Abstract
Purpose
To determine the effects of a mandibular advancement appliance (MAA) on sequences of jaw-closing muscle activity (JCMA) and apneic or hypopneic event (AHE) in individuals with obstructive sleep apnea (OSA).
Methods
Individuals with OSA were included in a secondary analysis of a randomized controlled crossover trial, in which two ambulatory polysomnographic recordings were performed: one with MAA in situ and the other without MAA. A time span of 16 s between JCMA and AHE was applied to classify JCMAs into four sequences: (1) JCMA occurs before AHE (B-type); (2) both events occur simultaneously (S-type); (3) JCMA occurs after AHE (A-type); and (4) JCMA is time-unrelated to AHE (U-type). The effects of MAA on the distribution of these sequences were analyzed by Wilcoxon signed-rank test.
Results
Among 16 individuals (10 men, mean age 51.3 ± 8.5 years) baseline apnea–hypopnea index and JCMA index were 23.8 ± 16.0 events/h and 10.8 ± 10.3 events/h, respectively. In both conditions, i.e., without and with MAA, most JCMAs were U-type (48% and 65%, respectively), followed by A-type (41% and 22%), B-type (25% and 21%), and S-type (2% and 1%). With MAA in situ, only the A-type JCMA index decreased significantly (P = 0.005), while B-type, S-type, and U-type JCMA indices did not change significantly (all P > 0.05).
Conclusion
MAA therapy only significantly reduces the jaw-closing muscle activities that occur after apneic or hypopneic events in individuals with OSA.
Trial registration
www.clinicaltrials.gov (NCT02011425); December 13, 2013.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnea (OSA) is a sleep-related breathing disorder characterized by repetitive obstructions of the upper airway that may result in oxygen desaturations and arousals from sleep [1]. OSA is usually accompanied by loud snoring, morning headache, and excessive daytime sleepiness [1, 2]. The overall population prevalence of OSA ranges from 9 to 38%, and is higher in individuals with male gender, higher age, and obesity [3]. OSA has been reported to be a risk factor for several metabolic (e.g., diabetes, glucose dysregulation), cardiovascular (e.g., hypertension, stroke), psychiatric (e.g., depression), and sleep-related disorders (e.g., sleep bruxism, insomnia) [4, 5].
Jaw-closing muscle activity (JCMA) is an increased electromyography activity that commonly occurs during sleep in individuals with OSA [6]. JCMA includes rhythmic masticatory muscle activity (RMMA, i.e., muscle activity characterizing sleep bruxism) and orofacial activity (e.g., swallowing, yawning, lip movement, and sleep talking).
Previous studies suggested that in individuals with OSA, respiratory events (apneic or hypopneic events [AHEs]) are frequently followed by JCMA [7, 8], and JCMAs’ onset may be triggered by the AHEs [9, 10]. Two studies reported that the majority of RMMAs occurred after AHEs in individuals with OSA [11, 12], supporting a theory that JCMA in association with the termination of AHE may play a protective role by re-opening the upper airway [13]. However, other studies showed that most RMMAs were time-unrelated to AHEs [14, 15]. Therefore, the temporal relationship between JCMA and AHE in individuals with OSA is probably not characterized by one specific sequence of events at the level of the individual patient [13].
Additionally, some studies suggested that JCMA is a general motor response to sleep arousals [16,17,18]. In our primary study [19], in which the same sample was used as in the present study, we observed that the effect of a mandibular advancement appliance (MAA) significantly reduces JCMAs related to respiratory arousals in participants with OSA [19]. Based on this, we hypothesized that only JCMAs occurring after AHEs would decrease with MAA treatment, while JCMAs occurring before AHEs, during AHEs, and those time-unrelated to AHEs would not change. Furthermore, we hypothesized that only JCMAs occurring after AHEs in relation to respiratory arousals would decrease. Therefore, the primary aim of this study was to determine the effects of MAA on the distribution of sequences of JCMAs and AHEs. The secondary aim was to determine the effect of MAA on JCMA occurring after AHE in relation to arousal.
Methods
Study design
This study is a secondary analysis of a prospective randomized controlled crossover trial in which the effects of MAA therapy on JCMA in individuals with OSA were investigated [19]. This clinical study is registered at www.clinicaltrials.gov (NCT02011425). The scientific and ethical aspects of this study were approved by the Medical Ethics Committee of the Université de Montréal (13–105-CERES-D).
Participants
Individuals who were prescribed MAA therapy by a physician for their OSA were recruited in the primary study. The criteria of participants’ recruitment were described in detail by Aarab et al. [19]. In summary, participants aged between 30 and 65 years with an apnea–hypopnea index (AHI) of 15–45 events/h of sleep and OSA signs or symptoms (e.g., choking or gasping during sleep, daytime fatigue) were included. The exclusion criteria were as follows: presence of other respiratory or sleep disorders (except sleep bruxism), ongoing periodontal problems, reversible upper airway abnormalities, severe orofacial pain, or temporomandibular disorders; usage of medications that could influence respiratory or sleep; and lack of retention possibilities for an MAA.
Polysomnography
After a 3 to 6 months’ habituation period of wearing MAA (SomnoDent Flex; SomnoMed, Ontario, Canada), polysomnographic (PSG) recordings were conducted for participants at two conditions, i.e., without and with MAA in situ, in random order with an interval of 1 week to eliminate possible carryover effects. An ambulatory type II PSG system, Embla Titanium hardware (Embla, Ontario, Canada), was used to record the following channels: electroencephalography (F3M2, F4M1, C3M2, C4M1, O1M2, O2M1), electrooculography (left and right), electromyography (EMG; mentalis, masseter, temporalis, and tibialis muscles), airflow (nasal cannula), respiratory effort (abdominal and thoracic), oximetry, and sleep position.
PSG recordings, including standard sleep variables, respiratory events (e.g., apnea, hypopnea), and sleep arousals, were scored manually by an experienced and registered polysomnographic technologist from an independent company (Sleep Strategies, Ottawa, Canada), following the criteria of the American Academy of Sleep Medicine [20]. JCMAs (i.e., RMMA and orofacial activity) were scored by the first author (D.L.) according to previously published criteria [21]. EMG burst was scored when the mean amplitude was two times higher than the baseline EMG signal on at least three of the four EMG channels of the bilateral masseter and temporalis muscles. EMG burst was classified as phasic (duration: 0.25–2 s) or tonic (duration ≥ 2 s). EMG bursts occurring with an interval of shorter than 3 s were considered belonging to a single episode. Subsequently, an RMMA episode was scored as phasic (three or more continuous phasic EMG bursts), tonic (one or more tonic EMG bursts), or mixed (at least one phasic and one tonic EMG bursts). Orofacial activity was scored when EMG bursts did not meet the criteria for RMMA. The number of JCMA was defined as the sum of RMMA and orofacial activity.
The sequence of JCMA and AHE
Scored JCMAs were categorized into four possible sequences in association with AHE (see Fig. 1a): (1) JCMA occurs before AHE (B-type); (2) JCMA and AHE occur simultaneously (S-type); (3) JCMA occurs after AHE (A-type); and (4) JCMA is time-unrelated to AHE (U-type). A time span of 16 s [16, 22,23,24] was applied for the relation between the two events, starting at the termination of the preceding AHE or JCMA. When JCMA occurred between two AHEs and both time spans were within 16 s, the JCMA was scored twice, i.e., B-type and A-type. Consequently, the total percentages of the four types may be over 100%.
Relationship between jaw-closing muscle activity (JCMA), arousal, and apneic or hypopneic event (AHE). a Sequences of JCMA and AHE; b Respiratory or non-respiratory arousal-related JCMA. B-type, JCMA occurs before AHE; S-type, JCMA and AHE occur simultaneously; A-type, JCMA occurs after AHE; U-type, JCMA occurs before or after AHE but is time-unrelated to AHE
A-type JCMAs were considered in relation to arousal when they occurred within 5 s of the arousal [16, 19]. If the arousal occurred at the termination of or immediately after a respiratory event (i.e., AHE), the arousal was scored as respiratory arousal [25], and the JCMA was then scored as respiratory arousal-related JCMA [16, 19]. In contrast, if the arousal was scored as non-respiratory arousal, the JCMA was scored as non-respiratory arousal-related JCMA (see Fig. 1b).
Outcome variables
Some variables were transformed into indices, defined as the number of events per hour of sleep, such as the JCMA index. The primary outcome variables of this study were the indices of each sequence of JCMA, viz., B-type JCMA index, S-type JCMA index, A-type JCMA index, and U-type JCMA index. In order to compare our results with other studies, the number of JCMAs for each sequence was also expressed as a percentage of the total number of JCMAs. Secondary outcome variables were the index of A-type JCMA in relation to respiratory and non-respiratory arousal.
Statistical analysis
The normality of variables was tested by the Shapiro–Wilk test. Paired-samples t-tests or Wilcoxon signed-rank tests were used to compare variables between PSG recordings without and with MAA in situ. The effects of MAA on the indices of the four sequences of JCMA and the indices of A-type JCMA in relation to arousals were analyzed by the Wilcoxon signed-rank test. Statistical significance was set at 0.05. Statistical analysis was performed using SPSS Statistics (version 26, SPSS Inc., Chicago, IL, USA).
Results
Participants
Thirty-two individuals with OSA were initially invited to participate. For various reasons, fourteen individuals were excluded in the primary study of Aarab et al. [19]. In two participants, the scored AHEs were invisible on the respiratory traces of their PSG recordings. Hence, in this study, we included 16 participants, including 10 men. Mean age was 51.3 ± 8.5 years. Mean body mass index was 29.1 ± 3.6 kg/m2. Although all participants met the AHI criteria (i.e., 15–45 events/h) during their recruitment, 6 cases showed an AHI below 15 events/h in the PSG recordings without MAA in situ.
Descriptive analyses
Table 1 shows the descriptive analyses of the sleep, respiratory, and JCMA variables in PSG recordings without and with MAA in situ. The total sleep time and sleep efficiency did not show a significant difference between PSG recordings without and with MAA in situ. The percentage of sleep stage N2 decreased significantly with MAA in situ, while the percentage of stage N3 and REM increased significantly (P < 0.05). The AHI, oxygen desaturation index, total arousal index, and respiratory arousal index decreased significantly with MAA in situ (P < 0.05).
Sequences of JCMA and AHE
Table 2 shows the distribution of each sequence of JCMA and AHE without and with MAA in situ. In both conditions, i.e., without and with MAA in situ, the majority of JCMAs were classified as U-type (mean = 48% and 65%, respectively), followed by A-type (mean = 41% and 22%), and B-type (mean = 25% and 21%). Only a few JCMAs were scored as S-type (mean = 2% and 1%). In addition, without MAA, 16% (mean) of JCMAs were scored as both A-type and B-type, while with MAA in situ, the percentage of the double-scored JCMAs decreased to 8%.
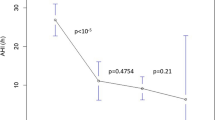
With MAA in situ, only the A-type JCMA index decreased significantly compared with that without MAA (P = 0.005), while B-type, S-type, and U-type JCMA showed no significant difference (P = 0.069, 0.401, and 0.501, respectively, see Table 2). This finding still holds after removing the double-scored JCMAs from A-type and B-type (for A-type, P = 0.023; for B-type, P = 0.326). Although the reduction of the B-type JCMA index was not significant, 10 of the 16 participants showed a decrease (Fig. 2). Also, in a few cases, the A-type, B-type, and U-type JCMA index increased with MAA in situ.
Individual values of JCMA index for each sequence without and with MAA in individuals with OSA. *A P value of < 0.05 is considered statistically significant; MAA, mandibular advancement appliance; JCMA, jaw-closing muscle activity; B-type, JCMA occurs before AHE; S-type, JCMA and AHE occur simultaneously; A-type, JCMA occurs after AHE; U-type, JCMA occurs before or after AHE but is time-unrelated to AHE
In addition, for the A-type JCMA index, only the respiratory arousal-related JCMA index decreased significantly with MAA in situ (0.40|2.15|3.67 vs 0.16|0.57|0.75, P = 0.001), whereas the non-respiratory arousal-related JCMA index did not show a significant difference (0.00|0.21|0.71 vs 0.00|0.06|0.37, P = 0.170).
Discussion
This study aimed to determine the effects of MAA on the distribution of the sequences of JCMA and AHE in individuals with OSA. Our results showed that MAA therapy significantly reduced only the JCMAs that occurred after AHEs in relation to respiratory arousals; not those that occurred after AHEs in relation to non-respiratory arousals, nor those that occurred before AHEs, during AHEs, or were time unrelated to AHEs.
A recent study demonstrated that both RMMA and other orofacial activities are involved in a cascade of arousal-related motor responses during sleep [26]. Considering that sleep arousals commonly follow AHEs in OSA, it can be assumed that RMMA and other orofacial activities have similar temporal relationship to AHEs. Also, the reliable distinction between RMMA and orofacial activities relies on audio–video recordings [21]. However, the PSG used in this study did not include such recordings. In addition, as previous studies on the temporal relationship between RMMAs/orofacial activities and AHEs are rare, especially for orofacial activity, comparisons between our study and others would be limited. For these reasons, in this study, we combined both types of oromotor events as JCMA to avoid potential bias as well as to analyze their temporal relationship to AHEs.
Currently, there is no evidence pointing out an appropriate time span to consider JCMA and AHE as being related [13]. Based on evidence gathered from several sources, we set the time span at 16 s. Hosoya et al. performed a PSG study to investigate the relationship between sleep bruxism and OSA, which concluded that RMMA is secondary to arousal that occurs after AHE [22]. Based on this, the time span between AHE and RMMA was regarded as involving three periods: (1) the time span between AHE and arousal; (2) the duration of arousal; and (3) the time span between arousal and RMMA. Based on the results of three studies, the time of the three periods was determined at 0.9 s [23], 10.8 s [24], and 4 s [16], respectively. Thus, by adding up the times of the three periods, the time span of AHE to RMMA was estimated to be 16 s. Based on this, when RMMA occurred after AHE within 16 s, we took these two events as being related and classified this RMMA into A-type. Unfortunately, to the best of our knowledge, available evidence regarding the possible time span between AHE and JCMA seems to be only available for RMMA that occur after AHE; not for RMMA that occur before AHE. Similarly, such evidence seems to be unavailable for orofacial activities. Therefore, in this study, we applied the 16-s rule to all types of JCMA and all possible sequences.
Theoretically, a longer time span would result in more JCMAs classified as AHE-related, i.e., B-type and A-type. This is indeed the case when comparing our results with those from Tsujisaka et al. [15] and those from Saito et al. [11]. In the study of Tsujisaka et al. [15], the 10-s rule resulted in around 40% of RMMAs and 18% of orofacial activities being related to AHEs, while the 16-s rule in our study resulted in 52% of JCMAs and the 5-min rule in the study of Saito et al. [11] even resulted in 80% of RMMA being related to AHEs.
Another possible reason for the discrepancy between these studies may be the participants’ OSA severity. The present study was composed of participants with OSA ranging from moderate to severe, while in the study of Tsujisaka et al. [15], which had a time-span setting comparable to ours, only mild cases were included. Also, our study showed more JCMAs that were associated with AHEs than the study of Tsujisaka et al. [15]. Based on this, we speculate that in severe cases, more JCMAs would be related to AHEs than in mild cases. This notion could support, at least partially, the expert opinion that the relative predominance of one specific sequence of events varies at the level of the individual patient [13]. Future studies are needed to confirm this hypothesis.
Corresponding to the four possible sequences of JCMA and AHE, four theories of the role of JCMA in OSA were hypothesized [13], viz., (1) B-type: JCMA may have an OSA-inducing effect; (2) S-type: the genesis of two events may share the same stimulus and mechanism; (3) A-type: JCMA may have a potential OSA-protective role; and (4) U-type: two events are unrelated. Furthermore, a possible predominant sequence would support one of these four theories. According to our results (Table 2), the most common JCMA was U-type (48.3%). On the one hand, the result means that around half of the JCMAs were time-unrelated to AHEs. On the other hand, it indicates that the other half of JCMAs were time-related to AHEs (i.e., B-type, A-type, or S-type). Thus, we could not conclude that these two events are unrelated. Since only a few JCMAs were scored as S-type, the presence of S-type seems like a coincidental occurrence of JCMA and AHE. Besides, 25.1% of JCMAs were scored as B-type. However, considering that part of B-type had an overlap with A-type JCMAs and that 10 of the 16 participants showed a reduction in B-type JCMA index with MAA therapy, these overlapping B-type JCMAs were more likely responses to the preceding AHEs and respiratory arousals. Given this, if we subtract the number of overlapping JCMAs (15.5%), the percentage of remaining B-type JCMA will be around 10%, which weakens the rationality of the hypothesis that JCMA has an OSA-inducing effect. Finally, 40.1% of JCMAs were scored as A-type, and most A-type JCMAs were related to respiratory arousals. These results suggest that A-type JCMA is a response to preceding AHE and respiratory arousal, supporting the hypothesis that some JCMAs may have a positive protective role against OSA [13].
Based on our results, we accepted the hypothesis that only A-type JCMA would decrease with MAA therapy, while B-type, S-type, and U-type JCMA would not change. Also, we accepted our second hypothesis that only JCMA occurring after AHE in relation to respiratory arousal would decrease with MAA treatment. These results imply that with MAA therapy, the reduction of the A-type JCMA index is mainly due to the decrease of respiratory arousal-related A-type JCMA. Besides, our results showed that with MAA in situ, only respiratory arousals decreased significantly; not non-respiratory arousals. Considering all this evidence, we can speculate that successful MAA treatment may effectively reduce JCMA through decreasing respiratory arousal. Furthermore, the efficacy of MAA in reducing JCMA in individuals with OSA may vary at an individual level depending on the proportion of A-type JCMA related to respiratory arousal in the total JCMA.
Additionally, previous studies reported that in some cases, MAA might not be effective in managing OSA, or even aggravate the condition of OSA [26, 27]. Given this, it is not surprising that a few cases in our sample showed an increase in the A-type JCMA index. Furthermore, it could be hypothesized that MAA responders may show a higher reduction in the JCMA index than non-responders in individuals with OSA. Similar to A-type, the U-type JCMA index also displayed an increase in several cases with MAA in situ, suggesting that MAA may increase the frequency of JCMA, even in individuals without OSA. This has also been reported in previous studies [28, 29].
Although this study was performed in participants with OSA and not in a population with comorbid sleep bruxism, based on our results and considering the fact that RMMA is a common muscle activity observed in both OSA and healthy individuals [30], we hypothesize that only RMMAs that occur after AHEs in relation to arousals would be improved by MAA therapy in individuals with OSA. Also, the proportion of respiratory arousal-related A-type RMMAs may be able to predict the efficacy of MAA on reducing the comorbid sleep bruxism in individuals with OSA. Future studies are needed to verify these hypotheses in individuals with both OSA and sleep bruxism.
Although this study provides new findings on the relationship between JCMA and AHE, several limitations should be noted. First, we did not perform an a priori sample size calculation. However, based on the post hoc power analysis, a sample of 11 would be enough to detect the difference in the A-type JCMA index with MAA therapy. Thus, the significant reduction of A-type JCMA with MAA in situ found in this study is considered reliable. Second, the time span setting for scoring sequences was based on limited and indirect evidence. A specifically designed study is needed to define a solid evidence-based time span at which JCMA or RMMA and AHE can be considered being related.
Conclusion
This study showed that effective mandibular advancement appliance therapy in individuals with obstructive sleep apnea reduces only the jaw-closing muscle activities that occur after respiratory events with arousals; not those that occurred after AHEs in relation to non-respiratory arousals, nor those that occurred before AHEs, during AHEs, or were time-unrelated to AHEs. These results suggest that mandibular advancement appliances can relieve jaw-closing muscle activities that are secondary to OSA, and that the efficacy may vary at the level of individual patients depending on the distribution of jaw-closing muscle activities that occur after respiratory events.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Redline S (2005) Obstructive sleep apnea: phenotypes and genetics. In: Kryger MH, Roth T, Dement WC (eds) Principles and practice of sleep medicine, Sixth edit. Elsevier, Philadelphia, PA, pp 1102–1109
Semelka M, Wilson J, Floyd R (2016) Diagnosis and treatment of obstructive sleep apnea in adults. Am Fam Physician 94:355–360. https://doi.org/10.1037/e676562012-001
Senaratna CV, Perret JL, Lodge CJ et al (2017) Prevalence of obstructive sleep apnea in the general population: a systematic review. Sleep Med Rev 34:70–81. https://doi.org/10.1016/j.smrv.2016.07.002
Jordan AS, McSharry DG, Malhotra A (2014) Adult obstructive sleep apnoea. Lancet 383:736–747
Ohayon MM, Li KK, Guilleminault C (2001) Risk factors for sleep bruxism in the general population. Chest 119:53–61. https://doi.org/10.1378/chest.119.1.53
Kato T, Masuda Y, Yoshida A, Morimoto T (2011) Masseter EMG activity during sleep and sleep bruxism. Arch Ital Biol 149:478–491. https://doi.org/10.4449/aib.v149i4.1317
Hollowell DE, Suratt PM (1989) Activation of masseter muscles with inspiratory resistance loading. J Appl Physiol 67:270–275. https://doi.org/10.1152/jappl.1989.67.1.270
Hollowell DE, Suratt PM (1991) Mandible position and activation of submental and masseter muscles during sleep. J Appl Physiol 71:2267–2273. https://doi.org/10.1152/jappl.1991.71.6.2267
da Costa Lopes AJ, Cunha TCA, Monteiro MCM et al (2020) Is there an association between sleep bruxism and obstructive sleep apnea syndrome? A systematic review. Sleep Breath 24:913–921. https://doi.org/10.1007/s11325-019-01919-y
Lavigne GJ, Huynh N, Kato T et al (2007) Genesis of sleep bruxism: motor and autonomic-cardiac interactions. Arch Oral Biol 52:381–384. https://doi.org/10.1016/j.archoralbio.2006.11.017
Saito M, Yamaguchi T, Mikami S et al (2014) Temporal association between sleep apnea-hypopnea and sleep bruxism events. J Sleep Res 23:196–203. https://doi.org/10.1111/jsr.12099
Okeson JP, Phillips BA, Berry DT et al (1991) Nocturnal bruxing events in subjects with sleep-disordered breathing and control subjects. J Craniomandib Disord 5:258–264
Manfredini D, Guarda-Nardini L, Marchese-Ragona R, Lobbezoo F (2015) Theories on possible temporal relationships between sleep bruxism and obstructive sleep apnea events. An expert opinion Sleep Breath 19:1459–1465. https://doi.org/10.1007/s11325-015-1163-5
Sjöholm TT, Lowe AA, Miyamoto K et al (2000) Sleep bruxism in patients with sleep-disordered breathing. Arch Oral Biol 45:889–896. https://doi.org/10.1016/S0003-9969(00)00044-3
Tsujisaka A, Haraki S, Nonoue S et al (2018) The occurrence of respiratory events in young subjects with a frequent rhythmic masticatory muscle activity: a pilot study. J Prosthodont Res 62:317–323. https://doi.org/10.1016/j.jpor.2017.12.004
Kato T, Rompré P, Montplaisir JY et al (2001) Sleep bruxism: an oromotor activity secondary to micro-arousal. J Dent Res 80:1940–1944. https://doi.org/10.1177/00220345010800101501
Carra MC, Rompré PH, Kato T et al (2011) Sleep bruxism and sleep arousal: an experimental challenge to assess the role of cyclic alternating pattern. J Oral Rehabil 38:635–642. https://doi.org/10.1111/j.1365-2842.2011.02203.x
Huynh N, Kato T, Rompré PH et al (2006) Sleep bruxism is associated to micro-arousals and an increase in cardiac sympathetic activity. J Sleep Res 15:339–346. https://doi.org/10.1111/j.1365-2869.2006.00536.x
Aarab G, Arcache P, Lavigne GJ et al (2020) The effects of mandibular advancement appliance therapy on jaw-closing muscle activity during sleep in patients with obstructive sleep apnea: a 3–6 months follow-up. J Clin Sleep Med 16:1545–1553. https://doi.org/10.5664/jcsm.8612
Berry RB, Budhiraja R, Gottlieb DJ et al (2012) Rules for scoring respiratory events in sleep: update of the 2007 AASM manual for the scoring of sleep and associated events. J Clin Sleep Med 8:597–619. https://doi.org/10.5664/jcsm.2172
Carra MC, Huynh N, Lavigne GJ (2015) Diagnostic accuracy of sleep bruxism scoring in absence of audio-video recording: a pilot study. Sleep Breath 19:183–190. https://doi.org/10.1007/s11325-014-0986-9
Hosoya H, Kitaura H, Hashimoto T et al (2014) Relationship between sleep bruxism and sleep respiratory events in patients with obstructive sleep apnea syndrome. Sleep Breath 18:837–844. https://doi.org/10.1007/s11325-014-0953-5
Simms T, Brijbassi M, Montemurro LT, Bradley TD (2013) Differential timing of arousals in obstructive and central sleep apnea in patients with heart failure. J Clin Sleep Med 9:773–779. https://doi.org/10.5664/jcsm.2918
Schwartz DJ, Moxley P, Barker A, Longman M (2005) On a characteristic of cortical arousals in individuals with obstructive sleep apnea.
Kato T, Katase T, Yamashita S et al (2013) Responsiveness of jaw motor activation to arousals during sleep in patients with obstructive sleep apnea syndrome. J Clin Sleep Med 9:759–765. https://doi.org/10.5664/jcsm.2914
Barnes M, McEvoy RD, Banks S et al (2004) Efficacy of positive airway pressure and oral appliance in mild to moderate obstructive sleep apnea. Am J Respir Crit Care Med 170:656–664. https://doi.org/10.1164/rccm.200311-1571OC
Bloch KE, Iseli A, Zhang JN et al (2000) A randomized controlled crossover trial of two oral appliances for sleep apnea treatment. Am J Respir Crit Care Med 162:246–251. https://doi.org/10.1164/ajrccm.162.1.9908112
Landry M-L, Rompré PH, Manzini C et al (2006) Reduction of sleep bruxism using a mandibular advancement device: an experimental controlled study. Int J Prosthodont 19:549–556
Landry-Schönbeck A, de Grandmont P, Rompré PH, Lavigne GJ (2009) Effect of an adjustable mandibular advancement appliance on sleep bruxism: a crossover sleep laboratory study. Int J Prosthodont 22:251–259
Lavigne GJ, Rompré PH, Poirier G et al (2001) Rhythmic masticatory muscle activity during sleep in humans. J Dent Res 80:443–448. https://doi.org/10.1177/00220345010800020801
Acknowledgements
The authors thank SomnoMed (Ontario, Canada) for supplying free of charge the SomnoDent appliances used in this study. Deshui Li is sponsored by the China Scholarship Council, China; the grant has no role in the conception, design, and execution of this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to study design, data acquisition and analyses, manuscript preparation, and revision. All authors have approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Medical Ethics Committee of the Université de Montréal (13–105-CERES-D).
Consent
Informed consent was obtained from all individual participants included in the study.
Competing interests
The SomnoDent appliances (SomnoMed, Ontario, Canada) were provided free of charge. SomnoMed had no input in the data analyses and publication of this article. Deshui Li is sponsored by the China scholarship council (CSC). Frank Lobbezoo is a member of the Academic Advisory Boards for GrindCare and Oral Function of Sunstar Suisse S.A. and receives research grants from Sunstar Suisse S.A., SomnoMed, VIVISOL B.V., ResMed, Health Holland, and Airway Management. Ghizlane Aarab is a member of the Academic Advisory Board for Oral Function of Sunstar Suisse S.A. and receives research grants from Sunstar Suisse S.A., SomnoMed, VIVISOL B.V., ResMed, and Health Holland. Gilles J. Lavigne received a Canada Research Chair on Pain, Sleep and Trauma during the realization of the protocol and had no other conflict of interest related to the present work. Patrick Arcache and Nelly Huynh had no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, D., Aarab, G., Lobbezoo, F. et al. The effects of mandibular advancement appliance therapy on the sequence of jaw-closing muscle activity and respiratory events in individuals with obstructive sleep apnea. Sleep Breath 27, 757–764 (2023). https://doi.org/10.1007/s11325-022-02624-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-022-02624-z