Abstract
Purpose
Sleep apnea is associated with increased risk of cardiovascular disease. Elevated plasma galectin-3 levels, a biomarker associated with myocardial fibrosis, are also associated with adverse cardiovascular events, including heart failure. Our objective was to determine the relationship between severity of sleep apnea and plasma levels of galectin-3 and to determine whether this relationship was modified by sex.
Methods
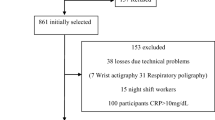
We performed a cross-sectional study of 471 Mexican Americans from Starr County, TX who underwent an overnight, in-home sleep evaluation, and plasma measurement of galectin-3. Severity of sleep apnea was based on apnea hypopnea index (AHI). Multivariable linear regression modeling was used to determine the association between categories of sleep apnea and galectin-3. We also tested for interactions by sex.
Results
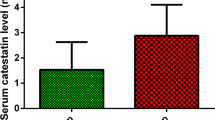
The mean age was 53 years, and 74% of the cohort was female. The prevalence of moderate to severe sleep apnea (AHI > 15 apnea–hypopnea events per hour) was 36.7%. Moderate to severe sleep apnea was associated with increased levels of galectin-3 in the entire population, but we identified a statistically significant interaction between galectin-3 levels and category of sleep apnea by sex (p for interaction = 0.02). Plasma galectin levels were significantly higher in women with moderate or severe sleep apnea than women with no/mild sleep apnea (multivariable adjusted p < 0.001), but not in men (p = 0.5).
Conclusions
Sleep apnea is associated elevated galectin-3 levels in women but not men. Our findings highlight a possible sex-specific relationship between sleep apnea and galectin-3, a biomarker of potential myocardial fibrosis that has been associated with increased cardiovascular risk.

Similar content being viewed by others
Abbreviations
- AHI:
-
Apnea hypopnea index
- CPAP:
-
Continuous positive airway pressure
- ODI:
-
Oxygen desaturation index
- T90:
-
Time spent (minutes) below nocturnal oxygen saturation of 90%
- hsTnT:
-
High-sensitivity troponin-T
- NT-proBNP:
-
N-terminal pro-B-type natriuretic peptide
References
Suthahar N, Meijers WC, Sillje HHW, Ho JE, Liu FT, de Boer RA (2018) Galectin-3 activation and inhibition in heart failure and cardiovascular disease: an update. Theranostics 8(3):593–609. https://doi.org/10.7150/thno.22196
Filipe MD, Meijers WC, Rogier van der Velde A, de Boer RA (2015) Galectin-3 and heart failure: prognosis, prediction & clinical utility. Clin Chim Acta 443:48–56. https://doi.org/10.1016/j.cca.2014.10.009
Sharma UC, Pokharel S, van Brakel TJ, van Berlo JH, Cleutjens JP, Schroen B, Andre S, Crijns HJ, Gabius HJ, Maessen J, Pinto YM (2004) Galectin-3 marks activated macrophages in failure-prone hyspertrophied hearts and contributes to cardiac dysfunction. Circulation 110(19):3121–3128. https://doi.org/10.1161/01.CIR.0000147181.65298.4D
Daniels LB, Clopton P, Laughlin GA, Maisel AS, Barrett-Connor E (2014) Galectin-3 is independently associated with cardiovascular mortality in community-dwelling older adults without known cardiovascular disease: the Rancho Bernardo Study. Am Heart J 167(5):674–682 e671. https://doi.org/10.1016/j.ahj.2013.12.031
Ho JE, Liu C, Lyass A, Courchesne P, Pencina MJ, Vasan RS, Larson MG, Levy D (2012) Galectin-3, a marker of cardiac fibrosis, predicts incident heart failure in the community. J Am Coll Cardiol 60(14):1249–1256. https://doi.org/10.1016/j.jacc.2012.04.053
de Boer RA, van Veldhuisen DJ, Gansevoort RT, Muller Kobold AC, van Gilst WH, Hillege HL, Bakker SJ, van der Harst P (2012) The fibrosis marker galectin-3 and outcome in the general population. J Intern Med 272(1):55–64. https://doi.org/10.1111/j.1365-2796.2011.02476.x
Djousse L, Matsumoto C, Petrone A, Weir NL, Tsai MY, Gaziano JM (2014) Plasma galectin 3 and heart failure risk in the Physicians’ Health Study. Eur J Heart Fail 16(3):350–354. https://doi.org/10.1002/ejhf.21
Jagodzinski A, Havulinna AS, Appelbaum S, Zeller T, Jousilahti P, Skytte-Johanssen S, Hughes MF, Blankenberg S, Salomaa V (2015) Predictive value of galectin-3 for incident cardiovascular disease and heart failure in the population-based FINRISK 1997 cohort. Int J Cardiol 192:33–39. https://doi.org/10.1016/j.ijcard.2015.05.040
Shahar E, Whitney CW, Redline S, Lee ET, Newman AB, Nieto FJ, O’Connor GT, Boland LL, Schwartz JE, Samet JM (2001) Sleep-disordered breathing and cardiovascular disease: cross-sectional results of the Sleep Heart Health Study. Am J Respir Crit Care Med 163(1):19–25. https://doi.org/10.1164/ajrccm.163.1.2001008
Cadby G, McArdle N, Briffa T, Hillman DR, Simpson L, Knuiman M, Hung J (2015) Severity of OSA is an independent predictor of incident atrial fibrillation hospitalization in a large sleep-clinic cohort. Chest 148(4):945–952. https://doi.org/10.1378/chest.15-0229
Gottlieb DJ, Yenokyan G, Newman AB, O’Connor GT, Punjabi NM, Quan SF, Redline S, Resnick HE, Tong EK, Diener-West M, Shahar E (2010) Prospective study of obstructive sleep apnea and incident coronary heart disease and heart failure: the sleep heart health study. Circulation 122(4):352–360. https://doi.org/10.1161/CIRCULATIONAHA.109.901801
Cowie MR (2017) Sleep apnea: state of the art. Trends Cardiovasc Med 27(4):280–289. https://doi.org/10.1016/j.tcm.2016.12.005
Querejeta Roca G, Redline S, Punjabi N, Claggett B, Ballantyne CM, Solomon SD, Shah AM (2013) Sleep apnea is associated with subclinical myocardial injury in the community. The ARIC-SHHS study. Am J Respir Crit Care Med 188(12):1460–1465. https://doi.org/10.1164/rccm.201309-1572OC
Roca GQ, Redline S, Claggett B, Bello N, Ballantyne CM, Solomon SD, Shah AM (2015) Sex-specific association of sleep apnea severity with subclinical myocardial injury, ventricular hypertrophy, and heart failure risk in a community-dwelling cohort: the atherosclerosis risk in communities-sleep heart health study. Circulation 132(14):1329–1337. https://doi.org/10.1161/CIRCULATIONAHA.115.016985
Randby A, Namtvedt SK, Einvik G, Hrubos-Strom H, Hagve TA, Somers VK, Omland T (2012) Obstructive sleep apnea is associated with increased high-sensitivity cardiac troponin T levels. Chest 142(3):639–646. https://doi.org/10.1378/chest.11-1779
Ljunggren M, Lindahl B, Theorell-Haglow J, Lindberg E (2012) Association between obstructive sleep apnea and elevated levels of type B natriuretic peptide in a community-based sample of women. Sleep 35(11):1521–1527. https://doi.org/10.5665/sleep.2202
Redline S, Sotres-Alvarez D, Loredo J, Hall M, Patel SR, Ramos A, Shah N, Ries A, Arens R, Barnhart J, Youngblood M, Zee P, Daviglus ML (2014) Sleep-disordered breathing in Hispanic/Latino individuals of diverse backgrounds. The Hispanic Community Health Study/Study of Latinos. Am J Respir Crit Care Med 189(3):335–344. https://doi.org/10.1164/rccm.201309-1735OC
Young T, Finn L (1998) Epidemiological insights into the public health burden of sleep disordered breathing: sex differences in survival among sleep clinic patients. Thorax 53(Suppl 3):S16–S19
Bureau USC Quick Facts: Starr County Texas. http://www.census.gov/quickfacts/table/PST045215/48427. Accessed September 4 2016
Hanis CL, Redline S, Cade BE, Bell GI, Cox NJ, Below JE, Brown EL, Aguilar D (2016) Beyond type 2 diabetes, obesity and hypertension: an axis including sleep apnea, left ventricular hypertrophy, endothelial dysfunction, and aortic stiffness among Mexican Americans in Starr County, Texas. Cardiovasc Diabetol 15:86. https://doi.org/10.1186/s12933-016-0405-6
Yalamanchali S, Farajian V, Hamilton C, Pott TR, Samuelson CG, Friedman M (2013) Diagnosis of obstructive sleep apnea by peripheral arterial tonometry: meta-analysis. JAMA Otolaryngol Head Neck Surg 139(12):1343–1350. https://doi.org/10.1001/jamaoto.2013.5338
Nambi V, Liu X, Chambless LE, de Lemos JA, Virani SS, Agarwal S, Boerwinkle E, Hoogeveen RC, Aguilar D, Astor BC, Srinivas PR, Deswal A, Mosley TH, Coresh J, Folsom AR, Heiss G, Ballantyne CM (2013) Troponin T and N-terminal pro-B-type natriuretic peptide: a biomarker approach to predict heart failure risk—the atherosclerosis risk in communities study. Clin Chem 59(12):1802–1810. https://doi.org/10.1373/clinchem.2013.203638
deFilippi CR, de Lemos JA, Christenson RH, Gottdiener JS, Kop WJ, Zhan M, Seliger SL (2010) Association of serial measures of cardiac troponin T using a sensitive assay with incident heart failure and cardiovascular mortality in older adults. JAMA 304(22):2494–2502. https://doi.org/10.1001/jama.2010.1708
Hill SA, Balion CM, Santaguida P, McQueen MJ, Ismaila AS, Reichert SM, McKelvie R, Worster A, Raina PS (2008) Evidence for the use of B-type natriuretic peptides for screening asymptomatic populations and for diagnosis in primary care. Clin Biochem 41(4–5):240–249. https://doi.org/10.1016/j.clinbiochem.2007.08.016
Saunders JT, Nambi V, de Lemos JA, Chambless LE, Virani SS, Boerwinkle E, Hoogeveen RC, Liu X, Astor BC, Mosley TH, Folsom AR, Heiss G, Coresh J, Ballantyne CM (2011) Cardiac troponin T measured by a highly sensitive assay predicts coronary heart disease, heart failure, and mortality in the Atherosclerosis Risk in Communities Study. Circulation 123(13):1367–1376. https://doi.org/10.1161/CIRCULATIONAHA.110.005264
Wang TJ, Larson MG, Levy D, Benjamin EJ, Leip EP, Omland T, Wolf PA, Vasan RS (2004) Plasma natriuretic peptide levels and the risk of cardiovascular events and death. N Engl J Med 350(7):655–663. https://doi.org/10.1056/NEJMoa031994
Calvier L, Miana M, Reboul P, Cachofeiro V, Martinez-Martinez E, de Boer RA, Poirier F, Lacolley P, Zannad F, Rossignol P, Lopez-Andres N (2013) Galectin-3 mediates aldosterone-induced vascular fibrosis. Arterioscler Thromb Vasc Biol 33(1):67–75. https://doi.org/10.1161/ATVBAHA.112.300569
Vergaro G, Prud’homme M, Fazal L, Merval R, Passino C, Emdin M, Samuel JL, Cohen Solal A, Delcayre C (2016) Inhibition of galectin-3 pathway prevents isoproterenol-induced left ventricular dysfunction and fibrosis in mice. Hypertension 67(3):606–612. https://doi.org/10.1161/HYPERTENSIONAHA.115.06161
Yu L, Ruifrok WP, Meissner M, Bos EM, van Goor H, Sanjabi B, van der Harst P, Pitt B, Goldstein IJ, Koerts JA, van Veldhuisen DJ, Bank RA, van Gilst WH, Sillje HH, de Boer RA (2013) Genetic and pharmacological inhibition of galectin-3 prevents cardiac remodeling by interfering with myocardial fibrogenesis. Circ Heart Fail 6(1):107–117. https://doi.org/10.1161/CIRCHEARTFAILURE.112.971168
Peppard PE, Young T, Palta M, Skatrud J (2000) Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 342(19):1378–1384. https://doi.org/10.1056/NEJM200005113421901
Otto ME, Belohlavek M, Romero-Corral A, Gami AS, Gilman G, Svatikova A, Amin RS, Lopez-Jimenez F, Khandheria BK, Somers VK (2007) Comparison of cardiac structural and functional changes in obese otherwise healthy adults with versus without obstructive sleep apnea. Am J Cardiol 99(9):1298–1302. https://doi.org/10.1016/j.amjcard.2006.12.052
Chami HA, Devereux RB, Gottdiener JS, Mehra R, Roman MJ, Benjamin EJ, Gottlieb DJ (2008) Left ventricular morphology and systolic function in sleep-disordered breathing: the Sleep Heart Health Study. Circulation 117(20):2599–2607. https://doi.org/10.1161/CIRCULATIONAHA.107.717892
Javaheri S, Barbe F, Campos-Rodriguez F, Dempsey JA, Khayat R, Javaheri S, Malhotra A, Martinez-Garcia MA, Mehra R, Pack AI, Polotsky VY, Redline S, Somers VK (2017) Sleep apnea: types, mechanisms, and clinical cardiovascular consequences. J Am Coll Cardiol 69(7):841–858. https://doi.org/10.1016/j.jacc.2016.11.069
Drager LF, McEvoy RD, Barbe F, Lorenzi-Filho G, Redline S, Initiative I (2017) Sleep apnea and cardiovascular disease: lessons from recent trials and need for team science. Circulation 136(19):1840–1850. https://doi.org/10.1161/CIRCULATIONAHA.117.029400
Chen TI, Tu WC (2016) Exercise attenuates intermittent hypoxia-induced cardiac fibrosis associated with sodium-hydrogen exchanger-1 in rats. Front Physiol 7:462. https://doi.org/10.3389/fphys.2016.00462
Ding WX, Dong YB, Ding N, Zhang XF, Zhang SJ, Zhang XL, Liu JN, Lu G (2014) Adiponectin protects rat heart from left ventricular remodeling induced by chronic intermittent hypoxia via inhibition of TGF-beta/smad2/3 pathway. J Thorac Dis 6(9):1278–1284. https://doi.org/10.3978/j.issn.2072-1439.2014.07.44
Punjabi NM, Caffo BS, Goodwin JL, Gottlieb DJ, Newman AB, O’Connor GT, Rapoport DM, Redline S, Resnick HE, Robbins JA, Shahar E, Unruh ML, Samet JM (2009) Sleep-disordered breathing and mortality: a prospective cohort study. PLoS Med 6(8):e1000132. https://doi.org/10.1371/journal.pmed.1000132
Faulx MD, Larkin EK, Hoit BD, Aylor JE, Wright AT, Redline S (2004) Sex influences endothelial function in sleep-disordered breathing. Sleep 27(6):1113–1120
Chew M, Xie J, Klein R, Klein B, Cotch MF, Redline S, Wong TY, Cheung N (2016) Sleep apnea and retinal signs in cardiovascular disease: the multi-ethnic study of atherosclerosis. Sleep Breath 20(1):15–23. https://doi.org/10.1007/s11325-015-1177-z
Macey PM, Kumar R, Woo MA, Yan-Go FL, Harper RM (2013) Heart rate responses to autonomic challenges in obstructive sleep apnea. PLoS One 8(10):e76631. https://doi.org/10.1371/journal.pone.0076631
McEvoy RD, Antic NA, Heeley E, Luo Y, Ou Q, Zhang X, Mediano O, Chen R, Drager LF, Liu Z, Chen G, Du B, McArdle N, Mukherjee S, Tripathi M, Billot L, Li Q, Lorenzi-Filho G, Barbe F, Redline S, Wang J, Arima H, Neal B, White DP, Grunstein RR, Zhong N, Anderson CS, Investigators S, Coordinators (2016) CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med 375(10):919–931. https://doi.org/10.1056/NEJMoa1606599
Foster GD, Sanders MH, Millman R, Zammit G, Borradaile KE, Newman AB, Wadden TA, Kelley D, Wing RR, Sunyer FX, Darcey V, Kuna ST, Sleep ARG (2009) Obstructive sleep apnea among obese patients with type 2 diabetes. Diabetes Care 32(6):1017–1019. https://doi.org/10.2337/dc08-1776
Acknowledgements
We thank the field staff in Starr County for their careful collection of these data and are especially grateful to the residents of Starr County who graciously participated in the study.
Funding
This work was supported in part by grants HL092585 and HL102830 from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Singh, M., Hanis, C.L., Redline, S. et al. Sleep apnea and galectin-3: possible sex-specific relationship. Sleep Breath 23, 1107–1114 (2019). https://doi.org/10.1007/s11325-019-01788-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-019-01788-5