Abstract
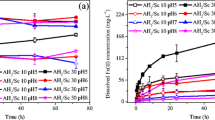
Schwertmannite, a Fe(III)-oxyhydroxysulfate mineral formed in acidic (pH 3~4), iron- and sulfate-rich acid mine drainage (AMD) environments, tends to undergo phase transformations with changes in pH and redox condition, which may depend on the presence of various trace anions and cations. In the present study, the effects of Cu(II) on the stability of arsenate-, chromate-, and molybdate-substituted schwertmannite were investigated. The release of Fe(III) and sulfate from schwertmannite seems to be accelerated in the presence of Cu(II) at pH ~ 3, while Cu(II) retarded the dissolution of schwertmannite at pH ~ 5. XRD and SEM results showed that pure schwertmannite and chromate-substituted schwertmannite underwent transformation to goethite over a 2-month period, the presence of Cu(II) enhanced the stability of the mineral’s structure at both pH 3 and 5. However, the structures of arsenate- and molybdate-substituted schwertmannites showed no significant changes in the presence or absence of Cu(II) at both pH 3 and 5. During the phase transformation process, the amount of released oxyanions followed the sequence of chromate > molybdate > arsenate. Moreover, the release of arsenate and chromate from schwertmannite was retarded in the presence of Cu(II) at pH 5, whereas the release of molybdate was promoted. These results have important environmental implications for the stability of schwertmannite and its potential to immobilize contaminant trace elements under AMD conditions.











Similar content being viewed by others
References
Acero P, Ayora C, Torrentó C, Nieto J (2006) The behavior of trace elements during schwertmannite precipitation and subsequent transformation into goethite and jarosite. Geochim Cosmochim Acta 70:4130–4139
Antelo J, Fiol S, Gondar D, López R, Arce F (2012) Comparison of arsenate, chromate and molybdate binding on schwertmannite: surface adsorption vs anion-exchange. J Colloid Interface Sci 386:338–343
Antelo J, Fiol S, Gondar D, Pérez C, López R, Arce F (2013) Cu(II) incorporation to schwertmannite: effect on stability and reactivity under AMD conditions. Geochim Cosmochim Acta 119:149–163
Azzali E, Marescotti P, Frau F, Dinelli E, Carbone C, Capitani G, Lucchetti G (2014) Mineralogical and chemical variations of ochreous precipitates from acid sulphate waters (asw) at the Rosia Montana gold mine (Romania). Environ Earth Sci 72:3567–3584
Ball JW, Nordstrom DK (1998) Critical evaluation and selection of standard state thermodynamic properties for chromium metal and its aqueous ions, hydrolysis species, oxides, and hydroxides. J Chem Eng Data 43:895–918
Bigham JM, Schwertmann U, Carlson L, Murad E (1990) A poorly crystallized oxyhydoxysulfate of iron formed by bacterial oxidation of Fe(II) in acid mine waters. Geochim Cosmochim Acta 54:2743–2758
Bigham JM, Schwertmann U, Traina SJ, Winland RL, Wolf M (1996) Schwertmannite and the chemical modeling of iron in acid sulfate waters. Geochim Cosmochim Acta 60:2111–2121
Burton ED, Bush RT, Sullivan LA, Mitchell DRG (2008) Schwertmannite transformation to goethite via the Fe(II) pathway: reaction rates and implications for iron-sulfate formation. Geochim Cosmochim Acta 72:4551–4564
Carlson L, Bigham JM, Schwertmann U, Kyek A, Wagner F (2002) Scavenging of As by schwertmannite and ferrihydrite from acid mine drainage: a comparison with synthetic analogues. Environ Sci Technol 36:1712–1719
Chen MQ, Lu GN, Guo CL, Yang CF, Wu JX, Huang WL, Yee N, Dang Z (2015) Sulfate migration in a river affected by acid mine drainage from the Dabaoshan mining area, South China. Chemosphere 119:734–743
Consani S, Carbone C, Salviulo G, Zorzi F, Dinelli E, Botter R, Nodari L, Badocco D, Lucchetti G (2016) Effect of temperature on the release and remobilization of ecotoxic elements in AMD colloidal precipitates: the example of Libiola copper mine, Liguria, (Italy). Environ Sci Pollution R 23(13):12900–12914
Cornell RM, Schwertmann U (2003) The iron oxides: structure, properties, reactions, occurrences and uses. Weinheim, Wiley-VHC
Cutting RS, Coker VS, Telling ND, Kimber RL, Laan G, Pattrick RAD, Vaughan DJ, Arenholz E, Lloyd JR (2012) Microbial reduction of arsenic-doped schwertmannite by Geobacter sulfurreducens. Environ Sci Technol 46:12591–12599
Dzombak DA, Morel FMM (1990) Surface complexation modeling: hydrous ferric oxide. Wiley-Interscience, New York
Fulladosa E, Murat JC, Martinez M, Villaescusa I (2004) Effect of pH on arsenate and arsenite toxicity to luminescent Bateria (Vibrio fischeri). Arch. Environ. ContamToxicology 46:176–182
Gagliano WB, Brill MR, Bigham JM, Jones FS, Traina SJ (2004) Chemistry and mineralogy of ochreous sediments in a constructed mine drainage wetland. Geochim Cosmochim Acta 68:2119–2128
Guo YW, Zhu ZL, Qiu YL, Zhao JF (2013) Synthesis of mesoporous Cu/Mg/Fe layered double hydroxide and its adsorption performance for arsenate in aqueous solution. J Environ Sci 25:944–953
Houng Aloune S, Kawaai T, Hiroyoshi N, Ito M (2014) Study on schwertmannite production from copper heap leach solutions and its efficiency in arsenic removal from acidic sulfate solutions. Hydrometallurgy 146-148:30–40
Houng Aloune S, Hiroyoshi N, Ito M (2015) Stability of As(V)-sorbed schwertmannite under porphyry copper mine conditions. Miner Eng 74:51–59
Jönsson J, Persson P, Sjöberg S, Lövgren L (2005) Schwertmannite precipitated from acid mine drainage: phase transformation, sulphate release and surface properties. Appl Geochem 20:179–191
Liu YY, Mou HY, Chen LC, Mirza ZA, Liu L (2015) Cr(VI)-contaminated groundwater remediation with simulated permeable reactive barrier (PRB) filled with natural pyrite as reactive material: environmental factors and effectiveness. J Hazard Mater 298:83–90
Liu LD, Wang WM, Liu L, Yu B, Zhang YX, Wu XQ, Zhang HW, Han X (2016) Catalytic activities of dissolved and Sch-immobilized Mo in H2O2 decomposition: implications for phenol oxidation under acidic conditions. Appl Catal B Environ 185:371–377
Mahadevaiah N, Venkataramani B, Prakash BSJ (2007) Restrictive entry of aqueous molybdate species into surfactant modified montmorillonite-a breakthrough curve study. Chem Mater 19:4606–4612
Mohapatra M, Sahoo SK, Anand S, Das RP (2006) Removal of As(V) by Cu(II)-, Ni(II)-, or Co(II)-doped goethite samples. J Colloid Interface Sci 298:6–12
Nagano T, Yanase N, Hanzawa Y, Takada M, Mitamura H, Sato T, Naganawa H (2011) Evaluation of the affinity of some toxic elements to schwertmannite in natural streams contaminated with acid mine drainage. Water Air Soil Pollut 216:153–166
Otero-Fariña A, Gago R, Antelo J, Fiol S, Arce F (2015) Surface complexation modelling of arsenic and copper immobilization by iron oxide precipitates derived from acid mine drainage. Bol Soc Geol Mex 67:493–508
Paikaray S, Peiffer S (2010) Dissolution kinetics of sulfate from schwertmannite under variable pH conditions. Mine Water Environ 29:263–269
Ponthieu M, Juillot F, Hiemstra T, Riemsdijk WH, Benedetti MF (2006) Metal ion binding to iron oxides. Geochim Cosmochim Acta 70:2679–2698
Regenspurg S, Peiffer S (2005) Arsenate and chromate incorporation in schwertmannite. Appl Geochem 20:1226–1239
Regenspurg S, Brand A, Peiffer S (2004) Formation and stability of schwertmannite in acidic mining lakes. Geochim Cosmochim Acta 68:1185–1197
Rodda DP, Wells JD, Johnson BB (1996) Anomalous adsorption of copper (II) on goethite. J Colloid Interface Sci 184:564–569
Saglam ES, Akçay M, Çolak DN, Bektas KI, Beldüz AO (2016) Generation of acid mine drainage around the Karaerik copper mine (Espiye, Giresun, NE Turkey): implications from the bacterial population in the Acısu effluent. Extremophiles 20:673–685
Schroth AW, Parnell RA (2005) Trace metal retention through the schwertmannite to goethite transformation as observed in a field setting, Alta mine, MT. Appl Geochem 20:907–917
Swedlund PJ, Webster JG (2001) Cu and Zn ternary surface complex formation with SO4 on ferrihydrite and schwertmannite. Appl Geochem 16:503–511
Waychunas GA, Xu N, Fuller CC, Davis JA, Bigham JM (1995) XAS study of AsO4 3− and SeO4 2− substituted schwertmannites. Physica B 208&209: 481–483, 208-209
Webster JG, Swedlund PJ, Webster KS (1998) Trace metal adsorption onto an acid mine drainage iron oxy hydroxyl sulfate. Environ Sci Technol 32:1361–1368
Xie YY, Lu GN, Ye H, Yang CF, Yi XY, Dang Z (2017) Role of dissolved organic matter in the release of chromium from schwertmannite: kinetics, repartition, and mechanisms. J Environ Qual 46:1088–1097
Funding
This work was financially supported by the National Natural Science Foundation of China (Nos.41330639 and 41720104004), the National Key Research and Development Program of China (2017YFD0801000), the National Key Technology Support Program of China (No. 2015BAD05B05), the Tip-top Scientific and Technical Innovative Youth Talents of Guangdong Special Support Program (No. 2015TQ01Z233), and the Guangdong Innovative and Entrepreneurial Research Team Program (No. 2014ZT05N200).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Li, J., Xie, Y., Lu, G. et al. Effect of Cu(II) on the stability of oxyanion-substituted schwertmannite. Environ Sci Pollut Res 25, 15492–15506 (2018). https://doi.org/10.1007/s11356-018-1773-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1773-0