–684. https://doi.org/10.1016/j.tiv.2007.01.015
Martelli A, Rousselet E, Dycke C et al (2006) Cadmium toxicity in animal cells by interference with essential metals. Biochimie 88. https://doi.org/10.1016/j.biochi.2006.05.013
Messaoudi I, El Heni J, Hammouda F et al (2009) Protective effects of selenium, zinc, or their combination on cadmium-induced oxidative stress in rat kidney. Biol Trace Elem Res 130:152–161. https://doi.org/10.1007/s12011-009-8324-y
Mukherjee A, Sharma A, Talukder G (1988) Effect of selenium on cadmium-induced chromosomal aberrations in bone marrow cells of mice. Toxicol Lett 41:23–29. https://doi.org/10.1016/0378-4274(88)90004-5
Murugavel P, Pari L (2007) Effects of diallyl tetrasulfide on cadmium-induced oxidative damage in the liver of rats. Hum Exp Toxicol 26:527–534. https://doi.org/10.1177/0960327107073810
Nehru LB, Bansal MP (1997) Effect of selenium supplementation on the glutathione redox system in the kidney of mice after chronic cadmium exposures. J Appl Toxicol 17:81–84. https://doi.org/10.1002/(SICI)1099-1263(199701)17:1<81::AID-JAT398>3.0.CO;2-K
Nordberg GF (2009) Historical perspectives on cadmium toxicology. Toxicol Appl Pharmacol 238:192–200. https://doi.org/10.1016/j.taap.2009.03.015
Pappas A, Zoidis E, Fegeros K, et al. (2011) Relation of cadmium to other elements and the antioxidant system. In: Cadmium in the environment. Nova Science, New York, USA
Prohaska JR, Mowafy M, Ganther HE (1977) Interactions between cadmium, selenium and glutathione peroxidase in rat testis. Chem Biol Interact 18:253–265. https://doi.org/10.1016/0009-2797(77)90012-6
Saygi S, Deniz G, Kutsal O, Vural N (1991) Chronic effects of cadmium on kidney, liver, testis, and fertility of male rats. Biol Trace Elem Res 31:209–214. https://doi.org/10.1007/BF02990191
Schwudke D, Schuhmann K, Herzog R et al (2011) Shotgun lipidomics on high resolution mass spectrometers. Cold Spring Harb Perspect Biol 3:1–13. https://doi.org/10.1101/cshperspect.a004614
Sidhu M, Sharma M, Bhatia M et al (1993) Effect of chronic cadmium exposure on glutathione S-transferase and glutathione peroxidase activities in Rhesus monkey: the role of selenium. Toxicology 83:203–213. https://doi.org/10.1016/0300-483X(93)90102-X
Skipski VP, Barclay M, Barclay RK et al (1967) Lipid composition of human serum lipoproteins. Biochem J 104:340–352
Stadtman TC (1991) Biosynthesis and function of selenocysteine-containing enzymes. J Biol Chem 266:16257–16260
Sugawara N, Sugawara C (1984) Selenium protection against testicular lipid peroxidation from cadmium. J Appl Biochem 6:199–204
Sun G, Yang K, Zhao Z et al (2007) Shotgun metabolomics approach for the analysis of negatively charged water-soluble cellular metabolites from mouse heart tissue. Anal Chem 79:6629–6640. https://doi.org/10.1021/ac070843+
Teodorova S, Metcheva R, Topashka-Ancheva M (2003) Bioaccumulation and damaging action of polymetal industrial dust on laboratory mice Mus musculus alba: I. Analysis of Zn, Cu, Pb, and Cd disposition and mathematical model for Zn and Cd bioaccumulations. Environ Res 91:85–94. https://doi.org/10.1016/S0013-9351(02)00013-0
Vuckovic D (2012) Current trends and challenges in sample preparation for global metabolomics using liquid chromatography-mass spectrometry. Anal Bioanal Chem 403:1523–1548. https://doi.org/10.1007/s00216-012-6039-y
Waalkes MP (2003) Cadmium carcinogenesis. Mutat Res Fundam Mol Mech Mutagen 533:107–120. https://doi.org/10.1016/j.mrfmmm.2003.07.011
Zhai Q, Wang G, Zhao J et al (2013) Protective effects of Lactobacillus plantarum CCFM8610 against acute cadmium toxicity in mice. Appl Environ Microbiol 79:1508–1515. https://doi.org/10.1128/AEM.03417-12
Funding
This work has been financially supported by projects CTM2015-67902-C2-1-P and PG2018-096608-B-C21 from the Spanish Ministry of Economy and Competitiveness and P12-FQM-0442 from the Regional Ministry of Economy, Innovation, Science and Employment (Andalusian Government, Spain). Gema Rodríguez Moro would like to thank the Spanish Ministry of Economy and Competitiveness for a PhD scholarship (BES-2013-064501). Finally, authors are grateful to FEDER (European Community) for the financial support, grant numbers UNHU13-1E-1611 and UNHU15-CE-3140.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All animals received humane care in compliance with the guidelines of the animal care and use of the European Community. The investigation was performed with the consent of the Ethical Committee of the University of Huelva (Spain).
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 14.9 kb)
Rights and permissions
About this article
Cite this article
Rodríguez-Moro, G., Roldán, F.N., Baya-Arenas, R. et al. Metabolic impairments, metal traffic, and dyshomeostasis caused by the antagonistic interaction of cadmium and selenium using organic and inorganic mass spectrometry. Environ Sci Pollut Res 27, 1762–1775 (2020). https://doi.org/10.1007/s11356-019-06573-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-06573-1