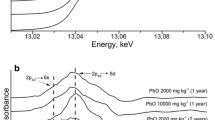
Abstract
Purpose The Pb absorption processes on a heavy textured calcareous soil, typical of central Italy, were studied using synchrotron X-ray absorption spectroscopy (XAS) in order to probe, at molecular scale, the structure and chemical nature of Pb in contaminated soils and achieve precise description of Pb ions localization into these contaminated soils.
Materials and methods In order to distinguish the role of the different components of soils in Pb retention, samples were prepared from the original soils removing the carbonate fractions, the organic matter, the metal oxides, or selecting the clay fractions. Then these samples were fortified with Pb simulating the natural interactions processes of heavy metal solutions with soils. The quantitative analysis of near edge (XANES) as well extended (EXAFS) regions of Pb LIII edge absorption spectra, in comparison with Pb XAS data of selected reference compounds, allowed the precise determination of local structure and chemical environment of Pb ions in these soil samples.
Results Four components were individuated as the major responsible of Pb retention in calcareous soils: the carbonates, the metal oxide surfaces, the organic matter, and the colloidal inorganic surfaces containing clay components. The structural analysis suggests that, within these experimental conditions, the Pb adsorbed on the soil is generally present as Pb hydroxide with poor crystallization degree. However, the presence of carbonates (CaCO3) induces the co-precipitation of PbCO3-like phases with some degree of crystallinity.



Similar content being viewed by others
Notes
Attempts to use the principal component analysis (Klementev 2001) to determine the chemical species in the samples failed to reach stable minima; therefore, the main components were determined as follows: for each soil sample spectrum (exp), we select a suitable reference signal to fit it (model). Then one other component is added to the model, and if the \(R^2_{\rm f\/it} = ({\rm exp} - {\rm model})^2/{\rm exp}^2\) decreases of at least 15%, it is accepted and one other component is selected.
References
Borda MJ, Sparks DL (2008) Kinetics and mechanisms of sorption–desorption in soils: a multiscale assessment. In: Violante A, Huang PM, Gadd GM (eds.) Biophysico-chemical processes of heavy metals and metalloids in soil environments. Wiley, New York, pp 75–124
Bres EF, Voegel JC, Barry JC, Waddington WG, Frank RM (1986) Feasibility study for the detection of lead substitution sites in the hydroxyapatite crystal structure using high resolution electron microscopy (HREM) at optimum focus. J Appl Crystallogr 19:168–173
Businelli D, Casciari F, Gigliotti G (2004) Sorption mechanisms determining Ni(II) retention by a calcareous soil. Soil Sci 169:355–362
Businelli D, Tombesi E (2009) Factors involved in the retention and release of lead by a central Italy soil: a stirred-flow approach. Soil Sci 174:380–384
Businelli D, Massaccesi L, Onofri A (2009) Evaluation of Pb and Ni mobility to groundwater in calcareous urban soils of Ancona, Italy. Water Air Soil Pollut 201:185–193
Dermatas D, Dadachov MS (2003) Rietveld quantification of montmorillonites in lead-contaminated soils. Appl Clay Sci 23:245–255
Harris W, White GN (2008): X-ray diffraction techniques for soil mineral identification. In: Ulery AL, Dress RD (eds) Methods of soil analysis, Part 5, SSSA Book Ser 5. SSSA, Madison, p 81-1
Jackson ML, Lim CH, Zelazny (1986) Oxydes, hydroxides, and aluminosilicates. In: Klute A (ed) Methods of soil analysis, part 1, 2nd edn. Physical and mineralogical methods. ASA, Madison, pp 101–150
Klementev KV (2001) Extraction of the fine structure from X-ray absorption spectra. J Phys D 34:209–217
Kunze GW, Dixon GB (1986) Pretreatment for mineralogical analysis. In: Klute A (ed) Methods of soil analysis, part 1, 2nd edn. Physical and mineralogical methods. ASA, Madison, pp 99–100
Lavkulich LM, Wiens JH (1970) Comparison of organic matter destruction by hydrogen peroxide and sodium hypochlorite and its effects on selected mineral constituents. Soil Sci Soc Am Proc 34:755–758
Lee PA, Citrin PH, Eisenberger P, Kincaid BM (1981) Extended X-ray absorption fine structure—its strengths and limitations as a structural tool. Rev Mod Phys 53:769–806
Manceau A, Bosset MC, Sarret G, Hazemann JL, Mench M, Cambier P, Prost R (1996) Direct determination of lead speciation in contaminated soils by EXAFS spectroscopy. Environ Sci Technol 30:1540–1552
Matocha CJ, Scheckel KJ, Sparks DL (2005) Kinetics and mechanisms of soil biogeochemical processes. In: Tabatabai MA, Sparks DL (eds) Chemical processes in soils. Soil Science Society of America, Madison, pp 309–342
Monesi C, Meneghini C, Bardelli F, Benfatto M, Mobilio S, Manju U and Sarma DD (2005) Local structure in LaMnO3 and CaMnO3 perovskites: a quantitative structural refinement of Mn-K edge XANES data. Phys Rev B 72:174104/1-174104/9
Moore DM, Reynolds RC (1989) X-ray diffraction and the identification and analysis of clay minerals. Oxford University Press, New York
Morin G, Juillot F, Ildefonse P, Calas G, Samama J-C, Chevallier P, Brown GE (2001) Mineralogy of lead in a soil developed on a Pb-mineralized sandstone (Largentiere, France). J Am Mineral 86:92–104
Roberts D, Nachtegaal M, Sparks DL (2005) Speciation of metals in soils. In: Tabatabai MA, Sparks DL (eds) Chemical processes in soils. Soil Science Society of America, Madison, pp 619–654
Sparks DL (1995) Environmental soil chemistry. Academic, San Diego
Sposito G (2008) The chemical composition of soils. In: The chemistry of soils. Oxford University Press, New York, pp 3–27
Strawn DG, Scheidegger AM, Sparks DL (1998) Kinetics and mechanisms of Pb(II) sorption and desorption at the aluminum oxide–water interface. Environ Sci Technol 32:2596–2601
Strawn DG, Sparks DL (1999) The use of XAFS to distinguish between inner- and outer-sphere lead adsorption complexes on montmorillonite. J Colloid Interface Sci 216:257–269
Strawn DG, Sparks DL (2000) Effects of soil organic matter on the kinetics and mechanisms of Pb(II) sorption and desorption in soil. Soil Sci Soc Am J 64:144–156
Xia K, Bleam W, Helmke PA (1997) Studies of the nature of Cu2 + and Pb2 + binding sites in soil humic substances using X-ray absorption spectroscopy. Geochim Cosmochim Acta 61:2211–2221
Zabinsky SI, Rehr JJ, Ankudinov A, Albers RC, Eller MJ (1997) Multiple-scattering calculations of X-ray absorption spectra. Phys Rev B 52:2995–3009
Acknowledgements
The research was partially supported by the Italian Ministry of University and Research (PRIN). The authors would like to thank Dr. F. Casciari for her help in preparing the investigated samples and Prof. G. Vlaic for his scientific support during the XAFS data collection and preliminary data analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Vera Slaveykova
Rights and permissions
About this article
Cite this article
Comaschi, T., Meneghini, C., Businelli, D. et al. XAS study of lead speciation in a central Italy calcareous soil. Environ Sci Pollut Res 18, 669–676 (2011). https://doi.org/10.1007/s11356-010-0412-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-010-0412-1