Abstract
Background
Strength training as neuromuscular electrical stimulation (NMES) is effective in counteracting age-related postural impairments in elderly. However, it remains unknown whether training different muscle groups would entail in different adaptations.
Aim
To evaluate the effect of NMES training on balance function in healthy inactive elderly, targeting paravertebral muscles, in addition to thigh muscles.
Methods
Eleven healthy elderly were trained with NMES for 8 week allocated to combined training (CT: quadriceps and lumbar paraspinal muscles) or to quadriceps training (QT), after completing lifestyle questionnaire and spine morphology measurements. Functional balance, static stabilometry, and isometric strength tests were assessed before and after the training period.
Results and conclusion
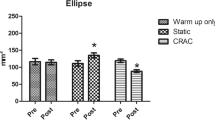
The CT group showed a greater improve in static balance control, i.e., reducing the CEA of the CoP displacement from 99 ± 38 to 76 ± 42 mm2 (Cohen’s d = 0.947). Benefits for improving static balance through CT might be due to NMES training, which increases spinal stabilization.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Healthy skeletal muscle promotes a broad range of functions, including the maintenance of postural control, mobility, strength, and metabolic functions, which makes it an essential tissue for life [1,2,3]. Severe sarcopenia is known to increase the chances of developing a disability and premature death [4, 5]. Of particular concern in old age is the high incidence of falls, which can lead to devastating consequences [6]. Although postural function impairment is not viewed as a typical risk factor for falls, associations between posture and body balance or falls in the elderly have been shown [7, 8].
The postural output is closely associated with lower limbs’ strength and power across all subjects [9]. Recently, Andrade and colleagues pointed out that the age-related impairment of postural control is mainly due to lower limb strength decline and not due to the changes in sensorial integration [10]. Furthermore, a growing body of evidence suggests that age-related postural changes in spine morphology contribute to the increased postural instability which in turn entails the increased risk of falls in the elderly [11,12,13]; for instance, Beserra Da Silva and colleagues [14] demonstrated on postmenopausal women that trunk extension strength is a predictor of falls. Indeed, to counteract the fall risk associated with age-related osteo-muscular diseases, in addition to the strengthening of lower limb muscles [15], and particularly knee extensors [16], it has been suggested to targeting trunk and core muscles [17].
Neuromuscular electrical stimulation (NMES) previously proved to be effective in attenuating muscle mass and strength in both healthy adults [18] as well in people with muscle weakness who cannot perform volitional training [19]. However, it is still missing evidence about a possible enhancement of postural adaptations due to training on paravertebral muscles, in addition to the usual training of large muscle groups of lower limbs. It has been argued that supervised protocols of resistance training in older adults improved balance to a greater extent, rather than unsupervised protocols [20]. Therefore, NMES training may be prospectively useful for targeting strength protocols in elderly in a self-training modality in those conditions where active and supervised protocols are hard to implement.
Hence, in this study, we aimed to evaluate the effect of paravertebral and thigh muscles NMES training on balance function in healthy older people. The authors hypothesized that targeting at lumbar paravertebral muscles with electrical stimulation in elderly, which is difficult to stimulate with active protocols in elderly in addition to thigh muscles, will enhance the training-related benefits to postural health. The authors also hypothesized that changes in postural control in response to 8-week of NMES training would be detectable by stabilometry.
Materials and methods
Participants
Volunteers were recruited from the community according to the inclusion criteria: (a) > 65 years old, (b) availability to take part in a 8-week lasting training plan in our laboratory, (c) being able to carry out stabilometry assessment with closed eyes. The exclusion criteria were irregular ECG, osteoarticular pathologies, mild to medium cardio-circulatory pathologies, cancer, non-controlled hypertension, metabolic, psychiatric, respiratory, neuromuscular or genetic disease, and sarcopenia. Only one participant (the one coded as Q + Lm1 in the tables) suffered from a vestibular disease in the past. The current report refers to an experimental group of 11 Italian volunteers, aged 66–74 years, retired, living in Abruzzo region, Italy (see Table 1 for details). Our participants were not involved in physical activity or exercise protocols. None of them was taking any medication targeting muscle function. The study was approved by the Ethics Committee of the University “G. d’Annunzio” (n.16 of 05/09/2019), conforming to the ethical standards set by the Declaration of Helsinki and later amendments [21]. Informed consent was obtained from all individual participants included in the study. A medical anamnesis was applied to all the participants by a medical doctor prior to the effective recruitment.
Materials
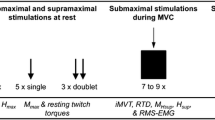
The neuromuscular activation (warm-up) was performed pedaling on a stationary bike (XTPRO Bike600, Technogym, Gambetolla, Italy). The transcutaneous electrical stimulation was delivered through a set of four fully gelled electrodes with a contact area of 5 × 5 cm2 using a customizable NMES device (Genesy 1200 Pro; Globus Srl, Codogne, Italy). Spine morphology was assessed through the 6TVC G.O.A.L.S. (Global Opto-electronic Approach for Locomotion & Spine) (Bioengineering & Biomedicine Company S.r.l., Italy) stereo-photogrammetric opto-electronic system, derived from Optitrack System (Natur- alPoint Inc. USA). 3D parametric biomechanical model was processed with the commercial software package ASAP 3D Skeleton Model (Bioengineering & Biomedicine Company S.r.l. Italy). They were then tested for spine morphology in the external Laboratory of the “Bioengineering and Biomedicine Company”. In this study, a stabilometric and posturometric platform with related software (Lizard 3.01, Lizard S.r.l, Perugia, Italy) was used to record and report the CoP movements in the anterior–posterior (y) and lateral (x) directions. To assess bilateral isometric strength of quadriceps, we used Maximum Voluntary Isometric Contraction (MVIC) method on a leg extension machine (Nessfit NMI 1000, Bcube, Italy) equipped with a dynamometer (Tesys 800, Globus, Italy). The percentage of Fat Mass (FM%) was estimated based on the method of Jackson and Pollock [22]; skinfold thickness was measured with an accurate plicometer (Holtain Ltd, UK).
Physical activity (PA) levels were assessed by the Physical Activity Scale for Elderly (PASE) questionnaire. It is a brief tool that focuses on three domains (leisure, household and work-related), assessing the PA in older people over a 1-week time frame. It includes activities common to most oldest adults, providing a more comprehensive assessment of the overall physical activity of the elderly. In this study, the validated version of the original questionnaire adapted to Italian culture was used [23]. Health-Related Quality of Life (HRQoL) was assessed by the Short Form-12 (SF-12) questionnaire. It is a 12-item questionnaire that assesses generic health outcomes from the patient’s perspective, including the impact of possible illnesses on a wide range of functional domains. In this study, the Italian version of the SF-12 questionnaire was used, and data were scored according to Ware et al. [24] to calculate Physical and Mental Component Summary (respectively, PCS and MCS) [25].
Research design
This randomized non-controlled trial represents a pilot study, as a sub-set of the project “A.M.A. la terza età” (translation: “love the old age”), which addresses the exercise-related adaptations in elderly and the determinants of muscle healthy aging. Before starting with the training, participants underwent an inclusive check for sarcopenic status, controlling for walking speed and handgrip strength [26]. The eleven volunteers were randomly assigned to two groups and trained with NMES throughout an eight-week period to either a combined training (Q + L group: training of quadriceps and lumbar paraspinal muscles; six participants, age: 69.2 ± 1.5 years, BMI: 26.2 ± 2.3 kg/m2) or to a training of quadriceps only (Q group; five participants, age: 70.4 ± 3.5 years, BMI: 28.4 ± 1.7 kg/m2). All participants were tested at the beginning and the end of the 8-week training period to examine the static balance adaptations using stabilometry and the functional balance using single-leg static balance test (or One-Leg Stance test, hereinto Flamingo test) and stepping test (or Fukuda-Unterberger stepping test, hereinto Fukuda test). In addition, MVIC was measured to assess the leg strength of volunteers. At baseline, participants completed PASE and SF-12 questionnaire and underwent anthropometric measurement. In particular, body mass index (BMI) was calculated as weight-to-square height ratio, and %FM was estimated as reported in the "Materials" section. Physical activity levels, wellness components, and spine morphology were assessed to characterize the sample. In addition, we measured spine morphology as the assessment of structural components of posture is considered as a valid component of physical functions and health in elderly individuals [27]. At the end of the training, we provided the participants with a report of their results, including the baseline measures. On these bases, we suggested them the current recommendation for Active Aging [28] and specific tips based on our results.
Procedure
Intervention: NMES training
The NMES training consisted of three sessions per week for an 8-week period. All volunteers performed 5 min of neuromuscular activation (warm-up) pedaling on a stationary bike. NMES training of quadriceps muscle group was set according to our laboratory protocol previously described in [29], while NMES training of lumbar paraspinal muscles (targeting the lumbar multifidus) was experimentally set according to the previous work by the groups of Baek and Kim [30, 31]. In both cases, electrodes were placed following skin preparation with an alcohol wipe.
For the NMES of the quadriceps, volunteers were seated in a relaxed position with a knee joint fixed at a 90° knee extension. After cleaning the skin with an alcohol wipe, two active electrodes were placed over the motor points of vastus lateralis and vastus medialis previously identified by palpation, and two dispersive electrodes were placed approximately 5 cm below the inguinal crease. The NMES training lasted 18 min in total. Rectangular wave pulsed currents (75 Hz frequency, 400 µs pulse width) were delivered with a rise time of 1.5 s, a steady tetanic stimulation time of 4 s, and a fall time of 0.75 s (total duration of the contraction: 6.25 s) with a rest interval of 20 s between contractions.
Volunteers undergoing the combined NMES training followed the quadriceps NMES by 15 min NMES of lumbar paraspinal muscles. Four electrodes were placed bilaterally at the vertebral levels of L4 and L5, as described by the groups of Baek and Kim [30, 31], identified previously by iliac crest palpation. Volunteers were seated in a relaxed position, which was empirically set based on a prior set of trials involving different volunteers trying different positions. Biphasic wave pulsed currents (50 Hz frequency, 300 µs pulse width) were delivered with a ramp-up for 1 s, a steady tetanic stimulation time of 8 s, and a ramp down for 1 s (total duration of the contraction: 10 s) with a rest interval of 10 s between contractions. Frequency and time were applied according to previous work by [30, 31]. The transcutaneous electrical nerve stimulation (TENS) [32] was applied for the rest interval. This novel setting was applied to provide a more comfortable experience for the elderly volunteers, which was again previously tested empirically.
For both NMES training protocols, the intensity was monitored during each session and gradually increased to reach the maximum tolerable intensity, corresponding to the individual’s pain threshold. After the first instructed trials, volunteers were allowed to freely adjust the intensity during the training session without feeling discomforts. Maximum intensities reached at the end of the NMES training ranged from 25 to 35 mA and from 35 to 45 mA for quadriceps and lumbar multifidus, respectively.
Variables: spine morphology measurements
We used the method described in [33] and [34]. In brief, recordings were based on a 6TV cameras (resolution 1.3 Mp, 120 fps) stereo-photogrammetric opto-electronic system. The calibrated acquisition volume was 3 × 3 × 2 m with a computed mean error range of 0.3–0.4 mm. For data processing, a complete 3 day parametric biomechanical human skeleton model has been implemented. It is based on 27 passive retro-reflective markers accurately positioned and glued on the human body, referred to anatomical landmarks identified by palpation [35]. Participants were required to maintain the neutral standing posture, with the upper arms relaxed along the side of the body, eyes looking directly ahead in the horizontal plane, feet apart at about pelvis width, and heels aligned on a line parallel to the frontal plane. At least five subsequent two-second lasting acquisitions at 120 Hz sampling rate have been recorded. All measurements were taken between 12 noon and 7.00 pm. The subjects were asked to avoid any intensive training and/or hard physical activity before the postural assessment. For the current study, we used four quantitative biomechanical parameters. In the frontal plane, we used the average frontal offsets (ASO) and the average frontal global offsets (AGO). These are the computed means of horizontal distances of each labeled spine landmark with respect to: the vertical axis passing by S3 (ASO), and to the vertical axis passing through the mid-point between the heels (AGO). In the sagittal plane, we used the kyphosis angle (KA) and the lordosis angle (LA), as the computed angles in the identified kyphosis and lordosis, properly identified according to real spine curvature spatial changes at the limit-vertebrae and not strictly limited to the thoracic and lumbar anatomical regions.
Variables: static stabilometry
During the stabilometry, the room was silent, and the assessment was performed with the volunteer standing barefoot on the stabilometric platform (Lizard 3.01®, Italy). Arms were held at the sides, and volunteers stood still focusing on a target located directly ahead on the white wall, approximately 1.5 m apart. Heels were positioned apart, forming a 30° angle as described in [36], touching the baseline with the second metatarsal and the center of the heel of each foot on each of the two 15° reference lines. The standardization was provided by positioning the lateral malleolus perpendicularly to the smaller reference line (see Fig. 1). The studied stance was bipedal with the eyes open (EO) and eyes closed (EC), which was tested for 51.2 s. The parameters were: Path Length (PL), Mean Path Velocity (MVP), Confidence Ellipse Area (CEA), Variance of Speed (Var S), Load Distribution Difference (LDD), Confidence Ellipse Axis Ratio (AP/ML Range Ratio). Romberg Index (RI) of PL, MVP and CEA were calculated as (EC/EO) × 100. The test was performed only once [37], and the data were recorded by the Lizard processing software (Lizard 3.01®, Italy).
Variables: functional balance measurements
The Flamingo test was applied following literature [38]. To be specific, volunteers were asked to stand freely on one foot for as long as possible, with the other foot raised, focusing on a spot on the wall at eye level in front of him with eyes open. The investigator interrupted the test after 30 s, or if the subject touched the floor with the raised leg. Compensatory arm movements were accepted during the test. To prevent falls or injuries, an investigator stood close to the volunteer throughout the experimental session. The investigator used a stopwatch to measure the amount of time the subject was able to stand on one limb. The best time of three trials was recorded for each limb.
The Fukuda test was applied following literature [39]. To be specific, volunteers stood in the center of one circle (drawn on the ground) with feet close together. They were asked to raise one leg and then the other and to step in place at normal walking speed, with eyes closed. Both arms were stretched in front of the individual and raised to 90°. When 50 steps were completed, volunteers were asked to remain standing, and the angle of rotation and distance of displacement were measured. The angle of rotation was the amount of turning of the body around its vertical axis. The distance of displacement was the distance the individual moved from the start position.
Variables: maximum bilateral isometric contraction or leg strength test
The MVIC test was performed according to a previous study [40]. Briefly, volunteers sat on a leg extension machine (Nessfit NMI 1000, Bcube, Italy) equipped with a dynamometer (Tesys 800, Globus, Italy) with knee and hip joint angle positioned at 90°, fastened to the chair, and they were required to push as hard as possible for 5 s. The test was carried out three times with recovery time > 2 min, and the best performance was recorded and used for later statistical analysis.
Statistics
The statistical analysis was carried out using GraphPad Prism Software, version 8 (GraphPad Software, La Jolla, USA), and the R-based open-source software Jamovi Version 1.2.5.0 (retrieved from https://www.jamovi.org). PL, MVP, CEA, Var S, LDD, and AP/ML Range Ratio, both in EO and EC condition were tested for the comparison (pre vs post-intervention) × (combined training vs quadriceps training). RIs and MVIC were tested for the (pre vs post-intervention) comparison. After the assumption checks for normality of distribution (Shapiro Wilk’s test) and homoscedasticity (Levene’s test), considering the number of repeated measures (two) and the absence of missing point, ANOVA for repeated measure was used [41]. Partial η2 was used as the effect size measure. Tukey’s correction for multiple comparisons was used for post hoc tests.
Considering the results of two reliability studies, one referred to young [42] and the other to elderly [43] volunteers, PL, CEA and LDD were analyzed further. To be specific, each experimental group was tested with Bayesian paired-sample t test, hypothesizing a reduction of the values after the training period. Cauchy’s distribution with λ = 0.707 was used arbitrarily for a priori distributions. Bayes Factor (BF10) was calculated, and BF robustness checks were performed across a wide range of prior distributions. Considering the small sample size, Cohen’s d was adjusted to Cohen’s dunbiased [44] as follows:
Results
The volunteers were compliant to the proceeding of the study and successfully concluded the training period. Anthropometric characteristics of the participants revealed 1 out of 11 obese (9%), 9 overweight (82%) and 1 with normal weight, according to the typical BMI cut-offs (18–25, 25–30, and > 30 kg/m2 for normal weight, overweight, and obese, respectively). Waist-to-hip ratio, an index for predicting the risk for non-communicable diseases in elderly has been widely established (Corrêa, Thumé, De Oliveira & Tomasi, 2016), ranged from 0.51 to 0.64. Their waist-to-hip ratio was higher than the typically recommended value of 0.50 [45] in all the participants. Mean PASE total score was 124.90; this result cannot be used to contextualize physical activity levels of our participants, as usually PASE scores are used to stratify on tertiles the participants [46, 47]; our participants reported a PASE score lower than the Italian reference [23] (125 ± 31 and 159 ± 78, respectively). The SF-12 questionnaires results were: Physical Component Summary (PS) = 48.8 ± 4.0 and Mental Component Summary (MS) = 51.4 ± 8.0; basing on large references [48], these results identify our participants healthy, both for physical and mental components. Compared to the data obtained from ten countries about volunteers aged 65–74 years, the mean PCS of our participants was higher than all the references, while the mean MCS was higher than 3 out of 10 references, including the Italian one [49].
Results of spine morphology measurements are shown in Table 2. Concerning spine morphology, the participants had greater angles (both for frontal and sagittal plane measures) than normative values of young healthy adults [33], reflecting, as expected, an impaired spine health. In particular, their spine morphology showed a high heterogeneity in the frontal plane deformities, and consistent and marked deformities in the sagittal plane; in particular, all of the tested volunteers had a kyphosis angle > then 40°, confirming the age-related hyperkyphosis [13].
Our participants showed a good balance status: only 2 out of 11 had lower values than normative values of one-leg stance (open eyes, best of three trials, clustered by age group and gender) [50]. Concerning the Flamingo test, after the training period were shown impairments in 3/6 and 1/5 participants (Q + L and Q group, respectively) and improvements in 1/6 and 2/5 participants (Q + L and Q group, respectively) (see Table 3). Concerning the Fukuda test, the angle of rotation was reduced in 4/6 and 3/5 participants (Q + L and Q group, respectively) while the distance of displacement was reduced in 1/6 and 2/5 participants (Q + L and Q group, respectively) (see Table 3); to be specific, the distance of displacement moved from 110 ± 38 cm to 93 ± 42 cm after the training period in the Q + L group, and from 92 ± 59 to 89 ± 39 cm in the Q group, while the angle of rotation moved from 41 ± 33° to 56 ± 44° in the Q + L group and from 15 ± 17° to 28 ± 12° in the Q group.
Individual plus whole group values are shown in Table 3 (Q + L group) and Table 4 (Q group), and statistical comparisons are shown in Tables 5 and 6. MVIC did not significantly change after the training period, ranging from 381 ± 108 N to 373 ± 133 N in the Q + L group after the training period, and from 447 ± 100 N to 435 ± 111 N in the Q group. Taking into account RM-ANOVA results (Table 2), Bayesian statistics and dunb (Table 3), and Magnitude-Based Decision references [51], the Q + L training resulted better than the Q training in improving static balance control, with likely beneficial effect in reducing CEA (open eyes condition) and possibly beneficial effects in reducing PL (open eyes condition) and LDD (closed eyes condition). The achieved statistical power (1 − β) for within–between interaction of CEA with open eyes was computed as 0.98 with the software G*Power Version 3.1.9.3 [52]. To be specific, volunteers of Q + L group reduced the CEA with open eyes after the training of 25% on average, ranging from 99 ± 38 to 76 ± 42 mm2.
Discussion
To our knowledge, this is the first study evaluating static balance adaptations in response to NMES training of lumbar multifidus and vastus lateralis in healthy elderly. All in all, the participants of the current study can be defined as healthy elderly, substantially inactive, with an anthropometric status linked with high cardiovascular and metabolic risk, with an unhealthy spine morphology, but with a high wellness status, good strength and functional mobility, and non-sarcopenic. We compared the effect of two NMES training protocols with regard to strength, stabilometry and functional balance measures. From our results, the effect of NMES for improving maximum strength (measured by MVIC test) was unclear, as well as for improving functional balance (measured by Flamingo test and Fukuda test). Instead, despite the failing of finding statistical significance, a comprehensive evaluation of statistical testing revealed the combined training (of quadriceps and lumbar paraspinal muscles), rather than quadriceps training, to be likely or possibly beneficial for improving static balance. To be specific, the most likely results involved a reduction of CEA with open eyes, followed by the reduction of LDD with closed eyes and the reduction of PL with open eyes. These parameters were shown to be reliable in young adults [42], allowing further wide studies aiming to define the age-related normative values.
Basing on these prospective pieces of evidence, and according to the repeatability results of stabilometry parameters in elderly [43], we support the use of CEA parameter to determine training-related adaptation of static balance. Indeed, despite the condition “narrow stand eyes closed” has been proved to be the most reliable position [43], measuring CEA with normal standing position (feet slightly apart with an open angle) could also be adopted. PL with open eyes and LDD with closed eyes deserve further evaluations. With this regard, it should be of interest to go ahead with the results of Bernard and colleagues, who demonstrated the ones with the lowest initial level of balance control obtain more benefits from a postural control training program [53], defining which parameters may be the most adequate in monitoring balance control adaptations in healthy and clinical elderly.
The importance of lower limb strength for allowing a good postural control in elderly has been demonstrated [10]. In addition, a systematic review conducted by Granacher and colleagues [17] concluded trunk muscle strength to be important in fall prevention and rehabilitation program, as an additional or alternative method to balance and resistance training. One of the reasons behind the likely more beneficial effect of the combined training may lie in the effectiveness of NMES training for improving the spinal stabilization, thanks to an enhanced activation of the deep fibers of multifidus [32], as demonstrated also in patients with lumbar degenerative kyphosis [31]. Aging of the spine contributes to the development of painful and debilitating disorders, due to the main pathophysiological processes of degeneration and bone mass reduction [54]. We think that the time course of adaptations related to middle-term training protocols, as in the current study, did not allow to counteract these processes. However, targeting trunk extensor muscles may have caused a greater control of core, resulting in a reduced instability during static posture. NMES has the potential to increase muscle mass, both in quadriceps and lumbar muscles; interestingly, improving muscle mass of paravertebral muscles likely benefits spine morphology and posture maintenance [55].
Whereas the training of lower limbs, as well as upper limbs, can be performed in healthy elderly with several active methods, the training of back muscles represents a more difficult challenge and, often, the presence of back pain or functional mobility impairments severely limits the possibility of active training, unless it is tutored by physiotherapists and clinicians [56]. Even more in several clinical conditions, where passive training represents the most suitable method to produce muscular exercise [57]. With this regard, it has been demonstrated the effectiveness of trunk extension strength in limiting the risk of fall [14], a major problem in elderly. Therefore, NMES training of back muscles can be adopted where core training, resistance training or balance training [58] are severely limited or impossible. Since the time course on mobility function in elderly due to NMES training varies across tasks [59], both in training and detraining periods, the time course of postural adaptation due to NMES training shall be investigated with larger sample studies.
The study did not come without limits. Internal validity is threatened in this study, since the absence of control group. However, there were difficulties in recruiting a control group where the contextual variables affecting static balance were identical to our experimental group, such as physical activity level, sight and vestibular function, anthropometry, previous injuries, wellness, and lifestyle habits. Former physical lifestyle history may have biased the results, even though it has demonstrated that former athletic activity history provides no protection for the age-related-onset postural impairment, whereas current activity does [60]. Positively, participants were not aware of the effect of NMES on the static balance nor they received any feedback about their results of stabilometry before the end of the study protocol. One strength of this study was that we carefully controlled historical events during the training period, which may have influenced the outcomes, such as sight or vestibular diseases, or osteo-muscular disorders. Moreover, an 8-week period alone unlikely affects static balance in healthy elderly. External validity may also have been threatened in this study due to the general intrinsic limit of NMES interventions involving its intensity, which is based on individual threshold comfort [19].
Finally, larger sample would allow to discriminate the effectiveness of training diverse muscle groups, solely or combined, compared to a control group, and the time course of adaptations; to extend the results of this study, a third group who train only lumbar paraspinal muscles, plus a fourth group as control should be included.
Conclusions
Healthy elderly, substantially inactive, with unhealthy anthropometric status and spine morphology, non-sarcopenic, benefit from a NMES training applied on both quadriceps and lumbar paraspinal muscles. We demonstrated a more beneficial effect of the combined training (quadriceps and lumbar paraspinal muscles) in respect to the training applied on quadriceps only. This was possibly due to the effectiveness of NMES training for improving spinal stabilization. Therefore, passive training on lumbar paravertebral muscles can be a suitable method for positively affecting postural control in elderly. The possible beneficial effect in improving static balance, as found in this study, should be translated and verified in those clinical conditions where active training could not be performed to produce muscular exercise.
References
Marieb EN, Hoehn KN (2015) Human anatomy and physiology. Pearson Education
Schiaffino S, Reggiani C (2011) Fiber types in mammalian skeletal muscles. Physiol Rev 91:1447–1531. https://doi.org/10.1152/physrev.00031.2010
Frontera WR, Ochala J (2015) Skeletal muscle: a brief review of structure and function. Calcif Tissue Int 96:183–195
Cawthon PM, Marshall LM, Michael Y et al (2007) Frailty in older men: prevalence, progression, and relationship with mortality. J Am Geriatr Soc. https://doi.org/10.1111/j.1532-5415.2007.01259.x
Janssen I, Heymsfield SB, Ross R (2002) Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc 50:889–896
Ito T, Sakai Y, Yamazaki K et al (2019) Relationship between l4/5 lumbar multifidus cross-sectional area ratio and fall risk in older adults with lumbar spinal stenosis: a retrospective study. Geriatrics. https://doi.org/10.3390/geriatrics4020038
Ishikawa Y, Miyakoshi N, Kasukawa Y et al (2009) Spinal curvature and postural balance in patients with osteoporosis. Osteoporos Int. https://doi.org/10.1007/s00198-009-0919-9
Sinaki M, Brey RH, Hughes CA et al (2005) Balance disorder and increased risk of falls in osteoporosis and kyphosis: significance of kyphotic posture and muscle strength. Osteoporos Int. https://doi.org/10.1007/s00198-004-1791-2
Paillard T (2017) Relationship between muscle function, muscle typology and postural performance according to different postural conditions in young and older adults. Front Physiol 8. https://doi.org/10.3389/fphys.2017.00585
Andrade HB, Costa SM, Pirôpo US et al (2018) Lower limb strength, but not sensorial integration, explains the age-associated postural control impairment. Muscles Ligaments Tendons J. https://doi.org/10.11138/mltj/2018.8.1.113
Dreischarf M, Albiol L, Rohlmann A et al (2014) Age-related loss of lumbar spinal lordosis and mobility—a study of 323 asymptomatic volunteers. PLoS ONE. https://doi.org/10.1371/journal.pone.0116186
Fernandes VLS, Ribeiro DM, Fernandes LC, de Menezes RL (2018) Postural changes versus balance control and falls in community-living older adults: a systematic review. Fisioter Em Mov. https://doi.org/10.1590/1980-5918.031.ao25
Katzman WB, Wanek L, Shepherd JA, Sellmeyer DE (2010) Age-related hyperkyphosis: its causes, consequences, and management. J Orthop Sports Phys Ther 40:352–360. https://doi.org/10.2519/jospt.2010.3099
Beserra Da Silva R, Costa-Paiva L, Siani Morais S et al (2010) Predictors of falls in women with and without osteoporosis. J Orthop Sports Phys Ther 40:582–588. https://doi.org/10.2519/jospt.2010.3239
Burke TN, França FJR, Ferreira de Meneses SR et al (2010) Postural control in elderly persons with osteoporosis: efficacy of an intervention program to improve balance and muscle strength: a randomized controlled trial. Am J Phys Med Rehabil 89:549–556. https://doi.org/10.1097/PHM.0b013e3181ddccd2
Gomes MM, Reis JG, Carvalho RL et al (2015) Analysis of postural control and muscular performance in young and elderly women in different age groups. Braz J Phys Ther 19:1–9. https://doi.org/10.1590/bjpt-rbf.2014.0068
Granacher U, Gollhofer A, Hortobágyi T et al (2013) The importance of trunk muscle strength for balance, functional performance, and fall prevention in seniors: a systematic review. Sports Med 43:627–641. https://doi.org/10.1007/s40279-013-0041-1
Dirks ML, Wall BT, Snijders T et al (2014) Neuromuscular electrical stimulation prevents muscle disuse atrophy during leg immobilization in humans. Acta Physiol Oxf Engl 210:628–641. https://doi.org/10.1111/apha.12200
Maffiuletti NA, Roig M, Karatzanos E, Nanas S (2013) Neuromuscular electrical stimulation for preventing skeletal-muscle weakness and wasting in critically ill patients: a systematic review. BMC Med 11:137. https://doi.org/10.1186/1741-7015-11-137
Lacroix A, Hortobágyi T, Beurskens R, Granacher U (2017) Effects of supervised vs. unsupervised training programs on balance and muscle strength in older adults: a systematic review and meta-analysis. Sports Med Auckl NZ 47:2341–2361. https://doi.org/10.1007/s40279-017-0747-6
World Medical Association (2013) World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310:2191. https://doi.org/10.1001/jama.2013.281053
Jackson AS, Pollock ML (1985) Practical assessment of body composition. Phys Sportsmed 13:76–90. https://doi.org/10.1080/00913847.1985.11708790
Covotta A, Gagliardi M, Berardi A et al (2018) Physical activity scale for the elderly: translation, cultural adaptation, and validation of the Italian version. Curr Gerontol Geriatr Res. https://doi.org/10.1155/2018/8294568
Ware J, Kosinski M, Keller SD (1996) A 12-Item short-form health survey: construction of scales and preliminary tests of reliability and validity. Med Care 34:220–233. https://doi.org/10.1097/00005650-199603000-00003
Ottoboni G, Cherici A, Marzocchi M, Chattat R (2017) Algoritimi di calcolo per gli indici PCS e MCS del questinario SF-12. AMSActa. https://doi.org/10.6092/unibo/amsacta/5751
Cruz-Jentoft AJ, Bahat G, Bauer J et al (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48:16–31. https://doi.org/10.1093/ageing/afy169
Suzuki Y, Kawai H, Kojima M et al (2016) Construct validity of posture as a measure of physical function in elderly individuals: use of a digitalized inclinometer to assess trunk inclination. Geriatr Gerontol Int 16:1068–1073. https://doi.org/10.1111/ggi.12600
Bangsbo J, Blackwell J, Boraxbekk C-J et al (2019) Copenhagen Consensus statement 2019: physical activity and ageing. Br J Sports Med 53:856–858. https://doi.org/10.1136/bjsports-2018-100451
Di Filippo ES, Mancinelli R, Marrone M et al (2017) Neuromuscular electrical stimulation improves skeletal muscle regeneration through satellite cell fusion with myofibers in healthy elderly subjects. J Appl Physiol Bethesda MD 123:501–512. https://doi.org/10.1152/japplphysiol.00855.2016
Baek SO, Ahn SH, Jones R et al (2014) Activations of deep lumbar stabilizing muscles by transcutaneous neuromuscular electrical stimulation of lumbar paraspinal regions. Ann Rehabil Med 38:506–513. https://doi.org/10.5535/arm.2014.38.4.506
Kim SY, Kim JH, Jung GS et al (2016) The effects of transcutaneous neuromuscular electrical stimulation on the activation of deep lumbar stabilizing muscles of patients with lumbar degenerative kyphosis. J Phys Ther Sci 28:399–406. https://doi.org/10.1589/jpts.28.399
Coghlan S, Crowe L, McCarthypersson U et al (2011) Neuromuscular electrical stimulation training results in enhanced activation of spinal stabilizing muscles during spinal loading and improvements in pain ratings. Conf Proc IEEE Eng Med Biol Soc 2011:7622–7625. https://doi.org/10.1109/IEMBS.2011.6091878
D’Amico M, Kinel E, Roncoletta P (2017) Normative 3D opto-electronic stereo-photogrammetric posture and spine morphology data in young healthy adult population. PLoS ONE. https://doi.org/10.1371/journal.pone.0179619
D’amico M, Kinel E, Roncoletta P, (2018) 3D quantitative evaluation of spine proprioceptive perception/motor control through instinctive self-correction maneuver in healthy young subjects’ posture: an observational study. Eur J Phys Rehabil Med. https://doi.org/10.23736/S1973-9087.17.04738-4
D’amico M, D’amico G, Frascarello M, et al (2008) A 3-D skeleton model and SEMG approach for integrated neck and low back pain analysis test batteries. Stud Health Technol Inform 140:79–84
Scoppa F, Gallamini M, Belloni G, Messina G (2017) Clinical stabilometry standardization: feet position in the static stabilometric assessment of postural stability. Acta Med Mediterr. https://doi.org/10.19193/0393-6384_2017_4_105
Swanenburg J, de Bruin ED, Favero K et al (2008) The reliability of postural balance measures in single and dual tasking in elderly fallers and non-fallers. BMC Musculoskelet Disord 9:162. https://doi.org/10.1186/1471-2474-9-162
Jonsson E, Seiger A, Hirschfeld H (2004) One-leg stance in healthy young and elderly adults: a measure of postural steadiness? Clin Biomech Bristol Avon 19:688–694. https://doi.org/10.1016/j.clinbiomech.2004.04.002
Bonanni M, Newton R (1998) Test-retest reliability of the Fukuda Stepping Test. Physiother Res Int J Res Clin Phys Ther 3:58–68
Pietrangelo T, Bondi D, Kinel E, Verratti V (2019) The bottom–up rise strength transfer in elderly after endurance and resistance training: the BURST. Front Physiol. https://doi.org/10.3389/fphys.2018.01944
Armstrong RA (2017) Recommendations for analysis of repeated-measures designs: testing and correcting for sphericity and use of MANOVA and mixed model analysis. Ophthalmic Physiol Opt 37:585–593. https://doi.org/10.1111/opo.12399
Nagymáté G, Orlovits Z, Kiss RM (2018) Reliability analysis of a sensitive and independent stabilometry parameter set. PLoS ONE 13:e0195995. https://doi.org/10.1371/journal.pone.0195995
Bauer CM, Gröger I, Rupprecht R et al (2010) Reliability of static posturography in elderly persons. Z Gerontol Geriatr 43:245–248. https://doi.org/10.1007/s00391-009-0052-6
Fritz CO, Morris PE, Richler JJ (2012) Effect size estimates: current use, calculations, and interpretation. J Exp Psychol Gen 141:2–18. https://doi.org/10.1037/a0024338
Corrêa MM, Thumé E, De Oliveira ERA, Tomasi E (2016) Performance of the waist-to-height ratio in identifying obesity and predicting non-communicable diseases in the elderly population: a systematic literature review. Arch Gerontol Geriatr 65:174–182. https://doi.org/10.1016/j.archger.2016.03.021
Curcio F, Liguori I, Cellulare M et al (2019) Physical activity scale for the elderly (PASE) score is related to sarcopenia in noninstitutionalized older adults. J Geriatr Phys Ther 42:130–135. https://doi.org/10.1519/JPT.0000000000000139
Schuit AJ, Schouten EG, Westerterp KR, Saris WHM (1997) Validity of the physical activity scale for the elderly (PASE): according to energy expenditure assessed by the doubly labeled water method. J Clin Epidemiol 50:541–546. https://doi.org/10.1016/S0895-4356(97)00010-3
Kodraliu G, Mosconi P, Groth N et al (2001) Subjective health status assessment: evaluation of the Italian version of the SF-12 Health Survey. results from the MiOS Project. J Epidemiol Biostat 6:305–316
Gandek B, Ware JE, Aaronson NK et al (1998) Cross-validation of item selection and scoring for the SF-12 Health Survey in nine countries: results from the IQOLA Project. J Clin Epidemiol 51:1171–1178. https://doi.org/10.1016/S0895-4356(98)00109-7
Springer BA, Marin R, Cyhan T et al (2007) Normative values for the unipedal stance test with eyes open and closed. J Geriatr Phys Ther 30:8–15
Hopkins WG, Marshall SW, Batterham AM, Hanin J (2009) Progressive statistics for studies in sports medicine and exercise science. Med Sci Sports Exerc 41:3–13. https://doi.org/10.1249/MSS.0b013e31818cb278
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191. https://doi.org/10.3758/BF03193146
Bernard PL, Blain H, Gerazime A et al (2018) Relationship between a three-month physical conditioning “posture-balance-motricity and health education” (PBM-HE) program on postural and balance capacities of sedentary older adults: influence of initial motor profile. Eur Rev Aging Phys Act. https://doi.org/10.1186/s11556-018-0203-0
Papadakis M, Sapkas G, Papadopoulos EC, Katonis P (2011) Pathophysiology and biomechanics of the aging spine. Open Orthop J 5:335–342. https://doi.org/10.2174/1874325001105010335
Jandova T, Narici MV, Steffl M et al (2020) Muscle hypertrophy and architectural changes in response to eight-week neuromuscular electrical stimulation training in healthy older people. Life 10:184. https://doi.org/10.3390/life10090184
Ishak NA, Zahari Z, Justine M (2016) Effectiveness of strengthening exercises for the elderly with low back pain to improve symptoms and functions: a systematic review. Scientifica. https://doi.org/10.1155/2016/3230427
Heesterbeek M, Van der Zee EA, van Heuvelen MJG (2018) Passive exercise to improve quality of life, activities of daily living, care burden and cognitive functioning in institutionalized older adults with dementia—a randomized controlled trial study protocol. BMC Geriatr 18:182. https://doi.org/10.1186/s12877-018-0874-4
Granacher U, Lacroix A, Muehlbauer T et al (2013) Effects of core instability strength training on trunk muscle strength, spinal mobility, dynamic balance and functional mobility in older adults. Gerontology 59:105–113. https://doi.org/10.1159/000343152
Mani D, Almuklass AM, Amiridis IG, Enoka RM (2018) Neuromuscular electrical stimulation can improve mobility in older adults but the time course varies across tasks: double-blind, randomized trial. Exp Gerontol 108:269–275. https://doi.org/10.1016/j.exger.2018.04.018
Bulbulian R, Hargan ML (2000) The effect of activity history and current activity on static and dynamic postural balance in older adults. Physiol Behav 70:319–325. https://doi.org/10.1016/s0031-9384(00)00272-9
Funding
Open access funding provided by Università degli Studi G. D'Annunzio Chieti Pescara within the CRUI-CARE Agreement. This research was supported by the "Departments of Excellence 2018–2022" initiative of the Italian Ministry of Education, University and Research for the Department of Neuroscience, Imaging and Clinical Sciences (DNISC) of the University of Chieti-Pescara. ESDF was supported by Fondazione Umberto Veronesi.
Author information
Authors and Affiliations
Contributions
Conceptualization: DB, TJ, TP. Methodology: DB, TJ, MDAt, TP. Formal analysis: DB. Investigation: DB, TJ, VV. Resources: VV, TP, MDAt. Writing—original draft: DB, TJ, MDAm, EK, ESDF, SF, TP. Writing—review and editing: DB, TJ, VV, MDAm, EK, MDAt, ESDF, SF, TP. Visualization: DB. Supervision: VV, MDAt, TP. Project administration: MDAt, TP. Funding acquisition: MDAt, TP.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Ethical approval
The study was approved by the Ethics Committee of the University “G. d’Annunzio” (n.16 of 05/09/2019), conforming to the ethical standards set by the Declaration of Helsinki and later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bondi, D., Jandova, T., Verratti, V. et al. Static balance adaptations after neuromuscular electrical stimulation on quadriceps and lumbar paraspinal muscles in healthy elderly. Sport Sci Health 18, 85–96 (2022). https://doi.org/10.1007/s11332-021-00777-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-021-00777-0