Abstract
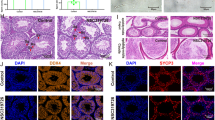
Hydroxyurea (HDU), a class of antineoplastic drugs, has a powerful efficacy in the treatment of several types of malignancies. However, it has multiple adverse effects including reduced fertility, especially in males. Thus, 60 male albino rats were used to investigate the chemoprotective potentials of royal jelly on HDU-induced testicular damage. Animals were gastro-gavaged with HDU (225 or 450 mg kg−1 bw day−1) before royal jelly (100 mg kg−1 bw day−1) for 60 days. Blood samples and testicles were collected, and spermatozoon was obtained. In a dose-dependent manner, the sperm count, motility and liveability, and testosterone, GSH, and catalase concentrations were decreased in HDU groups, whereas MDA, FSH, LH, IL-6, and IFN-γ expression levels were increased. Germinal epithelium degeneration, germ cell sloughing, reduction in the number of luminal spermatozoa, interstitial congestion, and severe leukocyte infiltration besides no glandular secretion in most of the acini were identified. However, royal jelly intake in HDU-treated rats successfully improved sperm quality, hormonal and antioxidant status, and reproductive organ histoarchitecture. Thus, it could be concluded that royal jelly is endowed with antioxidative and anti-inflammatory activities and could be, therefore, used as an adjuvant remedy to improve HDU-induced male subfertility.





Similar content being viewed by others
Abbreviations
- AR:
-
androgen receptor
- CAT:
-
catalase
- CTR:
-
control
- GSH:
-
reduced glutathione
- HDU:
-
hydroxyurea
- HE:
-
hematoxylin and eosin
- IL-6:
-
interleukin-6
- IFN-γ:
-
interferon-γ
- FSH:
-
follicular stimulating hormone
- LH:
-
luteinizing hormone
- MDA:
-
malondialdehyde
- PBS:
-
phosphate buffer saline
- RJ:
-
Royal jelly
- ROS:
-
reactive oxygen species
- SEM:
-
standard error of mean
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Agrawal RK, Patel RK, Shah V, Nainiwal L, Trivedi B (2014) Hydroxyurea in sickle cell disease: drug review. Indian J Hematol Blood Transfus 30:91–96
Ahmad MF, Ansari MO, Jameel S, Wani AL, Parveen N, Siddique HR, Shadab G (2018) Protective role of nimbolide against chemotherapeutic drug hydroxyurea induced genetic and oxidative damage in an animal model. Environ Toxicol Pharmacol 60:91–99
Amirshahi T, Najafi G, Nejati V (2014) Protective effect of royal jelly on fertility and biochemical parameters in bleomycin-induced male rats. Iran J Reprod Med 12:209–216
Berthaut I, Guignedoux G, Kirsch-Noir F, de Larouziere V, Ravel C, Bachir D, Galacteros F, Ancel PY, Kunstmann JM, Levy L, Jouannet P, Girot R, Mandelbaum J (2008) Influence of sickle cell disease and treatment with hydroxyurea on sperm parameters and fertility of human males. Haematologica 93:988–993
Berthaut I, Bachir D, Kotti S, Chalas C, Stankovic K, Eustache F, Ravel C, Habibi A, Brailly-Tabard S, Levy-Dutel L, Bleibtreu A, Simon T, Galacteros F, Lionnet F, Mandelbaum J (2017) Adverse effect of hydroxyurea on spermatogenesis in patients with sickle cell anemia after 6 months of treatment. Blood 130:2354–2356
Bjorndahl L, Soderlund I, Kvist U (2003) Evaluation of the one-step eosin-nigrosin staining technique for human sperm vitality assessment. Hum Reprod 18:813–816
Blanco-Rodriguez J, Martinez-Garcia C (1998) Apoptosis precedes detachment of germ cells from the seminiferous epithelium after hormone suppression by short-term oestradiol treatment of rats. Int J Androl 21:109–115
Charache S, Terrin ML, Moore RD, Dover GJ, Barton FB, Eckert SV, McMahon RP, Bonds DR (1995): Effect of hydroxyurea on the frequency of painful crises in sickle cell anemia. Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia. N Engl J Med 332, 1317–1322
Chen YF, Wang K, Zhang YZ, Zheng YF, Hu FL (2016) In vitro anti-inflammatory effects of three fatty acids from royal jelly. Mediat Inflamm 2016:3583684
Ciftci O, Aydin M, Ozdemir I, Vardi N (2012a) Quercetin prevents 2,3,7,8-tetrachlorodibenzo-p-dioxin-induced testicular damage in rats. Andrologia 44:164–173
Ciftci O, Ozdemir I, Aydin M, Beytur A (2012b) Beneficial effects of chrysin on the reproductive system of adult male rats. Andrologia 44:181–186
Cokic VP, Smith RD, Beleslin-Cokic BB, Njoroge JM, Miller JL, Gladwin MT, Schechter AN (2003) Hydroxyurea induces fetal hemoglobin by the nitric oxide-dependent activation of soluble guanylyl cyclase. J Clin Invest 111:231–239
Corradi PF, Corradi RB, Greene LW (2016) Physiology of the hypothalamic pituitary gonadal axis in the male. Urol Clin North Am 43:151–162
Creasy DM, Chapin RE (2013) Chapter 59 - male reproductive system. In: Haschek WM, Rousseaux CG, Wallig MA (eds) Haschek and Rousseaux’s handbook of toxicologic pathology, Third edn. Academic Press, Boston, pp 2493–2598
El-Maddawy ZK, El-Sayed YS (2018) Comparative analysis of the protective effects of curcumin and N-acetyl cysteine against paracetamol-induced hepatic, renal, and testicular toxicity in Wistar rats. Environ Sci Pollut Res Int 25:3468–3479
El-Neweshy MS, El-Maddawy ZK, El-Sayed YS (2013) Therapeutic effects of date palm (Phoenix dactylifera L.) pollen extract on cadmium-induced testicular toxicity. Andrologia 45:369–378
Florensa R, Bachs O, Agell N (2003) ATM/ATR-independent inhibition of cyclin B accumulation in response to hydroxyurea in nontransformed cell lines is altered in tumour cell lines. Oncogene 22:8283–8292
Friedrisch JR, Pra D, Maluf SW, Bittar CM, Mergener M, Pollo T, Kayser M, da Silva MA, Henriques JA, da Rocha Silla LM (2008) DNA damage in blood leukocytes of individuals with sickle cell disease treated with hydroxyurea. Mutat Res 649:213–220
Fujiwara S, Imai J, Fujiwara M, Yaeshima T, Kawashima T, Kobayashi K (1990) A potent antibacterial protein in royal jelly. Purification and determination of the primary structure of royalisin. J Biol Chem 265:11333–11337
Garozzo G, Disca S, Fidone C, Bonomo P (2000) Azoospermia in a patient with sickle cell disease treated with hydroxyurea. Haematologica 85:1216–1218
Ghanbari E, Nejati V, Najafi G, Khazaei M, Babaei M (2015) Study on the effect of royal jelly on reproductive parameters in streptozotocin-induced diabetic rats. Int J Fertil Steril 9:113–120
Gharagozloo P, Aitken RJ (2011) The role of sperm oxidative stress in male infertility and the significance of oral antioxidant therapy. Hum Reprod 26:1628–1640
Grigg A (2007) Effect of hydroxyurea on sperm count, motility and morphology in adult men with sickle cell or myeloproliferative disease. Intern Med J 37:190–192
Gu L, Xiong WT, Wang C, Sun HX, Li GF, Liu X (2013) Cistanche deserticola decoction alleviates the testicular toxicity induced by hydroxyurea in male mice. Asian J Androl 15:838–840
Guo H, Ekusa A, Iwai K, Yonekura M, Takahata Y, Morimatsu F (2008) Royal jelly peptides inhibit lipid peroxidation in vitro and in vivo. J Nutr Sci Vitaminol (Tokyo) 54:191–195
Hattori N, Nomoto H, Fukumitsu H, Mishima S, Furukawa S (2007) Royal jelly and its unique fatty acid, 10-hydroxy-trans-2-decenoic acid, promote neurogenesis by neural stem/progenitor cells in vitro. Biomed Res 28:261–266
Hodge DR, Hurt EM, Farrar WL (2005) The role of IL-6 and STAT3 in inflammation and cancer. Eur J Cancer 41:2502–2512
Hsu PC, Liu MY, Hsu CC, Chen LY, Guo YL (1998) Effects of vitamin E and/or C on reactive oxygen species-related lead toxicity in the rat sperm. Toxicology 128:169–179
Huang J, Yakubu M, Kim-Shapiro DB, King SB (2006) Rat liver-mediated metabolism of hydroxyurea to nitric oxide. Free Radic Biol Med 40:1675–1681
Italia K, Colah R, Ghosh K (2013) Hydroxyurea could be a good clinically relevant iron chelator. PLoS One 8:e82928
Jones KM, Niaz MS, Brooks CM, Roberson SI, Aguinaga MP, Hills ER, Rice VM, Bourne P, Bruce D, Archibong AE (2009) Adverse effects of a clinically relevant dose of hydroxyurea used for the treatment of sickle cell disease on male fertility endpoints. Int J Environ Res Public Health 6:1124–1144
Juul T, Malolepszy A, Dybkaer K, Kidmose R, Rasmussen JT, Andersen GR, Johnsen HE, Jorgensen JE, Andersen SU (2010) The in vivo toxicity of hydroxyurea depends on its direct target catalase. J Biol Chem 285:21411–21415
Kolayli S, Sahin H, Can Z, Yildiz O, Malkoc M, Asadov A (2016) A member of complementary medicinal food: anatolian royal jellies, their chemical compositions, and antioxidant properties. J Evid Based Complementary Altern Med 21:NP43–NP48
Kumar SG, Narayana K, Bairy KL, D'Souza UJ, Samuel VP, Gopalakrishna K (2006) Dacarbazine induces genotoxic and cytotoxic germ cell damage with concomitant decrease in testosterone and increase in lactate dehydrogenase concentration in the testis. Mutat Res 607:240–252
Lamanna C, Assisi L, Vittoria A, Botte V, Di Fiore MM (2007) D-Aspartic acid and nitric oxide as regulators of androgen production in boar testis. Theriogenology 67:249–254
Liebelt EL, Balk SJ, Faber W, Fisher JW, Hughes CL, Lanzkron SM, Lewis KM, Marchetti F, Mehendale HM, Rogers JM, Shad AT, Skalko RG, Stanek EJ (2007) NTP-CERHR expert panel report on the reproductive and developmental toxicity of hydroxyurea. Birth Defects Res B, Dev Reprod Toxicol 80:259–366
Muhl H, Pfeilschifter J (2003) Anti-inflammatory properties of pro-inflammatory interferon-gamma. Int Immunopharmacol 3:1247–1255
Nair AB, Jacob S (2016) A simple practice guide for dose conversion between animals and human. J Basic Clin Pharm 7:27–31
Najafi G, Nejati V, Shalizar Jalali A, Zahmatkesh E (2014) Protective role of royal jelly in oxymetholone-induced oxidative injury in mouse testis. Iranian J Toxicol 8:1073–1080
O'Connor JC, Frame SR, Ladics GS (2002) Evaluation of a 15-day screening assay using intact male rats for identifying antiandrogens. Toxicol Sci 69:92–108
O'Donnell L, Nicholls PK, O'Bryan MK, McLachlan RI, Stanton PG (2011) Spermiation: the process of sperm release. Spermatogenesis 1:14–35
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Ono Y, Suzuki K, Kashiwagi B, Shibata Y, Ito K, Fukabori Y, Yamanaka H (2004) Role of androgen on blood flow and capillary structure in rat seminal vesicles. Tohoku J Exp Med 202:193–201
Pasupuleti VR, Sammugam L, Ramesh N, Gan SH (2017) Honey, propolis, and royal jelly: a comprehensive review of their biological actions and health benefits. Oxidative Med Cell Longev 2017:1259510
Perreault S, Klinefelter G, Clegg E (2008) Assessment of male reproductive toxicity. In: Hayes AW (ed) Principles and methods of toxicology. Raven Press, New York, NY, pp 1605–1640
Richardson RJ, Murphy SD (1975) Effect of glutathione depletion on tissue deposition of methylmercury in rats. Toxicol Appl Pharmacol 31:505–519
Roy S, Banerjee A, Pandey HC, Singh G, Kumari GL (2001) Application of seminal germ cell morphology and semen biochemistry in the diagnosis and management of azoospermic subjects. Asian J Androl 3:55–62
Shalizar Jalali A, Najafi G, Hosseinchi M, Sedighnia A (2015) Royal jelly alleviates sperm toxicity and improves in vitro fertilization outcome in stanozolol-treated mice. Iran J Reprod Med 13:15–22
Shin JH, Mori C, Shiota K (1999) Involvement of germ cell apoptosis in the induction of testicular toxicity following hydroxyurea treatment. Toxicol Appl Pharmacol 155:139–149
Sver L, Orsolic N, Tadic Z, Njari B, Valpotic I, Basic I (1996) A royal jelly as a new potential immunomodulator in rats and mice. Comp Immunol Microbiol Infect Dis 19:31–38
Wiger R, Hongslo JK, Evenson DP, De Angelis P, Schwarze PE, Holme JA (1995) Effects of acetaminophen and hydroxyurea on spermatogenesis and sperm chromatin structure in laboratory mice. Reprod Toxicol 9:21–33
Xue X, Wu L, Wang K (2017) Chemical composition of royal jelly. In: Alvarez-Suarez JM (ed) Bee products - chemical and biological properties. Springer International Publishing, Cham, pp 181–190
Zahmatkesh E, Najafi G, Nejati V, Heidari R (2014) Protective effect of royal jelly on the sperm parameters and testosterone level and lipid peroxidation in adult mice treated with oxymetholone. Avicenna J Phytomed 4:43–52
Zhou L, Wu CQ, Luo YW, Liao MY, Sun ZY (2015) Studies on the characteristics and mechanisms of testicular toxicity induced by hydroxyurea. Toxicol Mech Methods 25:396–401
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that there are no conflicts of interest.
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tohamy, H.G., Gad El-Karim, D.R. & El-Sayed, Y.S. Attenuation potentials of royal jelly against hydroxyurea-induced infertility through inhibiting oxidation and release of pro-inflammatory cytokines in male rats. Environ Sci Pollut Res 26, 21524–21534 (2019). https://doi.org/10.1007/s11356-019-05521-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-05521-3