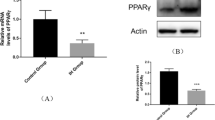
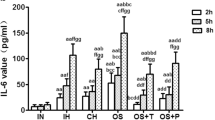
Abstract
Objective
Obstructive sleep apnea (OSA) has been implicated as a risk factor for atherosclerosis. Intermittent hypoxia (IH) induces oxidative and immuno-inflammatory alterations that could contribute to atherosclerosis. The aim of this study was to examine the effect of lymphocytes from intermittent hypoxia-exposed rats on the apoptotic signals in endothelial cells and the interventional role of tempol.
Method
Male Wistar rats were randomly distributed into three groups (n = 8 each): control group, IH group, and tempol group (exposed to IH and treated with tempol). Lymphocytes isolated from the rats were coincubated subsequently with endothelial cells under normoxia or IH condition. We analyzed endothelial apoptosis-related proteins (Bcl-2, Bax, and caspase-3) by Western blotting and measured the marks of oxidative stress (MDA, SOD, and CAT) and inflammation (TNF-α, IL-8, CRP, and ICAM-1) in cocultural supernatants by ELISA. We also determined endothelial p22phox, c-fos, and HIF-1α messenger RNA (mRNA) expressions using real time PCR.
Results
A significant decrease in the Bcl-2 level and the Bcl-2/Bax ratio and a significant increase in Bax and caspase-3 levels in endothelial cells were observed when coincubated normoxically with lymphocytes from IH-exposed rats compared to control (P < 0.01). Moreover, the oxidative stress, inflammation, and mRNA expressions of endothelial p22phox, c-fos, and HIF-1α were elevated significantly (P < 0.01). The alterations induced by lymphocytes were partially restored by tempol pretreatment while exacerbated by intermittent hypoxic coincubation.
Conclusions
Lymphocytes from intermittent hypoxia-exposed rats increased the apoptotic signals in endothelial cells via oxidative and inflammatory injury in vitro, which could be attenuated by tempol.





Similar content being viewed by others
References
Young T, Finn L, Peppard PE, Szklo-Coxe M, Austin D, Nieto FJ, Stubbs R, Hla KM (2008) Sleep disordered breathing and mortality: eighteen-year follow-up of the Wisconsin sleep cohort. Sleep 31:1071–1078
Quercioli A, Mach F, Montecucco F (2010) Inflammation accelerates atherosclerotic processes in obstructive sleep apnea syndrome (OSAS). Sleep Breath 14:261–269
Feng J, Chen BY, Cui LY, Wang BL, Liu CX, Chen PF, Guo MN, Dong LX, Li S (2009) Inflammation status of rabbit carotid artery model endothelium during intermittent hypoxia exposure and its relationship with leptin. Sleep Breath 13:277–283
Khayat R, Patt B, Hayes D Jr (2009) Obstructive sleep apnea: the new cardiovascular disease. Part I: obstructive sleep apnea and the pathogenesis of vascular disease. Heart Fail Rev 14:143–153
Choy J, Granville D, Hunt D, McManus B (2001) Endothelial cell apoptosis: biochemical characteristics and potential implications for atherosclerosis. J Mol Cell Cardiol 33:1673–1690
Tricot O, Mallat Z, Heymes C, Belmin J, Lesèche G, Tedgui A (2000) Relation between endothelial cell apoptosis and blood flow direction in human atherosclerotic plaques. Circulation 101:2450–2453
Arnaud C, Dematteis M, Pépin JL, Baguet JP, Lévy P (2009) Obstructive sleep apnea syndrome, immuno-inflammation and atherosclerosis. Semin Immunopathol 31:113–125
Dyugovskaya L, Lavie P, Lavie L (2005) Lymphocyte activation as a possible measure of atherosclerotic risk in patients with sleep apnea. Ann NY Acad Sci 1051:340–350
Dyugovskaya L, Lavie P, Hirsh M, Lavie L (2005) Activated CD8+ T-lymphocytes in obstructive sleep apnoea. Eur Respir J 25:820–828
Dyugovskaya L, Lavie P, Lavie L (2003) Phenotypic and functional characterization of blood gammadelta T cells in sleep apnea. Am J Respir Crit Care Med 168:242–249
Williams RJ, Motteram JM, Sharp CH, Gallagher PJ (1992) Dietary vitamin E and the attenuation of early lesion development in modified Watanabe rabbits. Atherosclerosis 94:153–159
Kita T, Nagano Y, Yokode M, Ishii K, Kume N, Ooshima A, Yoshida H, Kawai C (1987) Probucol prevents the progression of atherosclerosis in Watanabe heritable hyperlipidemic rabbit, an animal model for familial hypercholesterolemia. Proc Natl Acad Sci U S A 84:5928–5931
Cannizzo B, Quesada I, Militello R, Amaya C, Miatello R, Cruzado M, Castro C (2014) Tempol attenuates atherosclerosis associated with metabolic syndrome via decreased vascular inflammation and NADPH-2 oxidase expression. Free Radic Res 48:526–533
Caldwell CC, Kojima H, Lukashev D, Armstrong J, Farber M, Apasov SG, Sitkovsky MV (2001) Differential effects of physiologically relevant hypoxic conditions on T lymphocyte development and effector functions. J Immunol 167:6140–6149
Zhou W, Wan N, Zhang Z, Guo R, Chen B (2012) Effects of various degrees of oxidative stress induced by intermittent hypoxia in rat myocardial tissues. Respirology 17:821–829
He Q, Yang QC, Zhou Q, Zhu H, Niu WY, Feng J, Wang Y, Cao J, Chen BY (2014) Effects of varying degrees of intermittent hypoxia on proinflammatory cytokines and adipokines in rats and 3 T3-L1 adipocytes. PLoS One 9:e86326
Bauriedel G, Hutter R, Welsch U, Bach R, Sievert H, Lüderitz B (1999) Role of smooth muscle cell death in advanced coronary primary lesions: implications for plaque instability. Cardiovasc Res 41:480–488
Lavie L, Lavie P (2009) Molecular mechanisms of cardiovascular disease in OSAHS: the oxidative stress link. Eur Respir J 33:1467–1484
Chen F, Qian LH, Deng B, Liu ZM, Zhao Y, Le YY (2013) Resveratrol protects vascular endothelial cells from high glucose-induced apoptosis through inhibition of NADPH oxidase activation-driven oxidative stress. CNS Neurosci Ther 19:675–681
Fu R, Wang Q, Guo Q, Xu J, Wu X (2013) XJP-1 protects endothelial cells from oxidized low-density lipoprotein-induced apoptosis by inhibiting NADPH oxidase subunit expression and modulating the PI3K/Akt/eNOS pathway. Vascul Pharmacol 58:78–86
Quagliaro L, Piconi L, Assaloni R, Martinelli L, Motz E, Ceriello A (2003) Intermittent high glucose enhances apoptosis related to oxidative stress in human umbilical vein endothelial cells: the role of protein kinase C and NAD(P)H-oxidase activation. Diabetes 52:2795–2804
Spillmann F, Van Linthout S, Miteva K, Lorenz M, Stangl V, Schultheiss HP, Tschöpe C (2014) LXR agonism improves TNF-α-induced endothelial dysfunction in the absence of its cholesterol- modulating effects. Atherosclerosis 232:1–9
Lavie L (2003) Obstructive sleep apnoea syndrome—an oxidative stress disorder. Sleep Med Rev 7:35–51
Haverkate F, Thompson SG, Pyke SDM, Gallimore JR, Pepys MB (1997) Production of C-reactive protein and risk of coronary events in stable and unstable angina. Lancet 349:462–466
Nabata A, Kuroki M, Ueba H, Hashimoto S, Umemoto T, Wada H, Yasu T, Saito M, Momomura S, Kawakami M (2008) C-reactive protein induces endothelial cell apoptosis and matrix metalloproteinase-9 production in human mononuclear cells: implications for the destabilization of atherosclerotic plaque. Atherosclerosis 196:129–135
Zhang S, Zhao Y, Xu M, Yu L, Zhao Y, Chen J, Yuan Y, Zheng Q, Niu X (2013) FoxO3a modulates hypoxia stress induced oxidative stress and apoptosis in cardiac microvascular endothelial cells. PLoS One 8:e80342
Zhu J, Rebecchi MJ, Wang Q, Glass PS, Brink PR, Liu L (2013) Chronic Tempol treatment restores pharmacological preconditioning in the senescent rat heart. Am J Physiol Heart Circ Physiol 304:H649–H659
Lavie L (2012) Oxidative stress inflammation and endothelial dysfunction in obstructive sleep apnea. Front Biosci 4:1391–1403
Yang YY, Shang J, Liu HG (2013) Role of endoplasmic reticular stress in aortic endothelial apoptosis induced by intermittent/persistent hypoxia. Chin Med J 126:4517–4523
Acknowledgments
This work was supported by the grants from the National Natural Science Foundation of China (No. 81170071, 81270144 and 81100060).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hengjuan Guo and Jie Cao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Guo, H., Cao, J., Li, J. et al. Lymphocytes from intermittent hypoxia-exposed rats increase the apoptotic signals in endothelial cells via oxidative and inflammatory injury in vitro. Sleep Breath 19, 969–976 (2015). https://doi.org/10.1007/s11325-015-1128-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-015-1128-8