Abstract
Purpose
This study aimed to evaluate the effect of mandibular advancement splint (MAS) therapy on cardiac autonomic function in patients with obstructive sleep apnoea (OSA) using heart rate variability (HRV) analysis.
Methods
Electrocardiograms (ECG) derived from polysomnograms (PSG) of three prospective studies were used to study HRV of patients with OSA before and after MAS treatment. HRV parameters were averaged across the entire ECG signal during N2 sleep using 2-min epochs shifted by 30 s. Paired t-tests were used to compare PSG and HRV measures before and after treatment, and the percent change in HRV measures was regressed on the percent change in apnoea-hypopnea index (AHI).
Results
In 101 patients with OSA, 72% were Caucasian, 54% men, the mean age was 56 ± 11 years, BMI 29.8 ± 5.3 kg/m2, and treatment duration was 4.0 ± 3.2 months. After MAS therapy, there was a significant reduction in OSA severity (AHI, − 18 ± 16 events per hour, p < 0.001) and trends towards increased low-frequency to high-frequency ratio, low-frequency power, and reduced high-frequency power (LF:HF, − 0.4 ± 1.5, p = 0.01; LF, − 3 ± 16 nu, p = 0.02, HF, 3.5 ± 13.7 nu, p = 0.01). Change in NN intervals correlated with the change in AHI (β(SE) = − 2.21 (0.01), t = − 2.85, p = 0.005). No significant changes were observed in the time-domain HRV markers with MAS treatment.
Conclusion
The study findings suggest that successful MAS treatment correlates with changes in HRV, specifically the lengthening of NN intervals, a marker for improved cardiac autonomic adaptability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnoea (OSA) is a sleep-related breathing disorder characterized by the repeated collapse of the upper airway during sleep, leading to intermittent hypoxia, intrathoracic pressure swings, and fragmented sleep [1, 2]. OSA increases the risk of cardiovascular diseases, such as hypertension and coronary artery disease, partly due to changes in the autonomic nervous system, particularly upregulated sympathetic nerve activity [3].
Mandibular advancement splint (MAS) is an effective treatment for OSA and works by increasing upper airway patency and reducing collapsibility [4]. Currently, MAS is the recommended therapy for mild-moderate OSA and severe OSA when continuous positive airway pressure (CPAP) treatment is not tolerated [5]. Evidence suggests that health outcomes, at least in the short term, are comparable between MAS and CPAP therapy [6]. Randomized controlled trials of MAS treatment have demonstrated improved 24-h mean arterial blood pressure, which is more pronounced in hypertensive patients and those with resistant hypertension [7, 8]. However, the effects of MAS therapy on other intermediary cardiovascular markers, such as cardiac autonomic function, remain poorly understood [9], highlighting the need for further research on its potential to reduce cardiovascular risk.
Heart rate variability (HRV) analysis can be used non-invasively to evaluate beat-to-beat changes in cardiac autonomic control, which is particularly valuable for evaluating the relative activity of sympathetic and parasympathetic cardiac modulation [10]. OSA is associated with an HRV profile indicative of sympathetic predominance, which is a hallmark of hypertension, a major risk factor for cardiovascular disease [11]. Therefore, we aimed to use HRV to assess cardiac autonomic function following MAS therapy. We hypothesized that cardiac autonomic function would improve following successful MAS treatment i.e. that there would be a reduction in sympathetic and an increase in parasympathetic HRV markers.
Methods
Participants
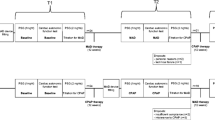
This study utilized data from three prospective MAS therapy research studies conducted at the Sleep Investigation Laboratory, Royal North Shore Hospital. Adult participants (age ≥ 18 years) diagnosed with OSA (AHI ≥ 10/h) and treated with MAS were included in the current analysis (Fig. 1). These studies collected baseline polysomnography (PSG) upon initial visit to the clinic, and this was followed by a treatment PSG with MAS after at least one month of MAS therapy [4, 12, 13]. Sutherland et al. [12] used this data to derive multimodal phenotypes to predict treatment response in patients using MAS. Phillips [4] performed a randomized controlled trial (RCT) comparing health outcomes of MAS and CPAP treatment. Jugé [13] used imaging techniques to characterize whether inspiratory tongue movement is associated with MAS treatment outcomes. Study characteristics and outcomes were compared across these three datasets and are presented in the Supplementary Material (Supplementary Tables 1–2). In all studies, dental eligibility was assessed by a dentist, and exclusion criteria were limited to contraindications to MAS therapy i.e. periodontal disease, temporomandibular joint diseases, active upper airway infection, and insufficient teeth.
We used the same MAS design for all studies — a titratable, custom-fitted two-piece device (SomnoMed Ltd., based in Sydney, Australia). Participants incrementally adjusted the MAS to the point of maximum comfortable mandibular advancement, as a means of self-titration and titration degree, with the titration degree confirmed by a dental specialist to ensure the correct adjustment. All studies were approved by Institutional Human Research Ethics Committees, and informed consent was obtained from all participants before the trials commenced.
For the present study, the participants included patients of varying response to treatment. Study characteristics and outcomes for different response groups were compared and presented in the Supplementary Material (Supplementary Table 3–4).
Polysomnography
During full night PSG, standard channels were utilized to record physiological parameters, including electrooculography (EOG), chin electromyography (EMG), nasal airflow pressure via nasal cannula, thoracic and abdominal respiratory effort, finger pulse oximetry to measure oxygen saturation (SpO2%), body position, and leg electromyography. Trained sleep scientists scored each recording according to the guidelines set by the American Academy of Sleep Medicine [14].
Holter monitor processing
As part of in-laboratory PSG, electrocardiogram (ECG) data was collected using three electrodes placed in the standard lead II configuration. The ECG signals were extracted from the polysomnograms and analysed using commercially available Holter software for QRS detection, ectopic beat detection, and labelling (Sentinel Holter Data Management System v11.5.1, Spacelabs Healthcare, Issaquah, WA, USA). QRS detection was performed with a resolution of 1 ms.
Heart rate variability
HRV analysis was performed on ECG recordings in accordance with standard guidelines [10] using HRV algorithms implemented in signal processing software (MATLAB 2017, version 9.2.0.538062 (R2017a), Mathworks, Natick, Massachusetts). HRV parameters were calculated using 2-min epochs, shifted by 30 s, across the entire ECG signal, and then averaged across segments of N2 sleep (stage 2 non-rapid eye movement). N2 sleep is the preferred stage for nocturnal HRV analysis due to its relative stability, as it is primarily controlled by the parasympathetic nervous system and has fewer arousals that may impact the sensitivity of HRV measures [15]. Epochs with mixed sleep stages and ECG sections associated with respiratory events or cardiac arrhythmia (including atrial and ventricular ectopic beats) were excluded from HRV analysis. If the total exclusion period exceeded 12 s (10% of epoch length), the entire epoch was excluded [16].
Time — domain measures
The time domain HRV metrics employed in the analysis were (1) Average NN interval (2) SDNN, standard deviation of NN intervals, utilized as a measure of global HRV, (3) RMSSD, which is the square root of the mean squared differences of successive NN intervals, and (4) pNN50, the percentage of absolute differences in successive NN values greater than 50 ms (Table 1). RMSSD and pNN50 are short-term measures that are strongly associated with high-frequency oscillations and reflect parasympathetic modulation of the heart [10].
Frequency — domain measures
Frequency-domain HRV parameters were calculated using the Lomb periodogram on the preprocessed data. Total power (TP, ms2) was defined as the power between 0.04 and 0.4 Hz and is associated with global HRV changes. Absolute power within the low frequency band (0.04–0.15 Hz; LF, ms2) reflects sympathetic cardiac modulation with a vagal component (Table 1), while absolute power within the high frequency band reflects vagal cardiac autonomic control (0.15–0.40 Hz; HF, ms2. Normalized values LFnu and HFnu are associated with sympathetic and parasympathetic activity, respectively [10]. The ratio between these indices (LF: HF) represent sympathovagal balance [10].
Statistical analysis
Mean and standard deviation (SD) were presented for continuous variables. Paired t-tests were utilized to compare polysomnographic characteristics and HRV markers before and after MAS treatment. Normality was evaluated using the Shapiro–Wilk test, and distribution plots were visually examined. Wilcoxon signed rank test was used for pairwise comparisons of nonparametric continuous data.
To investigate whether the responsiveness to MAS treatment influenced the changes in HRV, regression analyses were conducted for the change in HRV markers (∆HRV) (outcome variable) against treatment duration (predictor), and against treatment duration and change in AHI (∆AHI) (predictors). Treatment duration was calculated as the amount of time, in months, between the PSG collected at baseline and with treatment. The change (\(\Delta\)) was calculated using the following formula: \(\Delta = T-B\) where \(T\) referred to treatment outcomes and \(B\) indicated baseline outcomes. The p value was adjusted for multiple comparisons using Bonferroni correction; therefore, significance is denoted as p values less than 0.005 (10 comparators). Subgroup analyses were also performed comparing clinical and HRV outcomes between (1) all three data sources, (2) treatment response groups, and (3) treatment duration groups (Supplementary Tables 1 – 5). All data analyses were performed using SPSS software (version 24; SPSS Inc., Chicago, IL).
Results
Participant characteristics
Table 2 shows the clinical characteristics of the 101 study participants. The sample had a relatively even distribution of men and women (54% men), who were mostly middle-aged (age, 56 ± 11 years), overweight (BMI, 29.8 ± 5.3 kg/m2) and identified themselves as Caucasian (72% Caucasian). The average duration of treatment was 4.0 ± 3.2 months.
MAS treatment effect on polysomnographic indices
Table 3 shows the changes in polysomnographic parameters observed from baseline to treatment. The results show a significant reduction in AHI, AHINREM, AHIREM, and ODI4% (mean difference ± SE; − 18 ± 17); − 19 ± 24; − 19 ± 21; − 13 ± 15 events/hour), and a significant increase in SpO2 nadir (5 ± 33%). No significant changes were observed in TST, NREM duration, REM duration, or sleep efficiency.
MAS treatment effect on HRV measures
Table 4 shows the changes in HRV markers before and after MAS treatment. The results show a trend towards increased LF:HF ratio and LFnu (0.4 ± 1.5, 3 ± 16nu), along with a reduction in HFnu (− 3.5 ± 13.7nu). No significant changes were observed in the time-domain HRV markers with MAS treatment.
Regression analyses comparing changes in HRV and AHI measures are presented in Table 5. The results indicate a correlation between change in average NN interval and change in AHI (β(SE) = − 2.21 (0.01), t = − 2.85, p = 0.005) (Fig. 2). Change in pNN50 and change in AHI also trend towards an association (β(SE) = − 0.17 (0.18), t = − 1.87, p = 0.065) (Fig. 2). Change in AHI following MAS treatment was not significantly correlated with changes in frequency domain HRV measures. In addition, treatment duration had no association with HRV measures.
Supplementary Table 5 (refer to Supplementary Material) compares changes in HRV markers across patients grouped by treatment duration. There were no significant differences between change in HRV markers across the three treatment time groups. However, there was a trend towards higher change in average NN interval in the longer term, 9–12-month treatment group. Although this group also had the lowest sample size with only 6 participants.
Discussion
This study investigated the effect of MAS treatment on cardiac autonomic function in patients with OSA. Our hypothesis was that successful MAS treatment would result in increased parasympathetic and reduced sympathetic cardiac autonomic modulation. The findings revealed a novel correlation between successful MAS therapy and lengthened NN intervals, a marker of improved cardiac autonomic adaptability [17]. Specifically, each unit of reduction in AHI was associated with a 2.21 unit increase in NN interval length, and a similar trend towards higher pNN50 with improved AHI. The lengthened NN interval observed resulted from increased cardiac vagal modulation, which reflected improved cardiac autonomic adaptability and may have implications for reducing cardiovascular risk [17].
Of the five similar studies undertaken [18], only one study has reported nocturnal vagal predominance after MAS use [19]. The authors showed in their retrospective study that after 3 months of MAS use (N = 58), patients demonstrated improved cardiac autonomic adaptability through increases in average NN, normalized HF power, and SDNN and reduced normalized LF power [19]. In addition, other studies have reported an increase in daytime normalized HF power after short-term MAS use, albeit using small sample sizes (N = 10) and only during a controlled breathing protocol, rather than normal breathing [19, 20]. In the other studies investigating changes to HRV markers after MAS, no changes to sympathetic or vagal markers were found [21, 22]. In their randomized crossover trial, Dal-Fabbro and colleagues [21] showed that compared with placebo oral appliance, global HRV marker, TP, was significantly lower, with no differences found between MAS and CPAP treatment periods. Collectively, these findings suggest that MAS use may result in improved cardiac autonomic adaptability, though different studies have yielded different conclusions.
Clinically, MAS use is hindered by its varying degrees of efficacy whereby around one-third of patients completely resolve OSA with MAS treatment, one-third will have a demonstrated halving of their OSA severity, and one-third will demonstrate minimal effect [23]. Therefore, patient responsiveness to MAS therapy must be considered when evaluating treatment effect on cardiac autonomic function. In our study, improvement in vagally mediated HRV markers was only observed after considering responsiveness to MAS treatment (i.e., those with a greater change in AHI). We found a trend towards increased pNN50 with improved AHI. Similar subgroup analyses of treatment responders (n = 34, defined as, > 50% reduction in AHI and AHI < 20/h after treatment) identified significant increases in average NN and normalized HF power, accompanied by reductions in TP, VLF, LF, LF:HF ratio, and normalized LF power after MAS treatment [19]. In contrast, no significant changes in cardiac autonomic function were observed in the non-responder group (n = 24), defined as those with a < 50% reduction in AHI and AHI > 20/h following treatment [19]. Taken together, these two investigations suggest responders to MAS treatment experience a marked shift towards parasympathetic predominance in sympathovagal balance, characteristic of improved cardiac autonomic regulation. However, the lack of uniformity in results across all MAS treatment trials suggests that responsiveness to MAS therapy is only one factor contributing to improved cardiovascular health.
Despite an inextricable link between OSA and cardiovascular risk, the largest randomized study to date with a 3.7-year follow-up period investigating OSA treatment failed to demonstrate any improvement in hard cardiovascular endpoints [24]. A significant limitation in this trial was poor treatment adherence to CPAP, which was only 3.3 h/night in a population with cardiovascular disease [24]. While MAS therapy is generally less efficacious than CPAP, its higher treatment adherence makes both treatment modalities equally effective [23]. The main set-back to ours and other MAS treatment trials has been a lack of access to technologies to objectively record daily adherence. Therefore, future MAS trials should consider the effects of treatment responsiveness and objective daily adherence when evaluating cardiovascular risk.
HRV is a useful tool to monitor the cardiac autonomic benefits of OSA treatment, yet there is substantial heterogeneity among treatment trials [18]. Differences in HRV indices used and HRV sensitivity to external factors affecting HRV measurements are observed. Not only do methodologies vary between whether or not HRV is measured during daytime, nighttime, or over 24 h [18]. Some studies used single 5-min segments to represent changes that occur over whole sleep stages [21], while others used a minimum number of beats per tachogram [25], or limited their nocturnal analyses to specific times irrespective of sleep stages [19]. This is problematic as time domain analysis of HRV is highly influenced by the condition and duration of the recording [10]. Routine recording of ECG during polysomnography makes assessment of HRV during sleep a widely used and easily accessible method for measuring cardiac autonomic function. However, disordered respiration experienced by patients with OSA reduces the sensitivity of HRV as a tool for evaluating the effectiveness of OSA therapy on global measures of HRV [10]. We addressed this concern by excluding ECG segments with respiratory events, ectopic beats, and other cardiac arrhythmias. In order to increase sensitivity of our HRV measure, we also adopted a methodology of averaging HRV markers across the entire PSG-derived ECG using a shifting window of 2 min, and generating HRV measurements for the N2 sleep stage, as is common practice [15]. However, our study could have benefitted from longer duration of data collection. Using 24-h Holter monitoring to assess the SDNN as a marker of global HRV is preferable for HRV assessment, as it can better account for metabolic and circadian variability, which are strongly associated with cardiovascular risk [10].
In some patients, MAS treatment can alter nocturnal cardiac autonomic activity. Multiple studies have shown that MAS treatment may enhance vagally mediated markers, thereby improving cardiac autonomic adaptability. However, research in the field of MAS is hampered by various challenges. As previously highlighted, the lack of objective daily usage data is a major limitation to this and other similar studies. While some studies have provided preliminary data on average usage using objective adherence monitors for up to 1 year of treatment, these studies were small and did not evaluate cardiovascular outcomes. In contrast, our study had a relatively large sample size and captured a more comprehensive and accurate measure for cardiac autonomic function by averaging HRV markers over the entire ECG. However, our study did use data sets from three separate treatment trials and adherence was not measured uniformly across these studies. Therefore, we were unable to provide objective or subjective measures of adherence.
Conclusion
The current study investigated the effect of MAS treatment on cardiac autonomic function in patients with OSA. The findings suggested that successful treatment with MAS correlated with changes to HRV, specifically the increase in pNN50 and the lengthening of NN intervals, a marker for improved cardiac autonomic adaptability [10, 17]. However, change in AHI following MAS treatment was not significantly correlated with changes in frequency domain HRV measures.
Data availability
The datasets made available for this study and/or generated analysis during the current study are available from the corresponding author on reasonable request.
References
Kohler M, Stradling JR (2010) Mechanisms of vascular damage in obstructive sleep apnea. Nat Rev Cardiol 7(12):677–685. https://doi.org/10.1038/nrcardio.2010.145
Veasey SC, Rosen IM (2019) Obstructive sleep apnea in adults. N Engl J Med 380(15):1442–1449
Esquinas C, Sánchez-de-la Torre M, Aldomá A, Florés M, Martínez M, Barceló A, Barbé F, Network SS (2013) Rationale and methodology of the impact of continuous positive airway pressure on patients with ACS and nonsleepy OSA: the ISAACC Trial. Clin Cardiol 36(9):495–501
Phillips CL, Grunstein RR, Darendeliler MA, Mihailidou AS, Srinivasan VK, Yee BJ, Marks GB, Cistulli PA (2013) Health outcomes of continuous positive airway pressure versus oral appliance treatment for obstructive sleep apnea: a randomized controlled trial. Am J Respir Crit Care Med 187(8):879–887
Kapur VK, Auckley DH, Chowdhuri S, Kuhlmann DC, Mehra R, Ramar K, Harrod CG (2017) Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med 13(3):479–504
Bratton DJ, Gaisl T, Wons AM, Kohler M (2015) CPAP vs mandibular advancement devices and blood pressure in patients with obstructive sleep apnea: a systematic review and meta-analysis. JAMA 314(21):2280–2293
Andrén A, Hedberg P, Walker-Engström ML, Wahlén P, Tegelberg Å (2013) Effects of treatment with oral appliance on 24-h blood pressure in patients with obstructive sleep apnea and hypertension: a randomized clinical trial. Sleep Breath 17:705–712
Gotsopoulos H, Kelly JJ, Cistulli PA (2004) Oral appliance therapy reduces blood pressure in obstructive sleep apnea: a randomized, controlled trial. Sleep 27(5):934–941
Ucak S, Dissanayake HU, Sutherland K, de Chazal P, Cistulli PA (2021) Heart rate variability and obstructive sleep apnea: current perspectives and novel technologies. J Sleep Res 30(4):e13274
Malik M (1996) Heart rate variability: Standards of measurement, physiological interpretation, and clinical use: task force of the European Society of Cardiology and the North American Society for Pacing and Electrophysiology. Ann Noninvasive Electrocardiol 1(2):151–181
Dissanayake HU, Bin YS, Ucak S, de Chazal P, Sutherland K, Cistulli PA (2021) Association between autonomic function and obstructive sleep apnea: a systematic review. Sleep Med Rev 57:101470
Sutherland K, Chan AS, Ngiam J, Dalci O, Darendeliler MA, Cistulli PA (2018) Awake multimodal phenotyping for prediction of oral appliance treatment outcome. J Clin Sleep Med 14(11):1879–1887
Jugé L, Yeung J, Knapman FL, Burke PG, Lowth AB, Gan KZ, Brown EC, Butler JE, Eckert DJ, Ngiam J, Sutherland K (2021) Influence of mandibular advancement on tongue dilatory movement during wakefulness and how this is related to oral appliance therapy outcome for obstructive sleep apnea. Sleep 44(3):p.zsaa196
Berry RB, Quan SF, Abreu AR, Bibbs ML, DelRosso L, Harding SM, Mao MM, Plante DT, Pressman MR, Troester MM, Vaughn BV (2020) The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications. Am Acad Sleep Med
Qin H, Steenbergen N, Glos M, Wessel N, Kraemer JF, Vaquerizo-Villar F, Penzel T (2021) The different facets of heart rate variability in obstructive sleep apnea. Front Psych 12:642333
Mohammadieh AM, Dissanayake HU, Sutherland K, Ucak S, De Chazal P, Cistulli PA (2022) Does obstructive sleep apnoea modulate cardiac autonomic function in paroxysmal atrial fibrillation? J Interv Cardiac Electrophysiol 1–11
Kleiger RE, Miller JP, Bigger JT Jr, Moss AJ (1987) Decreased heart rate variability and its association with increased mortality after acute myocardial infarction. Am J Cardiol 59(4):256–262
Dissanayake HU, Bin YS, Sutherland K, Ucak S, de Chazal P, Cistulli PA (2022) The effect of obstructive sleep apnea therapy on cardiovascular autonomic function: a systematic review and meta-analysis. Sleep 45(12):p.zsac210
Kim JW, Kwon SO, Lee WH (2020) Nocturnal heart rate variability may be useful for determining the efficacy of mandibular advancement devices for obstructive sleep apnea. Sci Rep 10(1):1–8
Glos M, Penzel T, Schoebel C, Nitzsche GR, Zimmermann S, Rudolph C, Blau A, Baumann G, Jost-Brinkmann PG, Rautengarten S, Meier JC (2016) Comparison of effects of OSA treatment by MAD and by CPAP on cardiac autonomic function during daytime. Sleep Breath 20:635–646
Dal-Fabbro C, Garbuio S, D’Almeida V, Cintra FD, Tufik S, Bittencourt L (2014) Mandibular advancement device and CPAP upon cardiovascular parameters in OSA. Sleep Breath 18:749–759
Shiomi T, Guilleminault C, Sasanabe R, Hirota I, Maekawa M, Kobayashi T (1996) Augmented very low frequency component of heart rate variability during obstructive sleep apnea. Sleep 19(5):370–377
Sutherland K, Takaya H, Qian J, Petocz P, Ng AT, Cistulli PA (2015) Oral appliance treatment response and polysomnographic phenotypes of obstructive sleep apnea. J Clin Sleep Med 11(8):861–868
McEvoy RD, Antic NA, Heeley E, Luo Y, Ou Q, Zhang X, Mediano O, Chen R, Drager LF, Liu Z, Chen G (2016) CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med 375(10):919–931
Coruzzi P, Gualerzi M, Bernkopf E, Brambilla L, Brambilla V, Broia V, Lombardi C, Parati G (2006) Autonomic cardiac modulation in obstructive sleep apnea: effect of an oral jaw-positioning appliance. Chest 130(5):1362–1368
Acknowledgements
The authors gratefully acknowledge the prospective work conducted by investigators used in this study.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The data for this study came from three prospective studies. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Sydney Local Health District) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
P. A. C. has an appointment to an endowed academic Chair at the University of Sydney that was established from ResMed funding, has received research support from ResMed and SomnoMed, and is a consultant to ResMed, SomnoMed, Signifier Medical Technologies, Bayer, and Sunrise Medical. All other authors (S. U., H. D., K. S., Y. S. B., and, P. dC.) certify that they have no affiliations with or involvement in any organisation or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ucak, S., Dissanayake, H.U., Sutherland, K. et al. Effect of mandibular advancement splint therapy on cardiac autonomic function in obstructive sleep apnoea. Sleep Breath 28, 349–357 (2024). https://doi.org/10.1007/s11325-023-02924-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-023-02924-y