Abstract
Purpose
To find out if a moderate protrusion with a mandibular advancement device (MAD) can significantly increase the upper airway volume and, further, what signs and symptoms of obstructive sleep apnea (OSA) can be improved by this maneuver.
Methods
There were 58 adults diagnosed with OSA who were referred for MAD therapy. The mean apnea-hypopnea index (AHI) was 19.2 (SD 8.6). Five indicators of signs and symptoms of OSA (AHI, oxygen saturation, snoring, daytime sleepiness, and health-related quality of life) were evaluated at the baseline and after 6 months of MAD therapy. Nasal resistance and airway volume and cross-sectional areas with and without the MAD in situ were recorded. Based on AHI reduction, the treatment response was classified as complete, partial, or non-complete. Statistical analyses included the chi-square, t tests, Mann–Whitney U tests, and regression analyses (linear and logistic).
Results
Twenty-three patients attained a complete response (residual AHI < 5 events/h) to MAD therapy. In 13 subjects, the response was partial, and in 9 patients, it was non-complete. The complete responders were significantly younger, and they had a deeper overbite than partial/non-complete responders. A convex profile associated positively, but a vertically restricted throat and increased lower facial height associated negatively with the increase in airway volume.
Conclusions
Excellent MAD therapy outcomes were achieved in most patients. Only age and deep bite had some influence on AHI reduction, indicating multifactorial nature in the response to MAD therapy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In obstructive sleep apnea syndrome (OSAS), there are recurrent episodes of partial or complete obstruction of the upper airway during sleep, resulting in sleep fragmentation and oxygen desaturations. OSA has been identified as an independent risk factor for cardiovascular diseases [1] and is associated with reduced quality of life, increased healthcare utilization, and mortality [2, 3]. Continuous positive airway pressure (CPAP) is the gold standard of treatment, but its effectiveness is limited by poor compliance and intolerance. Although oral devices are not as effective as CPAP therapy in reducing the apnea-hypopnea index (AHI), they offer an alternative for patients with mild to moderate OSA who are unable to tolerate CPAP therapy.
There is no consensus regarding the most effective mandibular advancement with oral devices. In an earlier review, it was suggested that the more the mandible is stretched forward, the more the AHI improves [4], while in the recent review and meta-analysis, it was concluded that the AHI improvement is not proportional to the increase of mandibular advancement [5]. Since OSA patients may suffer from different underlining pathophysiologies such as an upper airway anatomical abnormality, increased pharyngeal collapsibility, an overly sensitive ventilator control system, or a reduced arousal threshold, the inter-individual response to mandibular advancement device (MAD) therapy also varies [6]. The focus of this study was to find out if a moderate mandibular advancement (60%) with MAD can significantly increase the upper airway volume and, further, what signs and symptoms of OSA can be improved by this maneuver.
Subjects and methods
Subjects
All patients in this study were diagnosed as having OSA by ambulatory polygraphic recording (APR) and were referred by sleep medicine specialists for oral device treatment to the Oral and Maxillofacial Department, Kuopio University Hospital (KUH). Patients were consecutively recruited from the autumn of 2016 to the end of the year 2017. Subjects were enrolled if they were 18 years or older, their AHI was at least 10 events/h, their body mass index (BMI) was less than 35 kg/m2, and they had at least 5 teeth/jaw. This study protocol was approved by the Research Ethics Committee of the Hospital District of Northern Savo, Kuopio, Finland (7 February 2017; 80/2017). All patients provided written informed consent before participating in the study. The study flowchart is presented in Fig. 1.
Study flowchart. CBCT, cone-beam computed tomography; ESS, Epworth sleepiness scale; MAD, mandibular advancement device; MMA, maxillomandibular advancement surgery; OSA, obstructive sleep apnea; SSI, snoring symptoms inventory; TMD, temporomandibular disorders; 15D, 15D health-related quality of life
Methods
Five indicators of signs and symptoms of OSA (AHI, arterial oxyhemoglobin saturation (SaO2), snoring symptoms inventory (SSI), Epworth sleepiness scale (ESS), 15 D health-related quality of life (15D)) were evaluated in this study. Besides, changes in nasal resistance with the MAD in situ and upper airway volume and minimal cross-sectional areas with the MAD in situ were recorded. Data were also collected on age, gender, marital status, education, BMI, smoking, alcohol consumption, comorbidity, prescribed medication, and previous treatment of OSA.
Ambulatory polygraphic recording
Nocturnal single-night ambulatory polygraphic recordings were conducted to diagnose OSA prior to this study and after 6 months with the MAD in situ. Home APRs were conducted by using either the Embletta (Embletta, Embla, Broomfield, CO, USA) or Nox A1 ambulatory polygraphic device (Nox Medical, Reykjavík, Iceland) with accepted guidelines for diagnosing OSA. Apneas and hypopneas were automatically scored (Remlogic, version 3.2, and Somnologica, version 3.2 software, Embla Co., Broomfield, CO, USA) and manually verified and edited. Apnea was defined as a cessation (≥ 90%) of airflow for ≥ 10 s. Determination of hypopneas was based on a decrease (≥ 30%) in airflow for ≥ 10 s and associated with oxygen desaturation ≥ 4. Trained physicians evaluated the recordings. The following parameters were assessed: AHI, supine AHI, SaO2, percentage of SaO2 below 90%, and proportion of sleep time spent snoring. Positional OSA was defined as a difference of 50% or more in apnea index between supine and non-supine sleep positions [7]. Based on the second APRs, patients were classified according to the treatment success of MAD therapy as follows: complete responders (residual AHI < 5 events/h), partial responders (residual AHI > 5 events/h, decrease in baseline AHI > 50%), and weak responders (residual AHI > 5 events/h, decrease in baseline AHI < 50%).
Daytime sleepiness and quality-of-life measurements
In order to evaluate the discomfort in life due to OSA and snoring, the subjects were asked to fulfill ESS, SSI, and 15D questionnaires at the baseline and 6 and 12 months after MAD therapy. Daytime sleepiness was assessed using ESS score (range 0–24), with a score of 11 or more being indicative of excessive daytime sleepiness [8]. The SSI score was composed of two components: the social-work and the physical-embarrassment component including 25 symptoms (range 0–100), with a score of 0 corresponding to no problems with snoring [9]. Health-related quality of life among patients was assessed with the 15D questionnaire [10]. It contains 15 dimensions of life (questions), each having five different levels of functional status. These dimensions can be presented as a 15-dimensional profile or as a single index score. The score can vary between 0 and 1, a low score indicating more pathology than a high score.
Clinical examination
Clinical data included occlusal findings (molar occlusion, overjet, overbite, crossbite, open bite, scissor bite, crowding, palatal morphology) assessed according to the modified method of Björk [11], examination of temporomandibular joints (TMJs), bite muscles, mandibular movements [12], craniofacial morphology (visual evaluation of the facial profile based on the angle formed by the soft tissue glabella, subnasale, and soft tissue pogonion, anteroposterior jaw relationships, and vertical proportions) [13, 14], and the Mallampati score [15]. The cephalometric measurements were not included in the original study protocol. Measurements were repeated at 3, 6, and 12 months after MAD therapy. All the examinations were based on routine orthodontic clinical examinations done by the same experienced orthodontist (RP). In our previous study [16], the accuracy of the evaluation of the facial profile by this method was good (kappa value 0.920 with the agreement of 93.3%).
Rhinomanometric measurement
Rhinomanometry was performed 3 months after MAD therapy. An NR6-rhinomanometer (GM Instruments Ltd., Kilwinning, UK) was used to conduct the rhinomanometric recordings, in which total inspiratory nasal resistance was recorded at a radius of 150 Pa. Recordings were made in the supine position without MAD and MAD in situ. Nasal decongestion was not used.
Upper airway imaging
Images were taken about 6 months after MAD therapy on a CBCT machine with Romexis software (Promax 3D Max: Planmeca, Helsinki, Finland). CBCT imaging was done in an upright position with the Frankfort horizontal line parallel to the floor. The patients were advised to keep their head in an upright position, hold light intercuspidation, and breathe normally but not to swallow or do any conscious movements during the imaging and measurement (13 s).
The airway analysis tool was used to define the portion of interest and to calculate the volume and cross-sectional areas of each section. The sections were determined using the boundaries described elsewhere [17]. The image orientation and selection of sensitive threshold values were conducted manually. There were two experienced oral radiologists who measured the parameters.
Mandibular advancement device therapy
Patients were treated with the SomnoDent Flex (SomnoMed Ltd., Sydney, Australia) custom-made acrylic duo block titratable oral device. In this device, the upper and lower splints are connected by adjustable interlocking acrylic buccal extensions. In order to determine the desired amount of mandibular advancement, the inter-occlusal bite was registered with the SOMGauge bite registration device to obtain 60% of the maximal protrusion of the mandible.
Statistical analysis
Statistical analyses were performed using the IBM SPSS statistics, version 22.0 (IBM Corp., Armonk, NY, USA). For the statistical analysis, partial and non-complete responders were combined. The chi-square test was used to analyze the differences in categorical variables between males and females and between complete and partial/non-complete responders. Fisher’s exact test was used when the numbers of subjects in some cells were small. The differences in continuous variables were analyzed using Student’s t test for normally distributed variables and the Mann–Whitney U test for variables with skewed distributions. Multivariate linear regression analysis was used to investigate the associations of age, gender, baseline and positional AHI, convex profile, extreme overjet, increased lower facial height, decreased palatal width, and vertically restricted throat with an increase in total airway volume. Furthermore, logistic regression analysis was used to study the association between treatment response dichotomized complete response vs. partial/non-complete response as a dependent variable and age, gender, BMI, convex profile, overjet, overbite, cross bite, increase in airway volume, and vertically restricted throat. The independent variables were added simultaneously in regression analyses and their choices were based on previous literature which suggests that they are related to sleep-disordered breathing and upper airway volume. Also, some of them contributed to the outcomes in bivariate analyses. Associations with p values of < 0.05 were considered statistically significant.
The intra- and inter-examiner reproducibility of CBCT measurements for airway volume and minimum cross-sectional areas were investigated by repeating measurements (n = 20) of those variables 2 weeks apart by two experienced oral radiologist using intra-class correlation coefficients (ICC) and its 95% confidence interval (CI). The intra-examiner repeatability of CBCT measurements varied from 0.945 (hypopharynx volume) to 0.996 (minimal cross-sectional area of the oropharynx), but it was only fair for the minimal cross-sectional area of the nasopharynx (0.387). The inter-examiner repeatability varied from 0.774 (minimal cross-sectional area of nasopharynx) to 0.995 (volume of oropharynx).
Results
During the recruitment period, 39 males and 19 females diagnosed to have OSA were consecutively referred for MAD therapy. The mean AHI of the subjects was 19.2 (SD 8.6), with no gender difference (Table 1). The mean age of the subjects was 50.7 years (SD 11.9). On average, women were significantly older than men, were more often single, and were more highly educated than men. In half of the participants (49%), OSA was positional, and fifteen patients had tried CPAP therapy.
More than half of the patients (n = 23) attained a complete response (AHI < 5/h) to MAD therapy; in 13 subjects, the response was partial, and in 9 patients, it was non-complete. The results showed that the complete responders were significantly younger, and they had a deeper overbite than partial/ non-complete responders (Table 2). MAD therapy significantly reduced AHI and increased SaO2 almost significantly (Table 3). Also snoring, the SSI score, and daytime sleepiness were significantly reduced. The increases in total upper airway volume and oropharyngeal volume with MAD in situ were both significant. Also the minimum cross-sectional areas increased significantly in the nasopharynx, oropharynx, and hypopharynx. Further, the decrease in nasal resistance with MAD in situ was significant.
In multivariate linear regression analyses, a convex profile associated positively but a vertically restricted throat and increased lower facial height associated negatively with the increase in upper airway volume (Table 4). No factor that influenced the response of MAD therapy on AHI was identified by logistic regression model using the criterion of complete response as a dependent variable (data not shown).
Discussion
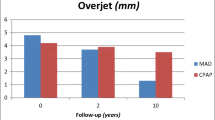
In the present study, convex facial profile proved to be a predictive factor for an improvement in the total pharyngeal airway volume with MAD therapy, whereas increased lower facial height and a vertically restricted throat seemed to worsen treatment outcome. In subjects with a convex profile, the overjet was significantly larger and the mean mandibular protrusion capacity was greater than in those with a straight or concave profile (Pahkala et al. MS). Thus, in subjects with a convex profile, the mandibular advancement (60%) in MAD was also larger, further increasing the pharyngeal airway volume. Interestingly, although not significantly related, the association between an extreme overjet and the increase in airway volume was negative. This tendency is parallel to previous 2D cephalometric and 3D volumetric studies on jaw orthopedic patients with mandibular and maxillomandibular advancements [18, 19], indicating that there is a certain limit after which no further improvement in airway space can be achieved after increasing the mandibular advancement. If this were also the case in MAD therapy, then a moderate mandibular advancement with MAD should be recommended. In addition, increased lower facial height seemed to limit the airway volume increase with treatment. This kind of craniofacial anatomy predisposes the mandible to rotate clockwise with the MAD in situ, thus reducing the retrolingual pharyngeal space.
Clear improvement in AHI by MAD therapy has been found to correlate to a retrognathic mandible [20], large upper inter-molar width [21], and wide lower inter-canine width [22]. In the present report complete responders had significantly deeper (over)bite and they tended to have more often mandibular retrusion than partial /non-complete responders. Our findings indicate that in subjects with a deep bite the intermaxillary space for the tongue in the oral cavity is reduced, which is then normalized by mandibular advancement with MAD. Thus our finding strengthens the previous suggestion [22] that the ratio between the tongue and bony enclosure size may be a predictive factor for the response of MAD therapy. Besides changing the position of the jaw and tongue MAD therapy is likely to increase the m. genioglossus tone and thus reduce the collapsibility of the upper airways. Furthermore, it has been found that in patients with Mallampati classes 3 and 4 the incidence of sleep apnea is high [23]. In the linear regression analysis, a vertically restricted throat (Mallampati class 4) was negatively related to an improvement in upper airway volume, indicating that soft tissue enlargement in the throat impairs the treatment response to MAD therapy.
Altogether low-dose CBCT proved to be an ideal diagnostic aid to study the pharyngeal airway in 3D. In the present study, the validity of CBCT to assess the upper airway space was good since both intra- and inter-examiner consistencies were high and acceptable. Generally, an increase in the airway volume increases airway flow and thus is likely to improve AHI and oxygen saturation level. Our results confirm this perception. The most critical site that causes maximum resistance to airflow is the smallest cross-section area of the airway. In our study, the smallest mean cross-sectional area in the pharynx was in the oropharyngeal region, which was increased 202 mm2 by MAD in situ in complete responders and half of that in partial/non-complete responders.
Concerning the effect of age on MAD treatment success, previous studies are contradictory [21, 22, 24]. In the present sample, complete responders were significantly younger than partial/non-complete responders. As observed previously, natural pharyngeal structures continue to grow until 13 years of age [25] and begin to narrow from about 20 until 50 years most likely because the soft palate becomes longer and thicker [26]. This narrowing might also be due to weight gain and an increase in the pharyngeal fat pad.
In normal subjects, upper airway resistance is lower during sleep when breathing through the nose. With nasal obstruction nose breathing switches to mouth breathing which is associated with increased propensity to OSA [27]. Nasal resistance is suggested to be elevated in patients with OSA [28, 29], but it is not related to the severity of OSA [30], or to AHI [31]. It also seems that high nasal resistance is a result rather than a cause of OSA, since nasal resistance decreases after tonsillectomy and AHI reduction [32]. Our result is parallel to this finding since nasal resistance was decreased significantly by upper airway enlargement with MAD in situ.
Our results showed that the prevalence of snoring was significantly reduced by MAD therapy. However, some patients still complained about persistent snoring, which may partly be related to the device design allowing mouth opening during sleep [33]. Interestingly, although in partial/non-complete responders AHI decreased less but the SSI score more than in complete responders, the adherence to MAD therapy seemed to be equal in the groups (Pahkala et al. MS). This underlines patients’ interest to reduce snoring in order to eliminate disturbances in everyday life. In the present study, the mean ESS scores both at the baseline and 6 months after MAD therapy were within the normal limit, which agrees the suggestion that mild to moderate OSA causes less pronounced daytime sleepiness than previously assumed [33]. Concerning the 15D quality of life, certain dimension scores such as “sleeping,” “discomfort and symptoms,” “psychological distress,” and “vitality,” however, were lower at the baseline than after using MAD for 6 months, indicating some minor improvements in those dimensions of life (Fig. 2).
15D health-related quality of life dimension scores at the baseline and 6 months after MAD therapy. Move, mobility; see, vision; hear, hearing; sleep, sleeping; eat, eating; speech, speech; excret, excretion; uact, usual activities; mental, mental function; disco, discomfort and symptoms; depr, depression; distr, distress; vital, vitality; sex, sexual activity
There is a variety of definitions of good MAD therapy response, and a lack of consensus around this issue makes the comparison between studies difficult. Although MAD therapy on OSA has not been reported to be as effective as that of CPAP treatment, we suggest that the criteria concerning good MAD therapy outcome should be more ambitious with its goal being parallel to that of CPAP treatment. In previous studies, the complete response to MAD therapy has been reported to vary from 29 to 71% (mean 48%) [34]. In the present study, AHI normalized in 51% of the patients, indicating good treatment response.
All the patients in this sample were consecutively referred for MAD treatment from the catchment area of KUH, and thus, the referral bias is unlikely. The percentage of withdrawals (29%) in this study was in line with similar studies and is most likely due to consecutive patient recruitment protocol. Certain limitations should be noted when interpreting the results. In this study, the ambulatory polygraphic recordings were performed mainly in KUH, and also in other institutes. However, all analyses were made according to the standard AASM respiratory rules [35]. Rule 4A was used for scoring the hypopnea events. Furthermore, CBCT imaging was done in an upright position with the Frankfort horizontal line parallel to the floor. In order to capture the true upper airway dimensions and volume in an upright position, the patients should have been positioned in the natural head position since manual placing of the patient in a defined position has an effect on the airway parameters. However, neither of these upright positions during imaging reflects the true anatomy while sleeping in bed, and thus, the methodological bias is likely. In the present study, the CBCT images with and without MAD were taken consecutively thus eliminating the possible interference of seasonal change or different patient positioning during CBCT scannings.
Regarding the questionnaires, it should be noticed that not all the patients returned every questionnaire despite previously given instructions. Mainly the 15D questionnaire was omitted. Also some patients did not want the CBCT images to be taken, and in some patients, the rhinomanometric measurements failed due to nasal obstruction.
In summary, this study shows that even excellent treatment outcomes among OSA patients can be achieved with moderate mandibular advancement using an oral device. With this protocol, also the upper airway space improves remarkably. We also presume that patients’ adherence is good and that there are few side effects such as temporomandibular disorders and occlusal changes with this treatment protocol. Our further studies will focus on those issues.
Conclusions
Excellent MAD therapy outcomes were achieved in most OSA patients with moderate mandibular advancement. A convex profile proved to be a predictive factor for a greater increase in pharyngeal airway volume, while increased lower facial height and a vertically restricted throat limited the airway volume increase. Despite some differences in the variables between complete and partial/non-complete responders, only age and deep bite seemed to have some influence on the effectiveness of MAD therapy. This indicates a multifactorial nature both in the pathophysiology of OSA and in the response to MAD therapy.
References
Peppard PE, Young T, Palta M, Skatrud J (2000) Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 342(19):1378–1384
Young T, Finn L, Peppard PE, Szklo-Coxe M, Austin D, Nieto FJ, Stubbs R, Hla KM (2008) Sleep disordered breathing and mortality: eighteen-year follow-up of the Wisconsin sleep cohort. Sleep 31(8):1071–1078
Flemons WW, Reimer MA (2002) Measurement properties of the Calgary sleep apnea quality of life index. Am J Respir Crit Care Med 165(2):159–164
Ferguson KA, Cartwright R, Rogers R, Schmidt-Nowara W (2006) Oral appliances for snoring and obstructive sleep apnea: a review. Sleep 29(2):244–262
Bartolucci ML, Bortolotti F, Raffaelli E, D’Antò V, Michelotti A, Alessandri Bonetti G (2016) The effectiveness of different mandibular advancement amounts in OSA patients: a systematic review and meta-regression analysis. Sleep Breath 20(3):911–919. https://doi.org/10.1007/s11325-015-1307-7
Edwards BA, Andara C, Landry S, Sands SA, Joosten SA, Owens RL, White DP, Hamilton GS, Wellman A (2016) Upper-airway collapsibility and loop gain predict the response to oral appliance therapy in patients with obstructive sleep apnea. Am J Respir Crit Care Med 194(11):1413–1422
Cartwright RD (1984) Effect of sleep position on sleep apnea severity. Sleep 7(2):110–114
Johns MW (1991) A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 14(6):540–545
Douglas SA, Webster S, El Badawey MR, Drinnan M, Matthews JN, Gibson GJ, Wilson JA (2006) The development of a snoring symptoms inventory. Otolaryngol Head Neck Surg 134(1):56–62
Sintonen H (2001) The 15D instrument of health-related quality of life: properties and applications. Ann Med 33(5):328–336
Björk A, Krebs B, Solow B (1964) A method for epidemiological registration of malocclusion. Acta Odontol Scand 22:27–41
Helkimo M (1974) Studies on function and dysfunction of the masticatory system. II. Index for anamnestic and clinical dysfunction and occlusal state. Swed Dent J 65:7–21
Proffit WR (2001) Contemporary orthodontics. Mosby, St. Louis
Reyneke JP (2003) Essentials of orthognathic surgery. Quintessence Publishing, Carol Stream, IL
Mallampati SR, Gatt SP, Gugino LD, Desai SP, Waraksa B, Freiberger D, Liu PL (1985) A clinical sign to predict difficult tracheal intubation: a prospective study. Can Anaesth Soc J 32(4):429–434
Ikävalko T, Närhi M, Lakka T, Myllykangas R, Tuomilehto H, Vierola A, Pahkala R (2015) Lateral facial profile may reveal the risk for sleep disordered breathing in children--the PANIC-study. Acta Odontol Scand 73(7):550–555. https://doi.org/10.3109/00016357.2014.997795
Feng X, Li G, Qu Z, Liu L, Näsström K, Xie-Qi S (2015) Comparative analysis of upper airway volume with lateral cephalograms and cone-beam computed tomography. Am J Orthod Dentofac Orthop 147(2):197–204. https://doi.org/10.1016/j.ajodo.2014.10.025
Riepponen A, Myllykangas R, Savolainen J, Kilpeläinen P, Kellokoski J, Pahkala R (2017) Changes in posterior airway space and hyoid bone position after surgical mandibular advancement. Acta Odontol Scand 75(1):73–78. https://doi.org/10.1080/00016357.2016.1252851
Bianchi A, Betti E, Tarsitano A, Morselli-Labate AM, Lancellotti L, Marchetti C (2014) Volumetric three-dimensional computed tomographic evaluation of the upper airway in patients with obstructive sleep apnoea syndrome treated by maxillomandibular advancement. Br J Oral Maxillofac Surg 52(9):831–837. https://doi.org/10.1016/j.bjoms.2014.07.101
Hoekema A, Doff MH, de Bont LG, van der Hoeven JH, Wijkstra PJ, Pasma HR, Stegenga B (2007) Predictors of obstructive sleep apnea-hypopnea treatment outcome. J Dent Res 86(12):1181–1186
Mostafiz W, Dalci O, Sutherland K, Malhotra A, Srinivasan V, Darendeliler MA, Cistulli PA (2011) Influence of oral and craniofacial dimensions on mandibular advancement splint treatment outcome in patients with obstructive sleep apnea. Chest 139(6):1331–1339. https://doi.org/10.1378/chest.10-2224
Cunha TCA, Guimarães TM, Schultz TCB, Almeida FR, Cunha TM, Simamoto PC Jr, Bittencourt LRA (2017) Predictors of success for mandibular repositioning appliance in obstructive sleep apnea syndrome. Braz Oral Res 5:31–37. https://doi.org/10.1590/1807-3107BOR-2017.vol31.0037
Nuckton TJ, Glidden DV, Browner WS, Claman DM (2006) Physical examination: Mallampati score as an independent predictor of obstructive sleep apnea. Sleep 29(7):903–908
Lettieri CJ, Paolino N, Eliasson AH, Shah AA, Holley AB (2011) Comparison of adjustable and fixed oral appliances for the treatment of obstructive sleep apnea. J Clin Sleep Med 7(5):439–445. https://doi.org/10.5664/JCSM.1300
Jeans WD, Fernando DC, Maw AR, Leighton BC (1981) A longitudinal study of the growth of the nasopharynx and its contents in normal children. Br J Radiol 54(638):117–121
Kollias I, Krogstad O (1999) Adult craniocervical and pharyngeal changes--a longitudinal cephalometric study between 22 and 42 years of age. Part II: Morphological uvulo-glossopharyngeal changes. Eur J Orthod 21(4):345–355
Fitzpatrick MF, McLean H, Urton AM, Tan A, O’Donnell D, Driver HS (2003) Effect of nasal or oral breathing route on upper airway resistance during sleep. Eur Respir J 22(5):827–832
Lofaso F, Coste A, d’Ortho MP, Zerah-Lancner F, Delclaux C, Goldenberg F, Harf A (2000) Nasal obstruction as a risk factor for sleep apnoea syndrome. Eur Respir J 16(4):639–643
Li HY, Wang PC, Hsu CY, Cheng ML, Liou CC, Chen NH (2005) Nasal resistance in patients with obstructive sleep apnea. ORL J Otorhinolaryngol Relat Spec 67(2):70–74
Miljeteig H, Hoffstein V, Cole P (1992) The effect of unilateral and bilateral nasal obstruction on snoring and sleep apnea. Laryngoscope 102(10):1150–1152
Blomster H, Kemppainen T, Numminen J, Ruoppi P, Sahlman J, Peltonen M, Seppä J, Tuomilehto H (2011) Impaired nasal breathing may prevent the beneficial effect of weight loss in the treatment of OSA. Rhinology 49(5):587–592. https://doi.org/10.4193/Rhino11.131
Nakata S, Miyazaki S, Ohki M, Morinaga M, Noda A, Sugiura T, Sugiura M, Teranishi M, Katayama N, Nakashima T (2007) Reduced nasal resistance after simple tonsillectomy in patients with obstructive sleep apnea. Am J Rhinol 21(2):192–195
Marklund M (2017) Update on oral appliance therapy for OSA. Curr Sleep Med Rep 3(3):143–151. https://doi.org/10.1007/s40675-017-0080-5
Sutherland K, Takaya H, Qian J, Petocz P, Ng AT, Cistulli PA (2015) Oral appliance treatment response and polysomnographic phenotypes of obstructive sleep apnea. J Clin Sleep Med 11(8):861–868. https://doi.org/10.5664/jcsm.4934
Iber C, Ancoli-Israel S, Chesson AL Jr (2007) Quan SF for the American Academy of Sleep Medicine. In: The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications, 1st edn. American Academy of Sleep Medicine, Westchester, IL
Acknowledgments
SomnoMed Ltd. placed the components of the DentiTrack System and software at our disposal during the survey.
Funding
Open access funding provided by University of Eastern Finland (UEF) including Kuopio University Hospital. The first author received a grant for this study from the Finnish Dental Society Apollonia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study received ethics committee approval on February 7, 2017. All procedures performed in this study were in accordance with the international and national ethical standards and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all the participants included in the study.
Disclaimer
The funding sources had no role in the study design or in the collection, analysis or interpretation of the data.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Pahkala, R., Seppä, J., Myllykangas, R. et al. The impact of oral appliance therapy with moderate mandibular advancement on obstructive sleep apnea and upper airway volume. Sleep Breath 24, 865–873 (2020). https://doi.org/10.1007/s11325-019-01914-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-019-01914-3