Abstract
Background
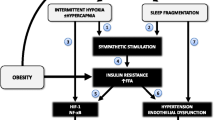
Obstructive sleep apnea syndrome (OSAS) has a negative impact on health and behavior of millions of individuals worldwide. The pathogenesis of this disorder is a multifactorial process related to a variety of mechanisms, including selective activation of inflammatory response pathways. A number of inflammatory factors, such as IL-6, IL-8, and TNF-α, can be found in high concentrations in subjects with OSAS and may serve as biological markers of this disease. The concentration of these cytokines contributes to weight gain in patients with OSAS and can also modify the risk of obesity-related metabolic disorders, especially insulin resistance. Nevertheless, the mechanisms by which specific genes are associated with these processes are still poorly known. In addition to gene expression studies, investigations aiming at the identification of epigenetic factors associated with OSAS are still scarce in the literature. The documented data support the hypothesis that the molecular changes that mediate inflammatory response are important mechanisms in the pathogenesis of OSAS, sleepiness, insulin resistance, visceral obesity, and cardiovascular disease, perhaps by leading to a more severe OSAS. Often, systemic changes may not be detected in mild OSA; however, molecular changes, which are much more sensitive to the mechanisms of intermittent hypoxia and oxidative stress, may be present.
Purpose
This review aimed to show an updated view on the studies evaluating the genetic basis of inflammatory response in many aspects of OSAS and to highlight potential research areas not fully explored to date in this field.
Similar content being viewed by others
References
Azagra-Calero E, Espinar-Escalona E, Barrera-Mora J-M, et al. (2012) Obstructive sleep apnea syndrome (OSAS). Review of the literature. Med Oral Patol Oral Cir Bucal 17:e925–e929
Casale M, Pappacena M, Rinaldi V, et al. (2009) Obstructive sleep apnea syndrome: from phenotype to genetic basis. Curr Genomics 10:119–126. doi:10.2174/138920209787846998
Grilo A, Ruiz-Granados ES, Moreno-Rey C, et al. (2013) Genetic analysis of candidate SNPs for metabolic syndrome in obstructive sleep apnea (OSA). Gene 521:150–154. doi:10.1016/j.gene.2013.03.024
Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research (1999). The Report of an American Academy of Sleep Medicine Task Force. Sleep 22:667–689.
Garvey JF, Taylor CT, McNicholas WT (2009) Cardiovascular disease in obstructive sleep apnoea syndrome: the role of intermittent hypoxia and inflammation. Eur Respir J 33:1195–1205. doi:10.1183/09031936.00111208
De Carvalho TBO, Suman M, Molina FD, et al. (2013) Relationship of obstructive sleep apnea syndrome with the 5-HT2A receptor gene in Brazilian patients. Sleep Breath Schlaf Atm 17:57–62. doi:10.1007/s11325-012-0645-y
Young T, Palta M, Dempsey J, et al. (1993) The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med 328:1230–1235. doi:10.1056/NEJM199304293281704
Peppard PE, Young T, Barnet JH, et al. (2013) Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol 177:1006–1014. doi:10.1093/aje/kws342
Tufik S, Santos-Silva R, Taddei JA, Bittencourt LRA (2010) Obstructive sleep apnea syndrome in the Sao Paulo Epidemiologic Sleep Study. Sleep Med 11:441–446. doi:10.1016/j.sleep.2009.10.005
Young T, Peppard PE, Gottlieb DJ (2002) Epidemiology of obstructive sleep apnea: a population health perspective. Am J Respir Crit Care Med 165:1217–1239
Jordan AS, McSharry DG, Malhotra A (2014) Adult obstructive sleep apnoea. Lancet 383:736–747. doi:10.1016/S0140-6736(13)60734-5
Schwartz AR, Patil SP, Laffan AM, et al. (2008) Obesity and obstructive sleep apnea: pathogenic mechanisms and therapeutic approaches. Proc Am Thorac Soc 5:185–192. doi:10.1513/pats.200708-137MG
Peppard PE, Young T, Palta M, et al. (2000) Longitudinal study of moderate weight change and sleep-disordered breathing. JAMA 284:3015–3021
Ronen O, Malhotra A, Pillar G (2007) Influence of gender and age on upper-airway length during development. Pediatrics 120:e1028–e1034. doi:10.1542/peds.2006-3433
Whittle AT, Marshall I, Mortimore IL, et al. (1999) Neck soft tissue and fat distribution: comparison between normal men and women by magnetic resonance imaging. Thorax 54:323–328
Malhotra A, Huang Y, Fogel RB, et al. (2002) The male predisposition to pharyngeal collapse: importance of airway length. Am J Respir Crit Care Med 166:1388–1395. doi:10.1164/rccm.2112072
Young T (2001) Menopause, hormone replacement therapy, and sleep-disordered breathing: are we ready for the heat? Am J Respir Crit Care Med 163:597–598. doi:10.1164/ajrccm.163.3.ed09-01a
McNicholas WT (2009) Obstructive sleep apnea and inflammation. Prog Cardiovasc Dis 51:392–399. doi:10.1016/j.pcad.2008.10.005
Testelmans D, Tamisier R, Barone-Rochette G, et al. (2013) Profile of circulating cytokines: impact of OSA, obesity and acute cardiovascular events. Cytokine 62:210–216. doi:10.1016/j.cyto.2013.02.021
Ryan S, Taylor CT, McNicholas WT (2009) Systemic inflammation: a key factor in the pathogenesis of cardiovascular complications in obstructive sleep apnoea syndrome? Postgrad Med J 85:693–698. doi:10.1136/thx.2008.105577
Lam S-Y, Liu Y, Ng K-M, et al. (2012) Chronic intermittent hypoxia induces local inflammation of the rat carotid body via functional upregulation of proinflammatory cytokine pathways. Histochem Cell Biol 137:303–317. doi:10.1007/s00418-011-0900-5
Grieve DJ, Shah AM (2003) Oxidative stress in heart failure. More than just damage. Eur Heart J 24:2161–2163
Lavie L, Lavie P (2009) Molecular mechanisms of cardiovascular disease in OSAHS: the oxidative stress link. Eur Respir J 33:1467–1484. doi:10.1183/09031936.00086608
Pilkauskaite G, Miliauskas S, Sakalauskas R (2013) Reactive oxygen species production in peripheral blood neutrophils of obstructive sleep apnea patients. ScientificWorldJournal 2013:421763. doi:10.1155/2013/421763
Lavie L, Lavie P (2012) CrossTalk opposing view: most cardiovascular diseases in sleep apnoea are not caused by sympathetic activation. J Physiol 590:2817–2819. doi:10.1113/jphysiol.2012.233833
Lui MM-S, Lam DC-L, Ip MS-M (2013) Significance of endothelial dysfunction in sleep-related breathing disorder. Respirol Carlton Vic 18:39–46. doi:10.1111/j.1440-1843.2012.02212.x
Flora Filho R, Zilberstein B (2000) Óxido nítrico: o simples mensageiro percorrendo a complexidade. Metabolismo, síntese e funções. Rev Assoc Médica Bras 46:265–271. doi:10.1590/S0104-42302000000300012
Lavie L (2015) Oxidative stress in obstructive sleep apnea and intermittent hypoxia—revisited—the bad ugly and good: implications to the heart and brain. Sleep Med Rev 20:27–45. doi:10.1016/j.smrv.2014.07.003
Hernández C, Abreu J, Abreu P, et al. (2006) Effects of nasal positive airway pressure treatment on oxidative stress in patients with sleep apnea-hypopnea syndrome. Arch Bronconeumol 42:125–129
Casella Filho A, Araújo RG, Galvão TG, Chagas ACP (2005) Inflamação e Aterosclerose: Integração de Novas Teorias e Valorização dos Novos Marcadores. Rev Bras Cardiol Invasiva 11:14–19
Kang WS, Park HJ, Chung J-H, Kim JW (2013) REM sleep deprivation increases the expression of interleukin genes in mice hypothalamus. Neurosci Lett 556:73–78. doi:10.1016/j.neulet.2013.09.050
Krueger JM, Majde JA (2003) Humoral links between sleep and the immune system: research issues. Ann N Y Acad Sci 992:9–20
Yokoe T, Minoguchi K, Matsuo H, et al. (2003) Elevated levels of C-reactive protein and interleukin-6 in patients with obstructive sleep apnea syndrome are decreased by nasal continuous positive airway pressure. Circulation 107:1129–1134
Ohga E, Tomita T, Wada H, et al. (1985) (2003) Effects of obstructive sleep apnea on circulating ICAM-1, IL-8, and MCP-1. J Appl Physiol Bethesda Md 94:179–184. doi:10.1152/japplphysiol.00177.2002
Ryan S, Taylor CT, McNicholas WT (2006) Predictors of elevated nuclear factor-kappaB-dependent genes in obstructive sleep apnea syndrome. Am J Respir Crit Care Med 174:824–830. doi:10.1164/rccm.200601-066OC
Schulz R, Hummel C, Heinemann S, et al. (2002) Serum levels of vascular endothelial growth factor are elevated in patients with obstructive sleep apnea and severe nighttime hypoxia. Am J Respir Crit Care Med 165:67–70. doi:10.1164/ajrccm.165.1.2101062
Ip MS, Lam KS, Ho C, et al. (2000) Serum leptin and vascular risk factors in obstructive sleep apnea. Chest 118:580–586
Al Lawati N, Mulgrew A, Cheema R, et al. (2009) Pro-atherogenic cytokine profile of patients with suspected obstructive sleep apnea. Sleep Breath Schlaf Atm 13:391–395. doi:10.1007/s11325-009-0259-1
Shamsuzzaman ASM, Winnicki M, Lanfranchi P, et al. (2002) Elevated C-reactive protein in patients with obstructive sleep apnea. Circulation 105:2462–2464
Guven SF, Turkkani MH, Ciftci B, et al. (2012) The relationship between high-sensitivity C-reactive protein levels and the severity of obstructive sleep apnea. Sleep Breath Schlaf Atm 16:217–221. doi:10.1007/s11325-011-0492-2
Quercioli A, Mach F, Montecucco F (2010) Inflammation accelerates atherosclerotic processes in obstructive sleep apnea syndrome (OSAS). Sleep Breath Schlaf Atm 14:261–269. doi:10.1007/s11325-010-0338-3
Popko K, Gorska E, Potapinska O, et al. (2008) Frequency of distribution of inflammatory cytokines IL-1, IL-6 and TNF-alpha gene polymorphism in patients with obstructive sleep apnea. J Physiol Pharmacol Off J Pol Physiol Soc 59(Suppl 6):607–614
Semenza GL (1985) (2004) O2-regulated gene expression: transcriptional control of cardiorespiratory physiology by HIF-1. J Appl Physiol Bethesda Md 96:1173–1177 discussion 1170–1172. doi:10.1152/japplphysiol.00770.2003
Ryan S, Taylor CT, McNicholas WT (2005) Selective activation of inflammatory pathways by intermittent hypoxia in obstructive sleep apnea syndrome. Circulation 112:2660–2667. doi:10.1161/CIRCULATIONAHA.105.556746
Baessler A, Nadeem R, Harvey M, et al. (2013) Treatment for sleep apnea by continuous positive airway pressure improves levels of inflammatory markers—a meta-analysis. J Inflamm Lond Engl 10:13. doi:10.1186/1476-9255-10-13
Nadeem R, Molnar J, Madbouly EM, et al. (2013) Serum inflammatory markers in obstructive sleep apnea: a meta-analysis. J Clin Sleep Med JCSM Off Publ Am Acad Sleep Med 9:1003–1012. doi:10.5664/jcsm.3070
Lui MM, Lam JC, Mak HK-F, et al. (2009) C-reactive protein is associated with obstructive sleep apnea independent of visceral obesity. Chest 135:950–956. doi:10.1378/chest.08-1798
Punjabi NM, Beamer BA (2007) C-reactive protein is associated with sleep disordered breathing independent of adiposity. Sleep 30:29–34
Guilleminault C, Kirisoglu C, Ohayon MM (2004) C-reactive protein and sleep-disordered breathing. Sleep 27:1507–1511
Mills PJ, Natarajan L, von Känel R, et al. (2009) Diurnal variability of C-reactive protein in obstructive sleep apnea. Sleep Breath Schlaf Atm 13:415–420. doi:10.1007/s11325-009-0268-0
Ishida K, Kato M, Kato Y, et al. (2009) Appropriate use of nasal continuous positive airway pressure decreases elevated C-reactive protein in patients with obstructive sleep apnea. Chest 136:125–129. doi:10.1378/chest.08-1431
Xie X, Pan L, Ren D, et al. (2013) Effects of continuous positive airway pressure therapy on systemic inflammation in obstructive sleep apnea: a meta-analysis. Sleep Med 14:1139–1150. doi:10.1016/j.sleep.2013.07.006
Guo Y, Pan L, Ren D, Xie X (2013) Impact of continuous positive airway pressure on C-reactive protein in patients with obstructive sleep apnea: a meta-analysis. Sleep Breath Schlaf Atm 17:495–503. doi:10.1007/s11325-012-0722-2
Mermigkis C, Bouloukaki I, Mermigkis D, et al. (2012) CRP evolution pattern in CPAP-treated obstructive sleep apnea patients. Does gender play a role? Sleep Breath Schlaf Atm 16:813–819. doi:10.1007/s11325-011-0580-3
Von Känel R, Natarajan L, Ancoli-Israel S, et al. (2013) Effect of continuous positive airway pressure on day/night rhythm of prothrombotic markers in obstructive sleep apnea. Sleep Med 14:58–65. doi:10.1016/j.sleep.2012.07.009
Larkin EK, Patel SR, Goodloe RJ, et al. (2010) A candidate gene study of obstructive sleep apnea in European Americans and African Americans. Am J Respir Crit Care Med 182:947–953. doi:10.1164/rccm.201002-0192OC
Petruco ACM, M da C B (2010) Aspectos genéticos da SAOS. J Bras Pneumol 36:13–16. doi:10.1590/S1806-37132010001400005
Zhang X, Liu R-Y, Lei Z, et al. (2009) Genetic variants in interleukin-6 modified risk of obstructive sleep apnea syndrome. Int J Mol Med 23:485–493
Jones J, Chen L-S, Baudhuin L, et al. (2009) Relationships between C-reactive protein concentration and genotype in healthy volunteers. Clin Chem Lab Med CCLM FESCC 47:20–25. doi:10.1515/CCLM.2009.005
Kaditis AG, Gozal D, Khalyfa A, et al. (2014) Variants in C-reactive protein and IL-6 genes and susceptibility to obstructive sleep apnea in children: a candidate-gene association study in European American and Southeast European populations. Sleep Med 15:228–235. doi:10.1016/j.sleep.2013.08.795
Riha RL, Brander P, Vennelle M, et al. (2005) Tumour necrosis factor-alpha (-308) gene polymorphism in obstructive sleep apnoea-hypopnoea syndrome. Eur Respir J 26:673–678. doi:10.1183/09031936.05.00130804
Bhushan B, Guleria R, Misra A, et al. (2009) TNF-alpha gene polymorphism and TNF-alpha levels in obese Asian Indians with obstructive sleep apnea. Respir Med 103:386–392. doi:10.1016/j.rmed.2008.10.001
Wu Y, Cao C, Wu Y, et al. (2014) TNF-α-308G/A polymorphism contributes to obstructive sleep apnea syndrome risk: evidence based on 10 case-control studies. PLoS One 9:e106183. doi:10.1371/journal.pone.0106183
Larkin EK, Patel SR, Zhu X, et al. (2010) Study of the relationship between the interleukin-6 gene and obstructive sleep apnea. Clin Transl Sci 3:337–339
Gozal D, Khalyfa A, Capdevila OS, et al. (2012) Cognitive function in prepubertal children with obstructive sleep apnea: a modifying role for NADPH oxidase p22 subunit gene polymorphisms? Antioxid Redox Signal 16:171–177. doi:10.1089/ars.2011.4189
Piérola J, Alemany A, Yañez A, et al. (2011) NADPH oxidase p22phox polymorphisms and oxidative stress in patients with obstructive sleep apnoea. Respir Med 105:1748–1754. doi:10.1016/j.rmed.2011.08.006
Arnardottir ES, Mackiewicz M, Gislason T, et al. (2009) Molecular signatures of obstructive sleep apnea in adults: a review and perspective. Sleep 32:447–470
Greenberg H, Ye X, Wilson D, et al. (2006) Chronic intermittent hypoxia activates nuclear factor-kappaB in cardiovascular tissues in vivo. Biochem Biophys Res Commun 343:591–596. doi:10.1016/j.bbrc.2006.03.015
Lavie L (2005) Sleep-disordered breathing and cerebrovascular disease: a mechanistic approach. Neurol Clin 23:1059–1075. doi:10.1016/j.ncl.2005.05.005
Lavie L (2003) Obstructive sleep apnoea syndrome—an oxidative stress disorder. Sleep Med Rev 7:35–51
Meier-Ewert HK, Ridker PM, Rifai N, et al. (2004) Effect of sleep loss on C-reactive protein, an inflammatory marker of cardiovascular risk. J Am Coll Cardiol 43:678–683. doi:10.1016/j.jacc.2003.07.050
Vgontzas AN, Zoumakis E, Bixler EO, et al. (2004) Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. J Clin Endocrinol Metab 89:2119–2126. doi:10.1210/jc.2003-031562
Irwin MR, Wang M, Campomayor CO, et al. (2006) Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation. Arch Intern Med 166:1756–1762. doi:10.1001/archinte.166.16.1756
Clement K, Langin D (2007) Regulation of inflammation-related genes in human adipose tissue. J Intern Med 262:422–430. doi:10.1111/j.1365-2796.2007.01851.x
Taylor CT, Kent BD, Crinion SJ, et al. (2014) Human adipocytes are highly sensitive to intermittent hypoxia induced NF-kappaB activity and subsequent inflammatory gene expression. Biochem Biophys Res Commun 447:660–665. doi:10.1016/j.bbrc.2014.04.062
Kimoff RJ, Hamid Q, Divangahi M, et al. (2011) Increased upper airway cytokines and oxidative stress in severe obstructive sleep apnoea. Eur Respir J 38:89–97. doi:10.1183/09031936.00048610
Broytman O, Braun RK, Morgan BJ, et al. (2014) Effects of chronic intermittent hypoxia on allergen-induced airway inflammation in rats. Am J Respir Cell Mol Biol. doi:10.1165/rcmb.2014-0213OC
Perry JC, Guindalini C, Bittencourt L, et al. (2013) Whole blood hypoxia-related gene expression reveals novel pathways to obstructive sleep apnea in humans. Respir Physiol Neurobiol 189:649–654. doi:10.1016/j.resp.2013.08.012
Liu Y, Patel S, Nibbe R, et al. (2011) Systems biology analyses of gene expression and genome wide association study data in obstructive sleep apnea. Pac Symp Biocomput Pac Symp Biocomput 14–25
Gharib SA, Hayes AL, Rosen MJ, Patel SR (2013) A pathway-based analysis on the effects of obstructive sleep apnea in modulating visceral fat transcriptome. Sleep 36:23–30. doi:10.5665/sleep.2294
Kim J, Bhattacharjee R, Khalyfa A, et al. (2012) DNA methylation in inflammatory genes among children with obstructive sleep apnea. Am J Respir Crit Care Med 185:330–338. doi:10.1164/rccm.201106-1026OC
Zhang J, Guo X, Shi Y, et al. (2014) Intermittent hypoxia with or without hypercapnia is associated with tumorigenesis by decreasing the expression of brain derived neurotrophic factor and miR-34a in rats. Chin Med J 127:43–47
Liu C, Kelnar K, Liu B, et al. (2011) The microRNA miR-34a inhibits prostate cancer stem cells and metastasis by directly repressing CD44. Nat Med 17:211–215. doi:10.1038/nm.2284
Jiang P, Liu R, Zheng Y, et al. (2012) MiR-34a inhibits lipopolysaccharide-induced inflammatory response through targeting Notch1 in murine macrophages. Exp Cell Res 318:1175–1184. doi:10.1016/j.yexcr.2012.03.018
Acknowledgments
This work was supported by grants from the Associação Fundo de Incentivo à Pesquisa (AFIP), Fundação de Amparo à Pesquisa do Estado de São Paulo, Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior (CAPES), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Conflict of interest
The authors declare that they have no competinginterests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Lima, F.F.F., Mazzotti, D.R., Tufik, S. et al. The role inflammatory response genes in obstructive sleep apnea syndrome: a review. Sleep Breath 20, 331–338 (2016). https://doi.org/10.1007/s11325-015-1226-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-015-1226-7