Abstract
Purpose
Little is known about cancer survivors’ perceptions of the prognostic impact of physical activity (PA) and the association between perceptions and PA. Therefore, the present narrative review aimed to identify cancer survivors' perceptions of the prognostic impact of PA, perception barriers, and association with PA.
Methods
A literature search was performed via PubMed and Scopus with search terms including “physical activity”, “cancer survivor”, and “perception” in November 2022. Inclusion criteria were studies that investigated cancer survivors’ perceptions of the prognostic impact of PA written in English. Studies were excluded for patients undergoing cancer treatment or advanced cancer patients only.
Results
Our search identified a total of 13,080 articles, and 14 studies met the inclusion criteria. The study sample size ranged from 8 to 1003 participants. Most studies showed that cancer survivors poorly understood the prognostic impact of PA. Perception barriers were cited as a lack of advice from health care professionals (HCPs). Several studies reported a positive association between the perception of the prognostic impact of PA and actual PA.
Conclusions
This narrative review revealed that most reports showed that cancer survivors have a poor perception of the prognostic impact of PA. In addition, the lack of advice from HCPs may also be contributing to the lack of perception among cancer survivors. Therefore, to increase the amount of PA among cancer survivors, it is necessary to improve the perception of the prognostic impact of PA in both cancer survivors and HCPs.
Similar content being viewed by others
Introduction
Physical activity (PA) is an effective non-pharmacological treatment for improving the physical and mental health of cancer survivors. Many studies have suggested that PA may improve muscle strength, cardiovascular fitness, body composition, bone health, sleep quality, fatigue, depression, and quality of life in cancer survivors [1,2,3,4,5,6,7]. Studies have demonstrated that PA has prognostic benefits in breast cancer and colorectal cancer survivors [8, 9]. For example, participation in PA after diagnosis showed a 24% reduction in recurrence and a 45% reduction in mortality in 2987 patients with stage I-III breast cancer [8]. Similarly, in 832 patients with stage III colorectal cancer, participation in PA after diagnosis showed a 40% reduction in recurrence and a 63% reduction in mortality [9]. In addition, biological mechanisms of exercise effects are postulated to modulate metabolism [10, 11], reduce inflammation [10, 12], and improve immune function [10, 13]. However, despite the evidence for prognostic benefits of PA, most cancer survivors poorly engage in PA [14,15,16]. Psychological interventions would be recommended to promote PA in cancer survivors [17]. Previous studies suggest that informing adults about the link between PA and preventing colorectal and breast cancer increased motivation and promoted PA [18,19,20]. Therefore, the perceived prognostic impact of PA on cancer survivors may promote PA.
Despite this, there is currently limited information about cancer survivors’ perceptions of the prognostic impact of PA [21]. Accordingly, we aimed to explore cancer survivors’ (1) perception of the prognostic impact of PA, (2) perception barriers, and (3) the relationship between perceptions and PA.
Methods
A literature search was conducted in PubMed and Scopus for articles published before November 2022. The search keywords included #1: (“Exercise” OR “exercises” OR “physical activity” OR “physical activities”) AND #2: (“Neoplasms” OR cancer OR oncology OR “cancer survivor” OR “cancer survivors” OR “cancer patient” OR “cancer patients”) AND #3: (“perception” OR “attitude” OR “recognition” OR “awareness” OR “knowledge” OR “experience”). The inclusion criteria were (1) surveys investigating the perception of the prognostic impact of PA (includes both qualitative and quantitative research) and (2) written in English. Studies were excluded for non-cancer populations, patients undergoing cancer treatment or advanced cancer patients only, for cannot calculate perception rate, surveys of other perceptions of PA effects, and studies not written in English. The studies excluded were case reports, editorials, literature reviews and systematic reviews, clinical guidelines, and conference papers. Perception levels were classified according to the perception rate of the prognostic impact of PA as follows: < 10% as low, > 50% as high, and the remainder as moderate. The perception rate is the percentage of the number of people who perceived the prognostic impact of PA on all participants. For studies that did not describe the perception rate, the author’s definition was used to calculate the perception rate and group the perception levels. In qualitative studies, the perception rate of the prognostic impact of PA was calculated from the percentage of all participants. In studies where the perception rate was described only on the Likert scale, the perception level was classified based on the median or mean value. Specifically, in the survey using a 5-point Likert scale, mean or median values of 1 to < 3 were classified as low, ≥ 4 to 5 as high, and the rest as moderate. In the survey using a 7-point Likert scale, mean or median values of 1 to < 4 were classified as low, ≥ 6 to 7 as high, and the rest as moderate. In studies that investigated both qualitative and quantitative research, the results of the qualitative studies were classified as a priority.
Results
Study characteristics
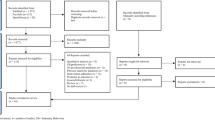
PRISMA 2020 flow diagram [22], adapted for the narrative review, is shown in Fig. 1. Our initial search identified 13,080 articles. After a title and abstract review, 59 studies were included, and after a full-text review, 45 studies were excluded, yielding 14 studies identified to have met the inclusion criteria. We included 14 studies published in English between 2013 and 2021 that enrolled 5109 adults (Tables 1, 2 and 3). The study sample sizes ranged from 8 to 1003 participants [23,24,25,26,27,28,29,30,31,32,33,34,35,36]. Eight studies were from North America: five from Canada [29, 30, 33,34,35], two from the USA [24, 31], and one study included the USA and Canada [36]. Three studies were from Europe: two from Germany [28, 32] and one from the United Kingdom [25]. Other studies included two from Australia [23, 26] and one from Nigeria [27]. The mean participant age was 44.9–68.0 years [23,24,25,26,27,28,29,30,31,32,33,34,35,36]. Breast cancer was the most reported type of cancer (four studies) [23, 24, 27, 31], followed by colorectal cancer (two studies) [25, 29], mixed cancer types (six studies) [26, 30, 32, 33, 35, 36], and two studies were unknown [28, 34]. Of the 14 studies, quantitative studies using questionnaires were the most common methods (12 studies) [24, 25, 27,28,29,30,31,32,33,34,35,36]. Quantitative research conducted in two studies used the open questionnaire or self-structured questionnaire [25, 27], nine studies used the closed questionnaire [24, 29,30,31,32,33,34,35,36], and one study used the opened and closed questionnaire [28]. The qualitative studies included three studies using semi-structured interviews [23, 24, 26].
Perception of the prognostic impact of PA among cancer survivors
Five of the 14 identified studies reported low perceptions of the prognostic impact of PA among cancer survivors (Table 1) [23,24,25,26,27]. Furthermore, nine of the 14 studies compared the perceptions of PA’s effect on improving cancer survivors’ physical and mental health and PA’s impact on prognosis [23,24,25, 27,28,29, 33, 34, 36]. Most reports showed that cancer survivors had a poor perception of the prognostic impact of PA compared to the effect of PA on improving physical and mental health [23,24,25, 27,28,29, 36].
Perception barriers
Two barriers to cancer survivor perceptions were identified [23, 24, 26]. One barrier was the lack of advice from healthcare professionals (HCPs) [24]. Hirschey R et al. [24] reported that cancer survivors did not learn the effect of PA on recurrence risk reduction from HCPs. The other barrier was that cancer survivors were themselves skeptical of the prognostic impact of PA. Cancer survivors denied the effectiveness of PA for recurrence [26] and felt that PA did not prevent cancer development [23].
Association between perception and behavior change or PA
Six of the 14 identified studies reported the relationship between PA and perceptions of the prognostic impact of PA among cancer survivors [28,29,30,31, 33, 36]. Eng et al. [33] showed that the perception of the effect of PA on survival contributed to achieving the PA guidelines (at least 150 min per week of moderate-intensity activity). Höh et al. [28] showed that nearly one in two subjects who answered that they regularly engaged in PA five or more times a week fully agreed that PA effectively reduced the risk of recurrence. In addition, perceptions of the prognostic impact of PA were positively associated with PA [29, 30, 36]. O’Neill et al. [31] reported that 18% of all participants perceived PA's effect as reducing cancer risk and were actually engaged in PA.
Discussion
To the best of our knowledge, this is the first study to review the perception of the prognostic impact of PA, perception barriers, and the relationship between perceptions and PA in cancer survivors. Our review revealed three major findings. First, most reports showed that cancer survivors poorly perceived the prognostic impact of PA. Second, the lack of advice from HCPs led to poor perception and engagement on PA among cancer survivors. Finally, multiple reports showed that cancer survivors’ PA perception was positively associated with actual PA.
PA guidelines for cancer survivors are published by the American Cancer Society [37]. However, cancer survivors poorly understand the PA guidelines [26, 38]. The results of our review are consistent with the results of previous reports on the lack of progress in implementing PA guidelines. Alternatively, most studies report that cancer survivors better perceived the PA’s effects on improving physical and mental functioning than the prognostic impact of PA. This finding suggests that education about the short-term benefits of PA has been effective. In future, it is necessary to educate cancer survivors regarding the long-term benefits of PA, including its impact on prognosis.
Advice from HCPs for PA is essential to increase cancer survivors’ perception. However, HCPs also poorly perceive the prognostic impact of PA [39,40,41]. Further, there is an association between the lack of PA promotion and low perception of PA [39,40,41]. Therefore, poor perception by HCPs may impact a lack of advice.
Cancer recurrence is a major concern for most cancer survivors [42]. Therefore, the positive impact of PA on prognosis may become a cancer survivors’ hope and promote behavior change. The health belief model (HBM) can explain behavior change among cancer survivors. The HBM is a behavior change theory model that has been widely used in health promotion and disease prevention studies [43, 44]. The HBM proposes that patients are more likely to engage in health-related behaviors when they perceive the threat of disease and believe that the benefits of the behavior outweigh the barriers [44]. Several previous studies have reported increased PA after HBM-based interventions for patients with type 2 diabetes [45, 46]. An overview of how HBM can be applied to promote behavior change in cancer survivors is shown in Fig. 2. The HBM suggests that when cancer survivors are educated about the prognostic impact of PA, they perceive threats to their prognosis, and the benefits of PA outweigh the barriers, thereby promoting behavior change. Therefore, a higher perception of the prognostic impact of PA among cancer survivors may increase PA through behavior change in HBM.
This review has several limitations. First, the literature search collected only literature written in English, which might have caused selection bias. Second, perception of the effects of PA was limited in comparison and integration because each study used different research methods. Third, studies that did not describe the perception rate were calculated by the author's definition, limiting the perception rate's interpretation. Finally, the population included some patients undergoing cancer treatment, which limited the interpretation of the perception of the effects of PA.
Based on the results of our review, we propose here methods to promote PA in cancer survivors (Fig. 3). The first is to educate HCPs on the importance of the prognostic impact of PA. The second is to encourage HCPs to advise cancer survivors on the prognostic impact of PA. The third is to enhance cancer survivors’ understanding by repeated counseling on the prognostic impact of PA. The fourth is to encourage cancer survivors to change their behavior to increase PA based on the HBM. The fifth is to follow-up longitudinally for the outcome of cancer survivors who increase PA. Finally, to inform HCPs and patients of the success story based on the accumulated experiences and analysis, thus generating a positive feedback cycle. Karvinen et al. [47] investigated the effect of an intervention of online learning of PA on oncology nurses. Exploratory data analysis revealed that PA counseling improved self-efficacy, but no significant change was observed in counseling practice or PA knowledge among cancer survivors. Therefore, further research is required on the education of HCPs on PA first to substantiate our hypothesis.
Conclusions
This narrative review revealed that most reports showed that cancer survivors have a poor perception of the prognostic impact of PA. In addition, the lack of advice from HCPs may also contribute to the lack of perception among cancer survivors. Higher perceptions were associated with higher PA. Therefore, to increase the levels of PA among cancer survivors, it is necessary to improve the perception of the prognostic impact of PA in both cancer survivors and HCPs.
Data availability
All data generated or analyzed during this study are included in this published article.
Code availability
Not applicable.
Abbreviations
- PA:
-
Physical activity
- HCPs:
-
Healthcare professionals
- HBM:
-
Health belief model
References
Fuller JT, Hartland MC, Maloney LT, Davison K (2018) Therapeutic effects of aerobic and resistance exercises for cancer survivors: a systematic review of meta-analyses of clinical trials. Br J Sports Med 52:1311. https://doi.org/10.1136/bjsports-2017-098285
Dieli-Conwright CM, Courneya KS, Demark-Wahnefried W, Sami N, Lee K, Sweeney FC et al (2018) Aerobic and resistance exercise improves physical fitness, bone health, and quality of life in overweight and obese breast cancer survivors: a randomized controlled trial. Breast Cancer Res 20:124. https://doi.org/10.1186/s13058-018-1051-6
Bruno E, Roveda E, Vitale J, Montaruli A, Berrino F, Villarini A et al (2018) Effect of aerobic exercise intervention on markers of insulin resistance in breast cancer women. Eur J Cancer Care (Engl) 27:e12617. https://doi.org/10.1111/ecc.12617
Roveda E, Vitale JA, Bruno E, Montaruli A, Pasanisi P, Villarini A et al (2017) Protective effect of aerobic physical activity on sleep behavior in breast cancer survivors. Integr Cancer Ther 16:21–31. https://doi.org/10.1177/1534735416651719
Mustian KM, Alfano CM, Heckler C, Kleckner AS, Kleckner IR, Leach CR et al (2017) Comparison of pharmaceutical, psychological, and exercise treatments for cancer-related fatigue: a meta-analysis. JAMA Oncol 3:961–968. https://doi.org/10.1001/jamaoncol.2016.6914
Gerritsen JK, Vincent AJ (2016) Exercise improves quality of life in patients with cancer: a systematic review and meta-analysis of randomised controlled trials. Br J Sports Med 50:796–803. https://doi.org/10.1136/bjsports-2015-094787
Brown JC, Huedo-Medina TB, Pescatello LS, Ryan SM, Pescatello SM, Moker E, LaCroix JM, Ferrer RA, Johnson BT (2012) The efficacy of exercise in reducing depressive symptoms among cancer survivors: a meta-analysis. PLoS One 7:e30955. https://doi.org/10.1371/journal.pone.0030955
Holmes MD, Chen WY, Feskanich D, Kroenke CH, Colditz GA (2005) Physical activity and survival after breast cancer diagnosis. JAMA 293:2479–2486. https://doi.org/10.1001/jama.293.20.2479
Meyerhardt JA, Heseltine D, Niedzwiecki D, Hollis D, Saltz LB, Mayer RJ et al (2006) Impact of physical activity on cancer recurrence and survival in patients with stage III colon cancer: findings from CALGB 89803. J Clin Oncol 24:3535–3541. https://doi.org/10.1200/JCO.2006.06.0863
Friedenreich CM, Ryder-Burbidge C, McNeil J (2021) Physical activity, obesity and sedentary behavior in cancer etiology: epidemiologic evidence and biologic mechanisms. Mol Oncol 15:790–800. https://doi.org/10.1002/1878-0261.12772
Brown JC, Rickels MR, Troxel AB, Zemel BS, Damjanov N, Ky B et al (2018) Dose–response effects of exercise on insulin among colon cancer survivors. Endocr Relat Cancer 25:11–19. https://doi.org/10.1530/erc-17-0377
Brown JC, Zhang S, Ligibel JA, Irwin ML, Jones LW, Campbell N et al (2020) Effect of exercise or metformin on biomarkers of inflammation in breast and colorectal cancer: a randomized trial. Cancer Prev Res (Phila) 13:1055–1062. https://doi.org/10.1158/1940-6207.CAPR-20-0188
Fairey AS, Courneya KS, Field CJ, Bell GJ, Jones LW, Mackey JR (2005) Randomized controlled trial of exercise and blood immune function in postmenopausal breast cancer survivors. J Appl Physiol 98:1534–1540. https://doi.org/10.1152/japplphysiol.00566.2004
Hyland KA, Jacobs JM, Lennes IT, Pirl WF, Park ER (2018) Are cancer survivors following the national comprehensive cancer network health behavior guidelines? An assessment of patients attending a cancer survivorship clinic. J Psychosoc Oncol 36:64–81. https://doi.org/10.1080/07347332.2017.1399193
Mason C, Alfano CM, Smith AW, Wang C-Y, Neuhouser ML, Duggan C et al (2013) Long-term physical activity trends in breast cancer survivors. Cancer Epidemiol Biomarkers Prev 22:1153–1161. https://doi.org/10.1158/1055-9965.EPI-13-0141
Sabiston CM, Brunet J, Vallance JK, Meterissian S (2014) Prospective examination of objectively assessed physical activity and sedentary time after breast cancer treatment: sitting on the crest of the teachable moment. Cancer Epidemiol Biomarkers Prev 23:1324–1330. https://doi.org/10.1158/1055-9965.EPI-13-1179
Pudkasam S, Polman R, Pitcher M, Fisher M, Chinlumprasert N, Stojanovska L et al (2018) Physical activity and breast cancer survivors: importance of adherence, motivational interviewing and psychological health. Maturitas 116:66–72. https://doi.org/10.1016/j.maturitas
Graham SP, Prapavessis H, Cameron LD (2006) Colon cancer information as a source of exercise motivation. Psychol Health 21:739–755. https://doi.org/10.1080/14768320600603554
Jalleh G, Donovan RJ, Slevin T, Dixon H (2005) Efficacy of bowel cancer appeals for promoting physical activity. Health Promot J Austr 16:107–109. https://doi.org/10.1071/he05107
Jalleh G, Donovan RJ, Slevin T, Lin CY (2009) Efficacy of breast cancer appeals for promoting physical activity. J Cancer Educ 24:33–35. https://doi.org/10.1080/08858190802664313
Elshahat S, Treanor C, Donnelly M (2021) Factors influencing physical activity participation among people living with or beyond cancer: a systematic scoping review. Int J Behav Nutr Phys Act 18:50. https://doi.org/10.1186/s12966-021-01116-9
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Short CE, James EL, Plotnikoff RC (2013) How social cognitive theory can help oncology-based health professionals promote physical activity among breast cancer survivors. Eur J Oncol Nurs 17:482–489. https://doi.org/10.1016/j.ejon.2012.10.009
Hirschey R, Docherty SL, Pan W, Lipkus I (2017) Exploration of exercise outcome expectations among breast cancer survivors. Cancer Nurs 40:E39-46. https://doi.org/10.1097/NCC.0000000000000362
Fisher A, Wardle J, Beeken RJ, Croker H, Williams K, Grimmett C (2016) Perceived barriers and benefits to physical activity in colorectal cancer patients. Support Care Cancer 24:903–910. https://doi.org/10.1007/s00520-015-2860-0
Hardcastle SJ, Galliott M, Lynch BM, Nguyen NH, Cohen PA, Mohan GR et al (2019) “If I had someone looking over my shoulder…”: Exploration of advice received and factors influencing physical activity among non-metropolitan cancer survivors. Int J Behav Med 26:551–561. https://doi.org/10.1007/s12529-019-09808-0
Odikpo LC, Chiejina EN (2021) Knowledge and perceived benefits of exercise among women with breast cancer in tertiary hospitals in Delta State. Breast Dis 40:101–108. https://doi.org/10.3233/BD-201062
Höh J-C, Schmidt T, Hübner J (2017) Physical activity among cancer survivors—what is their perception and experience? Support Care Cancer. https://doi.org/10.1007/s00520-017-3977-0
Speed-Andrews AE, McGowan EL, Rhodes RE, Blanchard CM, Culos-Reed SN, Friedenreich CM et al (2014) Identification and evaluation of the salient physical activity beliefs of colorectal cancer survivors. Cancer Nurs 37:14–22. https://doi.org/10.1097/ncc.0b013e3182813972
Price J, Barrett-Bernstein M, Wurz A, Karvinen KH, Brunet J (2021) Health beliefs and engagement in moderate-to-vigorous-intensity physical activity among cancer survivors: a cross-sectional study. Support Care Cancer 29:477–484. https://doi.org/10.1007/s00520-020-05515-9
O’Neill SC, DeFrank JT, Vegella P, Richman AR, Henry LR, Carey LA, Brewer NT (2013) Engaging in health behaviors to lower risk for breast cancer recurrence. PLoS One 8:e53607. https://doi.org/10.1371/journal.pone.0053607
Roth I, Dubois C, Schmidt T, Hübner J (2020) Attitude of cancer patients from online self-help groups towards physical activity. J Cancer Res Clin Oncol 146:1583–1590. https://doi.org/10.1007/s00432-020-03190-1
Eng L, Pringle D, Su J, Shen X, Mahler M, Niu C et al (2018) Patterns, perceptions, and perceived barriers to physical activity in adult cancer survivors. Support Care Cancer 26:3755–3763. https://doi.org/10.1007/s00520-018-4239-5
Liu SY, Lu L, Pringle D, Mahler M, Niu C, Charow R et al (2019) Impact of immigration status on health behaviors and perceptions in cancer survivors. Cancer Med 8:2623–2635. https://doi.org/10.1002/cam4.2079
Niu C, Eng L, Qiu X, Shen X, Espin-Garcia O, Song Y et al (2015) Lifestyle behaviors in elderly cancer survivors: a comparison with middle-age cancer survivors. J Oncol Pract 11:e450–e459. https://doi.org/10.1200/JOP.2014.002287
Karvinen K, Vallance J (2015) Breast and colon cancer survivors’ expectations about physical activity for improving survival. Oncol Nurs Forum 42:527–533. https://doi.org/10.1188/15.ONF.527-533
Rock CL, Doyle C, Demark-Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL et al (2012) Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin 62:243–274. https://doi.org/10.3322/caac.21142
Hardcastle SJ, Maxwell-Smith C, Kamarova S, Lamb S, Millar L, Cohen PA (2018) Factors influencing non-participation in an exercise program and attitudes towards physical activity amongst cancer survivors. Support Care Cancer 26:1289–1295. https://doi.org/10.1007/s00520-017-3952-9
Karvinen KH, DuBose KD, Carney B, Allison RR (2010) Promotion of physical activity among oncologists in the United States. J Support Oncol 8:35–41
Karvinen KH, McGourty S, Parent T, Walker PR (2012) Physical activity promotion among oncology nurses. Cancer Nurs 35:E41–E48. https://doi.org/10.1097/NCC.0b013e31822d9081
Park J-H, Oh M, Yoon YJ, Lee CW, Jones LW, Kim SI et al (2015) Characteristics of attitude and recommendation of oncologists toward exercise in South Korea: a cross sectional survey study. BMC Cancer 15:249. https://doi.org/10.1186/s12885-015-1250-9
Foster C, Wright D, Hill H, Hopkinson J, Roffe L (2009) Psychosocial implications of living 5 years or more following a cancer diagnosis: a systematic review of the research evidence. Eur J Cancer Care (Engl) 18:223–47. https://doi.org/10.1111/j.1365-2354.2008.01001.x
Carpenter CJ (2010) A meta-analysis of the effectiveness of health belief model variables in predicting behavior. Health Commun 25:661–669. https://doi.org/10.1080/10410236.2010.52190
Champion V, Skinner C (2008) The health belief model. Health behavior and health education: theory, research, and practice. In: Glanz K, Rimer BK, Jossey-Bass VK (eds) Book The health belief model health behavior and health education: theory research and practice. Jossey-Bass, San Francisco
Abdulah DM, Hassan AB, Saadi FS, Mohammed AH (2018) Impacts of self-management education on glycaemic control in patients with type 2 diabetes mellitus. Diabetes Metab Syndr 12:969–975. https://doi.org/10.1016/j.dsx.2018.06.007
Shabibi P, Zavareh MSA, Sayehmiri K, Qorbani M, Safari O, Rastegarimehr B et al (2017) Effect of educational intervention based on the health belief model on promoting self-care behaviors of type-2 diabetes patients. Electron Physician 9:5960–5968. https://doi.org/10.19082/5960
Karvinen K, Balneaves L, Courneya K, Perry B, Truant T, Vallance J (2017) Evaluation of online learning modules for improving physical activity counseling skills, practices, and knowledge of oncology nurses. Oncol Nurs Forum 44:729–738. https://doi.org/10.1188/17.onf.729-738
Funding
This work was supported by the Japan Agency for Medical Research and Development (AMED) under the grant number 21ck0106673h0001.
Author information
Authors and Affiliations
Contributions
Conceptualization, KS and TN and TO and YY and MK and RS and KS; methodology, KS and TN and TO and YY and MK and RS and KS; writing—original draft preparation, KS and TN and TO and YY and MK and RS and KS; writing—review and editing, KS and TN and TO and YY and MK and RS and KS and TI and NM and RU and AS and TN and HF. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
K.S. (Katsuyoshi Suzuki) and T.O. and Y.Y. and M.K. and R.S. and K.S. (Keisuke Suzuki) and T.I. and N.M. and R.U. and A.S. and T.N. (Tamae Nishizawa) and H.F have nothing to disclose. T.N. (Tateaki Naito) received a lecture fee from ONO Pharmaceutical CO. Ltd and Helsinn Healthcare SA and research funding from Otsuka Pharmaceutical CO. Ltd. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results. The authors report no other conflicts of interest in this work.
Ethical approval
This project is a narrative review, and thus, ethics approval is not applicable.
Consent to participate
Not applicable to this narrative review.
Consent to publish
Not applicable to this narrative review.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Suzuki, K., Naito, T., Okayama, T. et al. Perception of the prognostic impact of physical activity among cancer survivors: a narrative review. Sport Sci Health 19, 371–380 (2023). https://doi.org/10.1007/s11332-022-01033-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-022-01033-9