Abstract
Background
Seronegative elderly-onset rheumatoid arthritis (EORA)neg and polymyalgia rheumatica (PMR) have similar clinical characteristics making them difficult to distinguish based on clinical features. We hypothesized that the study of serum metabolome could identify potential biomarkers of PMR vs. EORAneg.
Methods
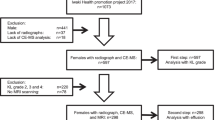
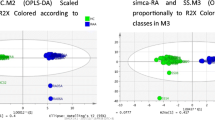
Arthritis in older adults (ARTIEL) is an observational prospective cohort with patients older than 60 years of age with newly diagnosed arthritis. Patients’ blood samples were compared at baseline with 18 controls. A thorough clinical examination was conducted. A Bruker Avance 600 MHz spectrometer was used to acquire Nuclear Magnetic Resonance (NMR) spectra of serum samples. Chenomx NMR suite 8.5 was used for metabolite identification and quantification.Student t-test, one-way ANOVA, binary linear regression and ROC curve, Pearson’s correlation along with pathway analyses were conducted.
Results
Twenty-eight patients were diagnosed with EORAneg and 20 with PMR. EORAneg patients had a mean disease activity score (DAS)-Erythrocyte Sedimentation Rate (ESR) of 6.21 ± 1.00. All PMR patients reported shoulder pain, and 90% reported pelvic pain. Fifty-eight polar metabolites were identified. Of these, 3-hydroxybutyrate, acetate, glucose, glycine, lactate, and o-acetylcholine (o-ACh), were significantly different between groups. Of interest, IL-6 correlated with different metabolites in PMR and EORAneg suggesting different inflammatory activated pathways. Finally, lactate, o-ACh, taurine, and sex (female) were identified as distinguishable factors of PMR from EORAneg with a sensitivity of 90%, specificity of 92.3%, and an AUC of 0.925 (p < 0.001).
Conclusion
These results suggest that EORAneg and PMR have different serum metabolomic profiles that might be related to their pathobiology and can be used as biomarker to discriminate between both diseases.




Similar content being viewed by others
Data Availability
The datasets of metabolites generated during the current study are available from the corresponding author on reasonable request. All data generated or analyzed during this study are included in this published article and its supplementary information files.
Abbreviations
- PMR:
-
Polymyalgia Rheumatica
- EORAneg :
-
Seronegative Elderly Onset Rheumatoid Arthritis. TMAO:Trimethylamine N-Oxide
- SG3PC:
-
Sn-Glycero-3-Phosphocholine. R:Responder
- NR:
-
Non-Responder. DM:Diabetes Mellitus
- HTA:
-
Hypertension
- DLP:
-
Dyslipidemia
- BMI:
-
Body Mass Index. ESR:Erythrocyte Sedimentation Rate
- CRP:
-
C-Reactive protein
- IL-6:
-
Interleukin-6
- CRP:
-
C-Reactive Protein. NSAIDs:Non-Steroidal Anti-Inflammatory Drugs. HAQ:Health Assessment Questionnaire. ANOVA:Analysis of Variance
- O-PLSDA:
-
Orthogonal Partial Least-Squares Discriminant Analysis
- VIP:
-
Variable Importance in Projection
- AUC:
-
Area Under the Curve. 1 H-NMR:Hydrogen-Nuclear Magnetic Resonance.
References
Aletaha, D., Neogi, T., Silman, A. J., Funovits, J., Felson, D. T., Bingham, C. O. 3rd, Birnbaum, N. S., Burmester, G. R., Bykerk, V. P., Cohen, M. D., Combe, B., Costenbader, K. H., Dougados, M., Emery, P., Ferraccioli, G., Hazes, J. M., Hobbs, K., Huizinga, T. W., Kavanaugh, A., Kay, J., Kvien, T. K., Laing, T., Mease, P., Ménard, H. A., Moreland, L. W., Naden, R. L., Pincus, T., Smolen, J. S., Stanislawska-Biernat, E., Symmons, D., Tak, P. P., Upchurch, K. S., Vencovský, J., Wolfe, F., & Hawker, G. (2010). 2010 rheumatoid arthritis classification criteria: An American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis And Rheumatism, 62(9), 2569–2581. https://doi.org/10.1002/art.27584.
Baghdadi, L. R. (2020). Effect of methotrexate use on the development of type 2 diabetes in rheumatoid arthritis patients: A systematic review and meta-analysis. PLoS One, 15(7), e0235637. https://doi.org/10.1371/journal.pone.0235637.
Biniecka, M., Canavan, M., McGarry, T., Gao, W., McCormick, J., Cregan, S., Gallagher, L., Smith, T., Phelan, J. J., Ryan, J., O’Sullivan, J., Ng, C. T., Veale, D. J., & Fearon, U. (2016). Dysregulated bioenergetics: A key regulator of joint inflammation. Annals Of The Rheumatic Diseases, 75(12), 2192–2200. https://doi.org/10.1136/annrheumdis-2015-208476.
Bustamante, M. F., Oliveira, P. G., Garcia-Carbonell, R., Croft, A. P., Smith, J. M., Serrano, R. L., Sanchez-Lopez, E., Liu, X., Kisseleva, T., Hay, N., Buckley, C. D., Firestein, G. S., Murphy, A. N., Miyamoto, S., & Guma, M. (2018). Hexokinase 2 as a novel selective metabolic target for rheumatoid arthritis. Annals Of The Rheumatic Diseases, 77(11), 1636–1643. https://doi.org/10.1136/annrheumdis-2018-213103.
Caporali, R., Montecucco, C., Epis, O., Bobbio-Pallavicini, F., Maio, T., & Cimmino, M. A. (2001). Presenting features of polymyalgia rheumatica (PMR) and rheumatoid arthritis with PMR-like onset: A prospective study. Annals of the Rheumatic Diseases, 60(11), 1021. https://doi.org/10.1136/ard.60.11.1021.
Chiba, T., Sakuma, K., Komatsu, T., Cao, X., Aimoto, M., Nagasawa, Y., Shimizu, K., Takahashi, M., Hori, Y., Shirai, K., & Takahara, A. (2019). Physiological role of nitric oxide for regulation of arterial stiffness in anesthetized rabbits. Journal of Pharmacological Sciences, 139(1), 42–45. https://doi.org/10.1016/j.jphs.2018.11.003.
Cho, K., Mahieu, N. G., Johnson, S. L., & Patti, G. J. (2014). After the feature presentation: Technologies bridging untargeted metabolomics and biology. Current Opinion In Biotechnology, 28, 143–148. https://doi.org/10.1016/j.copbio.2014.04.006.
Chong, I. G., & Jun, C. H. (2005). Performance of some variable selection methods when multicollinearity is present. Chemometrics and Intelligent Laboratory Systems, 78(1), 103–112. https://doi.org/10.1016/j.chemolab.2004.12.011.
Coras, R., Pedersen, B., Narasimhan, R., Brandy, A., Mateo, L., Prior-Español, A., Kavanaugh, A., Armando, A. M., Jain, M., Quehenberger, O., Martínez-Morillo, M., & Guma, M. (2021). Imbalance between Omega-6- and omega-3-Derived bioactive lipids in arthritis in older adults. Journals Of Gerontology. Series A, Biological Sciences And Medical Sciences, 76(3), 415–425. https://doi.org/10.1093/gerona/glaa113.
Costenbader, K. H., DiIorio, M., Chu, S. H., Cui, J., Sparks, J. A., Lu, B., Moss, L., Kelmenson, L., Feser, M., Edison, J., Clish, C., Lasky-Su, J., Deane, K. D., & Karlson, E. W. (2021). Circulating blood metabolite trajectories and risk of rheumatoid arthritis among military personnel in the Department of Defense Biorepository. Annals Of The Rheumatic Diseases. https://doi.org/10.1136/annrheumdis-2020-219682.
Cuppen, B. V., Fu, J., van Wietmarschen, H. A., Harms, A. C., Koval, S., Marijnissen, A. C., Peeters, J. J., Bijlsma, J. W., Tekstra, J., van Laar, J. M., Hankemeier, T., Lafeber, F. P., & van der Greef, J. (2016). Exploring the Inflammatory Metabolomic Profile to predict response to TNF-α inhibitors in rheumatoid arthritis. PLoS One, 11(9), e0163087. https://doi.org/10.1371/journal.pone.0163087.
Cutolo, M., Montecucco, C. M., Cavagna, L., Caporali, R., Capellino, S., Montagna, P., Fazzuoli, L., Villaggio, B., Seriolo, B., & Sulli, A. (2006). Serum cytokines and steroidal hormones in polymyalgia rheumatica and elderly-onset rheumatoid arthritis. Annals of the Rheumatic Diseases, 65(11), 1438. https://doi.org/10.1136/ard.2006.051979.
Dasgupta, B., Cimmino, M. A., Maradit-Kremers, H., Schmidt, W. A., Schirmer, M., Salvarani, C., Bachta, A., Dejaco, C., Duftner, C., Jensen, H. S., Duhaut, P., Poór, G., Kaposi, N. P., Mandl, P., Balint, P. V., Schmidt, Z., Iagnocco, A., Nannini, C., Cantini, F., Macchioni, P., Pipitone, N., Amo, M. D., Espígol-Frigolé, G., Cid, M. C., Martínez-Taboada, V. M., Nordborg, E., Direskeneli, H., Aydin, S. Z., Ahmed, K., Hazleman, B., Silverman, B., Pease, C., Wakefield, R. J., Luqmani, R., Abril, A., Michet, C. J., Marcus, R., Gonter, N. J., Maz, M., Carter, R. E., Crowson, C. S., & Matteson, E. L. (2012). 2012 provisional classification criteria for polymyalgia rheumatica: A European League Against Rheumatism/American College of Rheumatology collaborative initiative. Annals Of The Rheumatic Diseases, 71(4), 484–492. https://doi.org/10.1136/annrheumdis-2011-200329.
de Oliveira, P. G., Farinon, M., Sanchez-Lopez, E., Miyamoto, S., & Guma, M. (2019). Fibroblast-like Synoviocytes glucose metabolism as a therapeutic target in rheumatoid arthritis. Frontiers In Immunology, 10, 1743. https://doi.org/10.3389/fimmu.2019.01743.
Di Costanzo, A., Paris, D., Melck, D., Angiolillo, A., Corso, G., Maniscalco, M., & Motta, A. (2020). Blood biomarkers indicate that the preclinical stages of Alzheimer’s disease present overlapping molecular features. Scientific Reports, 10(1), 15612. https://doi.org/10.1038/s41598-020-71832-y.
Dumas, M. E., Maibaum, E. C., Teague, C., Ueshima, H., Zhou, B., Lindon, J. C., Nicholson, J. K., Stamler, J., Elliott, P., Chan, Q., & Holmes, E. (2006). Assessment of analytical reproducibility of 1H NMR spectroscopy based metabonomics for large-scale epidemiological research: The INTERMAP Study. Analytical chemistry, 78(7), 2199–2208. https://doi.org/10.1021/ac0517085.
Ehrenpreis, S. (1982). D-phenylalanine and other enkephalinase inhibitors as pharmacological agents: Implications for some important therapeutic application. Acupuncture And Electro-Therapeutics Research, 7(2–3), 157–172. https://doi.org/10.3727/036012982816952099.
Falconer, J., Murphy, A. N., Young, S. P., Clark, A. R., Tiziani, S., Guma, M., & Buckley, C. D. (2018). Review: Synovial cell metabolism and chronic inflammation in rheumatoid arthritis. Arthritis Rheumatol, 70(7), 984–999. https://doi.org/10.1002/art.40504.
Garcia-Carbonell, R., Divakaruni, A. S., Lodi, A., Vicente-Suarez, I., Saha, A., Cheroutre, H., Boss, G. R., Tiziani, S., Murphy, A. N., & Guma, M. (2016). Critical role of glucose metabolism in rheumatoid arthritis fibroblast-like synoviocytes. Arthritis Rheumatol, 68(7), 1614–1626. https://doi.org/10.1002/art.39608.
Hancock, A. T., Mallen, C. D., Muller, S., Belcher, J., Roddy, E., Helliwell, T., & Hider, S. L. (2014). Risk of vascular events in patients with polymyalgia rheumatica. Cmaj, 186(13), E495–501. https://doi.org/10.1503/cmaj.140266.
Holmberg, C. N., Åstrand, A., Wingren, C., Garnett, J. P., Mayer, G., Taylor, J. D., Baker, E. H., & Baines, D. L. (2017). Differential Effect of LPS on Glucose, Lactate and Inflammatory Markers in the Lungs of Normal and Diabetic Mice.J Pulm Respir Med, 2017(1).
Kanehisa, M., Sato, Y., Kawashima, M., Furumichi, M., & Tanabe, M. (2016). KEGG as a reference resource for gene and protein annotation. Nucleic Acids Research, 44(D1), D457–462. https://doi.org/10.1093/nar/gkv1070.
Kapoor, S. R., Filer, A., Fitzpatrick, M. A., Fisher, B. A., Taylor, P. C., Buckley, C. D., McInnes, I. B., Raza, K., & Young, S. P. (2013). Metabolic profiling predicts response to anti-tumor necrosis factor alpha therapy in patients with rheumatoid arthritis [Multicenter Study Research Support, N. U. S., & Gov’t]. Arthritis and rheumatism, 65(6), 1448–1456.https://doi.org/10.1002/art.37921.
Kim, S., Hwang, J., Xuan, J., Jung, Y. H., Cha, H. S., & Kim, K. H. (2014). Global metabolite profiling of synovial fluid for the specific diagnosis of rheumatoid arthritis from other inflammatory arthritis [Research Support, Non-U.S. Gov’t]. PLoS One, 9(6), e97501. https://doi.org/10.1371/journal.pone.0097501.
Kobak, S., & Bes, C. (2018). An autumn tale: Geriatric rheumatoid arthritis. Ther Adv Musculoskelet Dis, 10(1), 3–11. https://doi.org/10.1177/1759720x17740075.
Kosinska, M. K., Liebisch, G., Lochnit, G., Wilhelm, J., Klein, H., Kaesser, U., Lasczkowski, G., Rickert, M., Schmitz, G., & Steinmeyer, J. (2013). A lipidomic study of phospholipid classes and species in human synovial fluid [Research Support, Non-U.S. Gov’t]. Arthritis and rheumatism, 65(9), 2323–2333. https://doi.org/10.1002/art.38053.
Le Gall, G., Noor, S. O., Ridgway, K., Scovell, L., Jamieson, C., Johnson, I. T., Colquhoun, I. J., Kemsley, E. K., & Narbad, A. (2011). Metabolomics of fecal extracts detects altered metabolic activity of gut microbiota in ulcerative colitis and irritable bowel syndrome. Journal Of Proteome Research, 10(9), 4208–4218. https://doi.org/10.1021/pr2003598.
Luan, H., Gu, W., Li, H., Wang, Z., Lu, L., Ke, M., Lu, J., Chen, W., Lan, Z., Xiao, Y., Xu, J., Zhang, Y., Cai, Z., Liu, S., & Zhang, W. (2021). Serum metabolomic and lipidomic profiling identifies diagnostic biomarkers for seropositive and seronegative rheumatoid arthritis patients. J Transl Med, 19(1), 500. https://doi.org/10.1186/s12967-021-03169-7.
Madsen, R. K., Lundstedt, T., Gabrielsson, J., Sennbro, C. J., Alenius, G. M., Moritz, T., Rantapaa-Dahlqvist, S., & Trygg, J. (2011). Diagnostic properties of metabolic perturbations in rheumatoid arthritis [Research Support, Non-U.S. Gov’t Validation Studies]. Arthritis research & therapy, 13(1), R19. https://doi.org/10.1186/ar3243.
Manzo, C. (2019). Incidence and prevalence of Polymyalgia Rheumatica (PMR): The importance of the Epidemiological Context. The italian case. Med Sci (Basel), 7(9), https://doi.org/10.3390/medsci7090092.
Medcalf, M. R., Bhadbhade, P., Mikuls, T. R., O’Dell, J. R., Gundry, R. L., & Funk, R. S. (2021). Plasma metabolome normalization in rheumatoid arthritis following initiation of Methotrexate and the identification of metabolic biomarkers of efficacy. Metabolites, 11(12), https://doi.org/10.3390/metabo11120824.
Misra, B. B. (2020). Data normalization strategies in metabolomics: Current challenges, approaches, and tools. European Journal of Mass Spectrometry, 26(3), 165–174. https://doi.org/10.1177/1469066720918446.
Murillo-Saich, J. D., Diaz-Torne, C., Ortiz, M. A., Coras, R., Gil-Alabarse, P., Pedersen, A., Corominas, H., Vidal, S., & Guma, M. (2021). Metabolomics profiling predicts outcome of tocilizumab in rheumatoid arthritis: An exploratory study. Metabolomics, 17(9), 74. https://doi.org/10.1007/s11306-021-01822-2.
Nyamundanda, G., Gormley, I. C., Fan, Y., et al. (2013). MetSizeR: Selecting the optimal sample size for metabolomic studies using an analysis based approach. Bmc Bioinformatics, 14, 338. https://doi.org/10.1186/1471-2105-14-338.
Parada Venegas, D., De la Fuente, M. K., Landskron, G., González, M. J., Quera, R., Dijkstra, G., Harmsen, H. J. M., Faber, K. N., & Hermoso, M. A. (2019). Short chain fatty acids (SCFAs)-Mediated gut epithelial and Immune Regulation and its relevance for inflammatory Bowel Diseases [Review]. Frontiers in Immunology, 10(277), https://doi.org/10.3389/fimmu.2019.00277.
Rodríguez-Carrio, J., Coras, R., Alperi-López, M., López, P., Ulloa, C., Ballina-García, F. J., Armando, A. M., Quehenberger, O., Guma, M., & Suárez, A. (2021). Profiling of serum oxylipins during the earliest stages of rheumatoid arthritis. Arthritis & Rheumatology, 73(3), 401–413. https://doi.org/10.1002/art.41537.
Rosas-Ballina, M., Olofsson, P. S., Ochani, M., Valdés-Ferrer, S. I., Levine, Y. A., Reardon, C., Tusche, M. W., Pavlov, V. A., Andersson, U., Chavan, S., Mak, T. W., & Tracey, K. J. (2011). Acetylcholine-synthesizing T cells relay neural signals in a vagus nerve circuit. Science, 334(6052), 98–101. https://doi.org/10.1126/science.1209985.
Souto-Carneiro, M., Toth, L., Behnisch, R., Urbach, K., Klika, K. D., Carvalho, R. A., & Lorenz, H. M. (2020). Differences in the serum metabolome and lipidome identify potential biomarkers for seronegative rheumatoid arthritis versus psoriatic arthritis. Annals Of The Rheumatic Diseases, 79(4), 499–506. https://doi.org/10.1136/annrheumdis-2019-216374.
Sweeney, S. R., Kavanaugh, A., Lodi, A., Wang, B., Boyle, D., Tiziani, S., & Guma, M. (2016). Metabolomic profiling predicts outcome of rituximab therapy in rheumatoid arthritis. RMD open, 2(2), e000289. https://doi.org/10.1136/rmdopen-2016-000289.
Tatar, Z., Migne, C., Petera, M., Gaudin, P., Lequerre, T., Marotte, H., Tebib, J., Pujos Guillot, E., & Soubrier, M. (2016). Variations in the metabolome in response to disease activity of rheumatoid arthritis. Bmc Musculoskeletal Disorders, 17(1), 353. https://doi.org/10.1186/s12891-016-1214-5.
Teitsma, X. M., Yang, W., Jacobs, J. W. G., Pethö-Schramm, A., Borm, M. E. A., Harms, A. C., Hankemeier, T., van Laar, J. M., Bijlsma, J. W. J., & Lafeber, F. (2018). Baseline metabolic profiles of early rheumatoid arthritis patients achieving sustained drug-free remission after initiating treat-to-target tocilizumab, methotrexate, or the combination: Insights from systems biology. Arthritis Res Ther, 20(1), 230. https://doi.org/10.1186/s13075-018-1729-2.
Todeschini, R., Ballabio, D., Consonni, V., Sahigara, F., & Filzmoser, P. (2013). Locally centred Mahalanobis distance: A new distance measure with salient features towards outlier detection. Analytica Chimica Acta, 787, 1–9. https://doi.org/10.1016/j.aca.2013.04.034.
van Riel, P. L., & Renskers, L. (2016). The Disease activity score (DAS) and the Disease Activity score using 28 joint counts (DAS28) in the management of rheumatoid arthritis. Clinical And Experimental Rheumatology, 34(5 Suppl 101), S40–s44.
Viant, M. R., Bearden, D. W., Bundy, J. G., Burton, I. W., Collette, T. W., Ekman, D. R., Ezernieks, V., Karakach, T. K., Lin, C. Y., Rochfort, S., de Ropp, J. S., Teng, Q., Tjeerdema, R. S., Walter, J. A., & Wu, H. (2009). International NMR-based environmental metabolomics intercomparison exercise. Environmental Science And Technology, 43(1), 219–225. https://doi.org/10.1021/es802198z.
Wakura, D., Kotani, T., Takeuchi, T., Komori, T., Yoshida, S., Makino, S., & Hanafusa, T. (2016). Differentiation between Polymyalgia Rheumatica (PMR) and Elderly-Onset Rheumatoid Arthritis using 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed tomography: Is Enthesitis a New Pathological Lesion in PMR? PLoS One, 11(7), e0158509. https://doi.org/10.1371/journal.pone.0158509.
Wang, H., Liao, H., Ochani, M., Justiniani, M., Lin, X., Yang, L., Al-Abed, Y., Wang, H., Metz, C., Miller, E. J., Tracey, K. J., & Ulloa, L. (2004). Cholinergic agonists inhibit HMGB1 release and improve survival in experimental sepsis. Nature Medicine, 10(11), 1216–1221. https://doi.org/10.1038/nm1124.
Wessler, I., & Kirkpatrick, C. J. (2008). Acetylcholine beyond neurons: The non-neuronal cholinergic system in humans. British Journal Of Pharmacology, 154(8), 1558–1571. https://doi.org/10.1038/bjp.2008.185.
Wieder, C., Bundy, J. G., Frainay, C., Poupin, N., Rodríguez-Mier, P., Vinson, F., Cooke, J., Lai, R. P. J., Jourdan, F., & Ebbels, T. M. D. (2022). Avoiding the misuse of Pathway Analysis Tools in Environmental Metabolomics. Environmental Science & Technology, 56(20), 14219–14222. https://doi.org/10.1021/acs.est.2c05588.
Wishart, D. S., Jewison, T., Guo, A. C., Wilson, M., Knox, C., Liu, Y., Djoumbou, Y., Mandal, R., Aziat, F., & Dong, E. (2012). HMDB 3.0—the human metabolome database in 2013.Nucleic acids research,gks1065.
Xia, J., & Wishart, D. S. (2011). Metabolomic data processing, analysis, and interpretation using MetaboAnalyst. Curr Protoc Bioinformatics. https://doi.org/10.1002/0471250953.bi1410s34. Chap. 14, Unit 14.10.
Xu, M., Jiang, Z., Wang, C., Li, N., Bo, L., Zha, Y., Bian, J., Zhang, Y., & Deng, X. (2019). Acetate attenuates inflammasome activation through GPR43-mediated Ca2+-dependent NLRP3 ubiquitination. Experimental & Molecular Medicine, 51(7), 1–13. https://doi.org/10.1038/s12276-019-0276-5.
Young, S. P., Kapoor, S. R., Viant, M. R., Byrne, J. J., Filer, A., Buckley, C. D., Kitas, G. D., & Raza, K. (2013). The impact of inflammation on metabolomic profiles in patients with arthritis. Arthritis And Rheumatism, 65(8), 2015–2023. https://doi.org/10.1002/art.38021.
Zhang, S., Nagana Gowda, G. A., Ye, T., & Raftery, D. (2010). Advances in NMR-based biofluid analysis and metabolite profiling. The Analyst, 135(7), 1490–1498. https://doi.org/10.1039/c000091d.
Zhong, Z., Wheeler, M. D., Li, X., Froh, M., Schemmer, P., Yin, M., Bunzendaul, H., Bradford, B., & Lemasters, J. J. (2003). L-Glycine: A novel antiinflammatory, immunomodulatory, and cytoprotective agent. Current Opinion In Clinical Nutrition And Metabolic Care, 6(2), 229–240. https://doi.org/10.1097/00075197-200303000-00013.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Institutes of Health (R01AR073324 to M.G., T32AR064194 to JDM-S and RC).
Author information
Authors and Affiliations
Contributions
Supervision of overall project: MG, MM. Conception of design: MG. Patient recruitment: MM, AB, AP, LM. Sample collection: AB, AP, LM, MM. Acquisition of 1 H-NMR data: AP. Analysis: JDM-S, MC, FC, RC. Interpretation of results: JDM-S, MC, RC, FC, LM, MM, MG. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Institutional Board Review at Clinic of the Hospital Universitari Germans Trias i Pujol with the number IP-13-001. All procedures performed in this study were in accordance with the ethical standards of the Institutional Board Review at Clinic of the Hospital Universitari Germans Trias i Pujol and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Competing interests
All the authors declare that they have no competing interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cedeno, M., Murillo-Saich, J., Coras, R. et al. Serum metabolomic profiling identifies potential biomarkers in arthritis in older adults: an exploratory study. Metabolomics 19, 37 (2023). https://doi.org/10.1007/s11306-023-02004-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-023-02004-y