Abstract
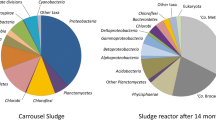
Methane is produced in anaerobic environments, such as reactors used to treat wastewaters, and can be consumed by methanotrophs. The composition and structure of a microbial community enriched from anaerobic sewage sludge under methane-oxidation condition coupled to denitrification were investigated. Denaturing gradient gel electrophoresis (DGGE) analysis retrieved sequences of Methylocaldum and Chloroflexi. Deep sequencing analysis revealed a complex community that changed over time and was affected by methane concentration. Methylocaldum (8.2%), Methylosinus (2.3%), Methylomonas (0.02%), Methylacidiphilales (0.45%), Nitrospirales (0.18%), and Methanosarcinales (0.3%) were detected. Despite denitrifying conditions provided, Nitrospirales and Methanosarcinales, known to perform anaerobic methane oxidation coupled to denitrification (DAMO) process, were in very low abundance. Results demonstrated that aerobic and anaerobic methanotrophs coexisted in the reactor together with heterotrophic microorganisms, suggesting that a diverse microbial community was important to sustain methanotrophic activity. The methanogenic sludge was a good inoculum to enrich methanotrophs, and cultivation conditions play a selective role in determining community composition.



Similar content being viewed by others
Abbreviations
- ANME:
-
Anaerobic methanotrophic archaea
- DAMO:
-
Denitrifying anaerobic methane oxidation
- DGGE:
-
Denaturing gradient gel electrophoresis
- OTU:
-
Operational taxonomic unit
- UASB:
-
Upflow anaerobic sludge blanket reactor
- VTS:
-
Volatile total solids
References
APHA (2005) Standard Methods for the Examination of Water and Wastewater Washington, DC, USA
Beck DAC, Kalyuzhnaya MG, Malfatti S, Tringe SG, Glavina del Rio T, Ivanova N, Lindstrom ME, Chistoserdova L (2013) A metagenomic insight into freshwater methane-utilizing communities and evidence for cooperation between the Methylococcaceae and the Methylophilaceae. PeerJ 1:e23
Cai Y, Zheng Y, Bodelier PLE, Conrad R, Jia Z (2016) Conventional methanotrophs are responsible for atmospheric methane oxidation in paddy soils. Nat Commun 7:11728
Cakir FY, Stenstrom MK (2005) Greenhouse gas production: a comparison between aerobic and anaerobic wastewater treatment technology. Water Res 39:4197–4203
Chistoserdova L, Lidstrom MEI (2013) Aerobic methylotrophic prokaryotes. In: DEF R, Thompson F, Lory S, Stackebrandt E (eds) The prokaryotes. Springer, New York
Chistoserdova L, Kalyuzhnaya MG, Lidstrom ME (2009) The expanding world of methylotrophic metabolism. Annu Rev Microbiol 63:477–499
Ding FL, Ding ZW, Lu Y,Z, Cheng SH, Zeng RJ (2016) Environmental evaluation of coexistence of denitrifying anaerobic methane oxidizing archaea and bacteria in a paddy field. Applied Microbiology Biotechnology 100:439–446
Dong J, Ding L, Wang X, Chi Z, Lei J (2015) Vertical profiles of community abundance and diversity of anaerobic methanotrophic archaea (ANME) and bacteria in a simple waste landfill in North China. Appl Biochem Biotechnol 175:2729–2740
Egli K, Langer C, Siegrist H-R, Zehnder AJB, Wagner M, van der Meer JR (2003) Community analysis of ammonia and nitrite oxidizers during start-up of nitritation reactors. Appl Environ Microbiol 69:3213–3222
Ettwig KF (2010) Nitrite-dependent methane oxidation. Radboud University Nijmegen, Netherlands 120 pp
Gupta RS, Mok A (2007) Phylogenomics and signature proteins for the alpha Proteobacteria and its main groups. BMC Microbiology 7:20
Hanson RS, Hanson TE (1996) Methanotrophic bacteria. Microbiol Mol Biol Rev 60:439–471
Hatamoto M, Kimura M, Sato T, Koizumi M, Takahashi M, Kawakami S, Araki N, Yamaguchi T (2014) Enrichment of denitrifying methane-oxidizing microorganisms using up-flow continuous reactors and batch cultures. PLoS One 9:e115823
Henkel T, Jackel U, Schnell S, Conrad R (2000) Molecular analyses of novel methanotrophic communities in forest soil that oxidize atmospheric methane. Applied Environmental Microbiology 66:1801–1808
Ho A, Kerckhof F-M, Luke C, Reim A, Krause S, Boon N, Bodelier PLE (2013a) Conceptualizing functional traits and ecological characteristics of methane-oxidizing bacteria as life strategies. Environ Microbiol Rep 5:335–345
Ho A, Vlaeminck SE, Ettwig KF, Schneider B, Frenzel P, Boon N (2013b) Revisiting methanotrophic communities in sewage treatment plants. Appl Environ Microbiol 79:2841–2846
Ho A, Angel R, Veraart AJ, Daebeler A, Jia Z, Kin SY, Kerckhof F-M, Boon N, Bodelier PLE (2016) Biotic interactions in microbial communities asmodulators of biogeochemical processes: methanotrophy as a model system. Front Microbiol 7:1285
Hu S, Zeng RJ, Burow LC, Lant P, Keller J, Yuan Z (2009) Enrichment of denitrifying anaerobic methane oxidizing microorganisms. Environ Microbiol Rep 1:377–384
Hug LA, Castelle CJ, Wrighton KC, et al. (2013) Genomic analyses constrain the distribution of metabolic traits across the Chloroflexi phylum and indicate roles in sediment carbon cycling. Microbiome 1:2–17
Huson DH, Auch AF, Qi J, Schuster SC (2007) MEGAN analysis of metagenomic data. Genome Res 17:377–386
Kalyuzhnaya MG, Puri AW, Lidstrom ME (2015) Metabolic engineeringin methanotrophic bacteria. Metab Eng 29:142–152
Kerckhof F-M, Courtens ENP, Geirnaert A, Hoefman A, Ho A, Vilchez-Vargas R (2014) Optimized cryopreservation of mixed microbial communities for conserved functionality and diversity. PLoS One 9:1–14
Knief C, Kolb S, Bodelier P,L, Lipski A, Dunfield PF (2006) The active methanotrophic community in hydromorphic soils changes in response to changing methane concentration. Environ Microbiol 8:21–333
Lee JH, Kim TG, Cho KS (2012) Isolation and characterization of a facultative methanotroph degrading malodor-causing volatile sulfur compounds. Jornal of Hazardous Materials 15:224–229
Luesken FA, van Alen TA, van der Biezen E, Frijters C, Toonen G, Kampman C, Hendrickx TL, Zeeman G, Temmink H, Strous M, Op den Camp HJ, Jetten MS (2011) Diversity and enrichment of nitrite-dependent anaerobic methane oxidizing bacteria from wastewater sludge. Appl Microbiol Biotechnol 92:845–854
Madigan MT, Martinko J, Dunlap PV, Clark DP (2010) Microbiologia de Brock. Editora Artmed, Porto Alegre 1160 pp
Meulepas RJW, Jagersma CG, Zhang Y, Petrillo M, Cai HZ, Buisman CJN, Stams AJM, Lens PNL (2010) Trace methane oxidation and the methane dependency of sulfate reduction in anaerobic granular sludge. FEMS Microbiol Ecol 72:261–271
Myhre G, Shindell D, Bréon F-M, Collins W, Fuglestvedt J, Huang J, Koch D, Lamarque D,J-F, Lee BM, Nakajima T, Robock A, Stephens G, Takemura T, Zhang H (2013) Climate change 2013: the physical science basis. In: Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, pp 731–738
Noll M, Frenzel P, Conrad R (2008) Selective stimulation of type I methanotrophs in a rice paddy soil by urea fertilization revealed by RNA-based stable isotope probing. FEMS Microbiol Ecol 65:125–132
Op den Camp HJM, Islam T, Stott MB, Harhangi HR, Hynes A, Schouten S, Jetten MS, Berkeland NK, Pol A, Dunfield PF (2009) Environmental, genomic and taxonomic perspectives on methanotrophic Verrucomicrobia. Environ Microbiol Rep 1:293–306
Oshkin IY, Beck DAC, Lamb AE, Tchesnokova V, Benuska G, McTaggart TL, Kalyuzhnaya MG, Dedysh SN, Lidstrom ME, Chistoserdova L (2015) Methane-fed microbial microcosms show differential community dynamics and pinpoint taxa involved in communal response. The ISME Journal 9:1119–1129
Raghoebarsing AA, Pol A, Pas-Schoonen KTVD, Smolders AJP, Ettwig KF, Rijpstra WIC, Schouten S, Damst JSS, Camp HJMO, Jetten MSM, Strous M (2006) A microbial consortium couples anaerobic methane oxidation to denitrification. Nature 440:918–921
Siljanen HM, Saari A, Krause S, Lensu A, Abell GC, Bodrossy L et al (2011) Hydrology is reflected in the functioning and community composition of methanotrophs in the littoral wetland of a boreal lake. FEMS Microbiol Ecol 75:430–445
Siniscalchi LAB, Vale IC, Dell’Isola J, Chernicharo CAL, Araújo JC (2015) Enrichment and activity of methanotrophic microorganisms from municipal wastewater sludge. Environ Technol 36:1–13
Souza CL, Chernicharo CA, Aquino SF (2011) Quantification of dissolved methane in UASB reactors treating domestic wastewater under different operating conditions. Water Science Technology 64:2259–2264
Strous M, Pelletier E, Mangenot S, Rattei T, Lehner A, Taylor MW et al. (2006) Deciphering the evolution and metabolism of an anammox bacterium from a community genome. Nature 440:790–794
Trotsenko YA, Murrell JC (2008) Metabolic aspects of aerobic obligate methanotrophy. Adv Appl Microbiol 63:183–229
Vos PD, Garrity GM, Jones D, Krieg NR, Ludwig W, Rainey FA, Schleifer KH, Whitman WB (2009) Bergey’s manual of systematic bacteriology, vol Vol. Three, The Firmicutes Second edn. Springer, New York 1422 pp
Yamada T, Sekiguchi Y, Hanada S, Imachi H, Ohashi A, Harada H, Kamagata Y (2006) Anaerolinea thermolimosa sp. nov., Levilinea saccharolytica gen. nov., sp. nov. and Leptolinea tardivitalis gen. nov., sp. nov., novel filamentous anaerobes, and description of the new classes Anaerolineae classis nov. and Caldilineae classis nov. in the. Int J Syst Evol Microbiol 56:1331–1340. doi:10.1099/ijs.0.64169-0
Acknowledgments
We wish to thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Fundação de Amparo a Pesquisa do Estado de Minas Gerais (FAPEMIG). We are also grateful to Dr. Emanuel F. Brandt for helping with the methane mass balance calculation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Gerald Thouand
Rights and permissions
About this article
Cite this article
Siniscalchi, L.A.B., Leite, L.R., Oliveira, G. et al. Illumina sequencing-based analysis of a microbial community enriched under anaerobic methane oxidation condition coupled to denitrification revealed coexistence of aerobic and anaerobic methanotrophs. Environ Sci Pollut Res 24, 16751–16764 (2017). https://doi.org/10.1007/s11356-017-9197-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9197-9