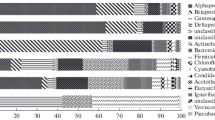
Abstract
Geothermal waters exploited in the southeastern region of Hungary are alkali-hydrogen-carbonate type, and beside the high amount of dissolved salt, they contain a variety of aromatic, heteroaromatic, and polyaromatic hydrocarbons. The majority of these geothermal waters used for heating are directed into surface waters following a temporary storage in reservoir lakes. The aim of this study was to gain information about the temporal and spatial changes of the water quality as well as the bacterial community composition of an alkaline and saline oxbow lake operated as reservoir of used geothermal water. On the basis of the water physical and chemical measurements as well as the denaturing gradient gel electrophoresis (DGGE) patterns of the bacterial communities, temporal changes were more pronounced than spatial differences. During the storage periods, the inflow, reservoir water, and sediment samples were characterized with different bacterial community structures in both studied years. The 16S ribosomal RNA (rRNA) gene sequences of the bacterial strains and molecular clones confirmed the differences among the studied habitats. Thermophilic bacteria were most abundant in the geothermal inflow, whereas the water of the reservoir was dominated by cyanobacteria and various anoxygenic phototrophic prokaryotes. In addition, members of several facultative anaerobic denitrifying, obligate anaerobic sulfate-reducing and syntrophic bacterial species capable of decomposition of different organic compounds including phenols were revealed from the water and sediment of the reservoir. Most of these alkaliphilic and/or halophilic species may participate in the local nitrogen and sulfur cycles and contribute to the bloom of phototrophs manifesting in a characteristic pink-reddish discoloration of the water of the reservoir.





Similar content being viewed by others
References
Abed RMM, Zein B, Al-Thukair A, de Beer D (2007) Phylogenetic diversity and activity of aerobic heterotrophic bacteria from a hypersaline oil-polluted microbial mat. Syst Appl Microbiol 30:319–330
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Bernal C, Vidal L, Valdivieso E, Coello N (2003) Keratinolytic activity of Kocuria rosea. World J Microbiol Biotechnol 19:255–261
Boldareva EN, Moskalenko AA, Makhneva ZK, Tourova TP, Kolganova TV, Gorlenko VM (2009) Rubribacterium polymorphum gen. nov., sp. nov., a novel alkaliphilic nonsulfur purple bacterium from an Eastern Siberian soda lake. Microbiology 78:732–740
Borsodi AK, Felföldi T, Máthé I, Bognár V, Knáb M, Krett G, Jurecska L, Tóth EM, Márialigeti K (2013a) Phylogenetic diversity of bacterial and archaeal communities inhabiting the saline Lake Red located in Sovata, Romania. Extremophiles 17:87–98
Borsodi AK, Knáb M, Czeibert K, Márialigeti K, Vörös L, Somogyi B (2013b) Planktonic bacterial community composition of an extremely shallow soda pond during a phytoplankton bloom revealed by cultivation and molecular cloning. Extremophiles 17:575–584
Castillo-Carvajal L, Sanz-Martin JL, Barragán-Huerta BE (2014) Biodegradation of organic pollutants in saline wastewater by halophilic microorganisms: a review. Environ Sci Pollut Res 21:9578–9588
Cerqueira VS, Hollenbach EB, Maboni F, Vainstein MH, Camargo FAO, Peralbo MCR, Bento FM (2011) Biodegradation potential of oily sludge by pure culture and mixed bacterial cultures. Bioresour Technol 102:11003–11010
Chen S, Liu X, Dong X (2005) Syntrophobacter sulfatireducens sp. nov., a novel syntrophic, propionate-oxidizing bacterium isolated from UASB reactors. Int J Syst Evol Microbiol 55:1319–1324
Das R, Kazy SK (2014) Microbial diversity, community composition and metabolic potential in hydrocarbon contaminated oily sludge: prospects for in situ bioremediation. Environ Sci Pollut Res 21:7369–7389
Dövényi P, Horváth F, Drahos D (2002) Hungary. In: Hurter, S., Haenel, R. (eds) Atlas of Geothermal Resources in Europe. Publication no. 17811 of the European Commission. Office for Official Publications of the European Communities, Luxembourg, pp. 36–38
Eichler B, Pfennig N (1986) Characterization of a new platelet-forming purple sulfur bacterium, Amoebobacter pedioformis sp. nov. Arch Microbiol 146:295–300
Guyoneaud R, Süling J, Petri R, Matheron R, Caumette P, Pfennig N, Imhoff JF (1998) Taxonomic rearrangements of the genera Thiocapsa and Amoebobacter on the basis of 16S rDNA sequence analyses, and description of Thiolamprovum gen. nov. Int J Syst Evol Microbiol 48:957–964
Harmsen HJM, Van Kuuk BLM, Plugge CM, Akkermans ADL, De Vos WM, Stams AJM (1998) Syntrophobacter fumaroxidans sp. nov., a syntrophic propionate-degrading sulfate-reducing bacterium. Int J Syst Evol Microbiol 48:1383–1387
Hudson JA, Morgan HW, Daniel RM (1989) Numerical classification of Thermus isolates from globally distributed hot springs. Syst Appl Microbiol 11:250–256
Kim OS, Cho YJ, Lee K, Yoon SH, Kim M, Na H, Park SC, Jeon YS, Lee JH, Yi H, Won S, Chun J (2012) Introducing EzTaxon-e: a prokaryotic 16S rRNA Gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62:716–721
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Kolekar YM, Pawar SP, Adav SS, Zheng L-Q, Li W-J, Shouche YS, Dastager SG, Kodam KM (2013) Alishewanella solinquinati sp. nov., isolated from soil contaminated with textile dyes. Curr Microbiol 67:454–459
Liu Y, Balkwill DL, Aldrich HC, Drake GR, Boone DR (1999) Characterization of the anaerobic propionate-degrading syntrophs Smithella propionica gen. nov., sp. nov. and Syntrophobacter wolinii. Int J Syst Evol Microbiol 49:545–556
Liu Y, Jiang J-T, Xu C-J, Liu Y-H, Song X-F, Li H, Liu Z-P (2012) Rheinheimera longhuensis sp. nov., isolated from a slightly alkaline lake, and emended description of genus Rheinheimera Brettar et al. 2002. Int J Syst Evol Microbiol 62:2927–2933
Lueders T, von Netzer F (2014) Primers: functional genes for anaerobic hydrocarbon degrading microbes. In: McGenity TJ et al (eds) Hydrocarbon and lipid microbiology protocols, springer protocols handbooks. Springer Verlag, Berlin
Manaia CM, Nogales B, Nunes OC (2003) Tepidiphilus margaritifer gen. nov., sp. nov., isolated from a thermophilic aerobic digester. Int J Syst Evol Microbiol 53:1405–1410
Margesin R, Schinner F (2001) Biodegradation and bioremediation of hydrocarbons in extreme environments. Appl Microbiol Biotechnol 56:650–663
Massol-Deya AA, Odelson DA, Hickey RF, Tiedje JM (1995) Bacterial community fingerprinting of amplified 16S and 16–23S ribosomal DNA sequences and restriction endonuclease analysis (ARDRA). In: Akkermans ADL, van Elsas JD, de Bruijn FJ (eds) Molecular microbial ecology manual. Kluwer Academic Publishers, Dordrecht, pp 3.3.2:1–3.3.2:8
Mechichi T, Stackebrandt E, Gad’on N, Fuchs G (2002) Phylogenetic and metabolic diversity of bacteria degrading aromatic compounds under denitrifying conditions, and description of Thauera phenylacetica sp. nov., Thauera aminoaromatica sp. nov., and Azoarcus buckelii sp. nov. Arch Microbiol 178:26–35
Muyzer G, Smalla K (1998) Application of denaturing gradient gel electrophoresis (DGGE) and temperature gradient gel electrophoresis (TGGE) in microbial ecology. Antonie Van Leeuwenhoek 73:127–141
Mwirichia R, Muigai AW, Tindall B, Boga HI, Stackebrandt E (2010) Isolation and characterisation of bacteria from the haloalkaline Lake Elmenteita, Kenya. Extremophiles 14:339–348
Németh A, Szirányi B, Krett G, Janurik E, Kosáros T, Pekár F, Márialigeti K, Borsodi AK (2014) Prokaryotic phylogenetic diversity of Hungarian deep subsurface geothermal well waters. Acta Microbiol Immunol Hung 61:363–377
Oshima T, Imahori K (1974) Description of Thermus thermophilus comb. nov., a nonsporulating thermophilic bacterium from a Japanese thermal spa. Int J Syst Bacteriol 24:102–112
Parshetti G, Kalme S, Saratale G, Govindwar S (2006) Biodegradation of Malachite Green by Kocuria rosea MTCC 1532. Acta Chim Slov 53:492–498
Parshetti GK, Telke AA, Kalyani DC, Govindwar SP (2010) Decolorization and detoxification of sulfonated azo dye methyl orange by Kocuria rosea MTCC 1532. J Hazard Mater 176:503–509
Piubeli F, Grossman MJ, Fantinatti-Garboggini F, Durrant LR (2014) Phylogenetic analysis of the microbial community in hypersaline petroleum produced water from the Campos Basin. Environ Sci Pollut Res 21:12006–12016
Podani J (2001) SYN-TAX 2000 Computer programs for data analysis in ecology and systematics (user’s manual, Scientia, Budapest)
Porter KG, Feig YS (1980) The use of DAPI for identifying and counting aquatic microflora. Limnol Oceanogr 25:943–948
Rusznyák A, Vladár P, Szabó G, Márialigeti K, Borsodi AK (2008) Phylogenetic and metabolic bacterial diversity of Phragmites australis periphyton communities in two Hungarian shallow soda lakes. Extremophiles 12:763–773
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Scholten E, Lukow T, Auling G, Kroppenstedt RM, Rainey FA, Diekmann H (1999) Thauera mechernichensis sp. nov., an aerobic denitrifier from a leachate treatment plant. Int J Syst Bacteriol 49:1045–1051
Song B, Palleroni NJ, Häggblom MM (2004) Isolation and characterization of diverse halobenzoate-degrading denitrifying bacteria from soils and sediments. Appl Environ Microbiol 66:3446–3453
Srinivas TNR, Kumar PA, Sasikala C, Spröer C, Ramana CV (2008) Rhodobacter ovatus sp. nov., a phototrophic alphaproteobacterium isolated from a polluted pond. Int J Syst Evol Microbiol 58:379–1383
Stackebrandt E, Goebel BM (1994) Taxonomic note: a place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. Int J Syst Evol Microbiol 44:846–849
Szanyi J, Kovács B (2010) Utilization of geothermal systems in South-East Hungary. Geothermics 39:357–364
Takai K, Komatsu T, Horikoshi K (2001) Hydrogenobacter subterraneus sp. nov., an extremely thermophilic, heterotrophic bacterium unable to grow on hydrogen gas, from deep subsurface geothermal water. Int J Syst Evol Microbiol 51:1425–1435
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Varsányi I, Kovács LÓ (2009) Origin, chemical and isotopic evolution of formation water in geopressured zones in the Pannonian Basin, Hungary. Chem Geol 264:187–196
Varsányi I, Matray JM, Kovács LÓ (1997) Geochemistry of formation waters in the Pannonian Basin (Southeast Hungary). Chem Geol 140:89–106
Varsányi I, Matray JM, Kovács LÓ (1999) Hydrogeochemistry in two adjacent areas in the Pannonian Basin (Southeast-Hungary). Chem Geol 156:25–39
Zhilina TN, Zavarzina DG, Kuever J, Lysenko AM, Zavarzin GA (2005) Desulfonatronum cooperativum sp. nov., a novel hydrogenotrophic, alkaliphilic, sulfate-reducing bacterium, from a syntrophic culture growing on acetate. Int J Syst Evol Microbiol 55:1001–1006
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Kenneth Mei Yee Leung
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
PCR primer sequences and thermal profiles used for the amplification of 16S rRNA gene fragments (DOCX 15.1 KB)
Supplementary Fig. 1
Bacterial cell numbers of the water of TO Reservoir at the two sampling sites (TO-0 and TO-4) in 2010 and 2011. (DOCX 17 kb)
Supplementary Fig. 2
Rarefaction curves for the different ARDRA patterns of 16S rRNA gene molecular clones from the influent, water and sediment of the TO Reservoir. (Abbreviations used: TO-IC influent clones, TO-WC water clones, TO-SC sediment clones) (DOCX 23 kb)
Rights and permissions
About this article
Cite this article
Borsodi, A.K., Szirányi, B., Krett, G. et al. Changes in the water quality and bacterial community composition of an alkaline and saline oxbow lake used for temporary reservoir of geothermal waters. Environ Sci Pollut Res 23, 17676–17688 (2016). https://doi.org/10.1007/s11356-016-6923-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6923-7