Abstract
P2X receptors are expressed on different functional groups of primary afferent fibers. P2X receptor-mediated sensory inputs can be either innocuous or nociceptive, depending on which dorsal horn regions receive these inputs. We provide a brief review of P2X receptor-mediated purinergic sensory pathways to different regions in the dorsal horn. These P2X purinergic pathways are identified in normal animals, which provides insights into their physiological functions. Future studies on P2X purinergic pathways in animal models of pathological conditions may provide insights on how P2X receptors play a role in pathological pain states.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
P2X receptors and their expression on primary afferent neurons
P2X receptors are cation channels on the plasma membranes that open in response to the binding of extracellular ATP. Seven P2X subunits have been identified and cloned [1, 2]. These subunits can form a number of functional subtypes of recombinant P2X receptors in a heterologous expression system [1, 2]. Biophysical and pharmacological characterization of these P2X receptor subtypes have been extensively reviewed [1, 2]. It should be pointed out that the two terms, P2X subunit and P2X subtype, have distinct meanings. The former only refers to a component of a functional P2X receptor; the later is used for a functional P2X receptor. A functional P2X receptor can be formed from same P2X subunits (i.e., homomeric P2X receptors) or different subunits (i.e., heteromeric P2X receptors).
All of the seven P2X receptor subunits appear to be expressed on primary afferent neurons in the spinal dorsal root ganglia (DRG) and trigeminal ganglia [3, 4]. ATP and other P2X receptor agonists can evoke membrane currents in many primary afferent neurons. The evoked currents show three distinct phenotypes: Fast current, slow current, and mixed current with both fast and slow components [5–10]. The fast current is manifested by rapid desensitization in the range of milliseconds in the presence of agonists. In contrast, the slow current displays weak or little desensitization in the range of seconds in the presence of agonists. By comparing the findings from primary afferent neurons with those recombinant P2X receptors, it has been suggested that homomeric P2X3 receptors (P2X3) account for fast currents, heteromeric P2X receptors composed of P2X2 and P2X3 subunits (P2X2 + 3 receptors) account for slow currents, and the co-expression of P2X3 and P2X2 + 3 receptors account for the mixed currents [11–16]. However, for many DRG neurons slow currents appear to also be mediated by P2X receptor subtypes other than P2X2 + 3 receptors [10].
Studies have shown that P2X receptors can be involved in both peripheral and central sensory signaling and processing [11, 12, 17–24]. In the periphery, ATP may be released as a result of tissue stretch, injury and inflammation, visceral distension, or sympathetic activation [25]. ATP release can excite afferent fibers by the activation of P2X receptors [25–27]. Behavioral studies indicate that ATP and P2X receptors are involved in both nociceptive and innocuous functions [28–32]. P2X receptors are suggested to play roles in nociception under conditions of acute tissue injury and inflammation. Furthermore, P2X receptors have been implicated in neuropathic pain conditions [19, 33–35]. At central sites in the spinal cord dorsal horn, sensory impulses can release ATP [23], which may arise from afferent central terminals, second-order neurons, or astrocytes [36]. ATP can also be released centrally during spinal cord tissue damage and inflammation as a result of numerous disorders in the spinal cord. Centrally released ATP [37] may act on the central terminals of afferent fibers, which may then modulate or directly evoke the release of neurotransmitters from afferent central terminals [23, 24, 38]. This action may represent a major function of ATP and P2X receptors at the central sites in sensory pathways. P2X receptors on afferent central terminals have novel and important implications in the centrally initiated sensory signals including neuropathic pain associated with disorders in the spinal cord. Furthermore, P2X receptor-mediated modulation of transmitter release at afferent central terminals can also be a novel mechanism for the sensitization of sensory inputs from the periphery [39].
P2X receptor-mediated sensory pathways to different regions of the spinal cord dorsal horn
Studies on the nociceptive functions of P2X receptors are still at an early stage. To understand the nociceptive functions of P2X receptors under both physiological and pathological conditions, it is essential to identify P2X-mediated nociceptive pathways and to know where and how P2X-mediated nociceptive inputs are transmitted and processed in the spinal cord dorsal horn (DH). The dorsal horn, comprising laminae I and II (superficial laminae), III and IV (intermediate part), and V and VI (deep laminae), is the primary central site for processing somatic sensory inputs [40, 41]. Both the superficial and the deep laminae of the DH are responsible in the reception, processing and transmission of nociceptive information [41–46]. In contrast, the intermediate part of the dorsal horn is mainly involved in processing non-nociceptive information [41].
P2X sensory pathways to the dorsal horn can be studied on spinal cord sections by immunochemistry with P2X antibodies and by synaptic physiology using the patch-clamp technique. These approaches are used to determine whether the central terminals of primary afferent fibers express P2X receptors and, if so, what types of P2X receptors are expressed and where these P2X-expressing terminals are located in the dorsal horn. These approaches for the study of P2X purinergic pathways are based on the assumption that if a type of P2X receptors is expressed at the central terminal of a primary afferent fiber, the peripheral site of the afferent fiber also expresses the same type of P2X receptors (Figure 1A). Thus, by studying P2X receptors at the central terminals of primary afferent fibers, one can map the sensory pathways of P2X purinergic inputs into the spinal cord dorsal horn. These approaches can help us understand the potential functions of different P2X subtypes in sensory transmission. However, although this assumption is likely to be true for most receptors, it should be noted that membrane receptors may not always be delivered to both ends of a primary afferent fiber.
An assumption of P2X purinergic pathway to the spinal cord dorsal horn and the synaptic physiological approach for the study of P2X purinergic pathway. A) The diagram illustrates a dorsal root ganglion with both peripheral nerve endings and central terminals. The central terminals are within the spinal cord dorsal horn. It is proposed that the expression of a P2X receptor at the central terminal also predicts its presence in other part of the primary afferent fibers including peripheral nerve endings. Thus, P2X purinergic pathway to the dorsal horn can be mapped by studying P2X receptors at the central terminals through synaptic physiology or immunochemistry. B) A schematic diagram illustrates the use of synaptic physiology to study P2X purinergic pathway to the dorsal horn using spinal cord slice preparations. Synaptic transmission between afferent central terminals and dorsal horn (DH) neurons is recorded using patch-clamp technique. Activation of P2X receptors on the central terminals of primary afferent fibers results in the release of glutamate, which in turn activate glutamate receptors (GluR) on dorsal horn neurons and generate excitatory postsynaptic currents (EPSCs).
In immunochemistry studies with P2X3 antibodies, P2X3-expressing afferent terminals [47–49] appear to be restricted to the inner part of lamina II (lamina IIi). Lamina distribution of afferent fibers that express other P2X subunits remains unclear, although there was a report that showed immunoreactivity of P2X1 and P2X2 subunits in superfacial lamina as well [50]. Except for P2X3 subunit antibodies, it appears that antibodies for other P2X receptor subunits have limited usefulness in the spinal cord sections. In addition, a further limitation of immunochemistry is that it only reveals P2X subunits rather than functional P2X receptors [36].
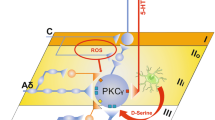
The use of synaptic physiology with patch-clamp recording technique for the study of P2X purinergic sensory pathways (Figure 1B) provides the opportunity to assess both function and structure in the system in several ways. First, this approach allows one to assess functional P2X receptors that are expressed on primary afferent central terminals. Second, it reveals the effects of P2X purinergic sensory inputs on dorsal horn neurons (i.e., the secondary-order sensory neurons within the dorsal horn). Third, it allows one to characterize neuronal circuits that are involved in processing P2X purinergic sensory inputs within the dorsal horn. To this end, we have applied patch-clamp recording technique to the spinal cord slice preparations to assess the effects of P2X receptor activation on monosynaptic and polysynaptic transmission from primary afferent fibers to dorsal horn neurons located in a number of lamina regions. We have found that P2X agonists increased monosynaptic transmission from afferent central terminals to the dorsal horn neurons located in lamina V (Figure 2) and lamina II [23, 24, 38]. The original aim of these studies was to explore the role of presynaptic P2X receptors, i.e., P2X receptors at the central terminal of primary afferent fibers in modulating glutamate release from P2X-expressing afferent central terminals. However, based on our assumption as illustrated in Figure 1, presynaptic P2X receptor-mediated increases of monosynaptic transmission have also revealed sensory pathways from some P2X-expressing afferent fibers to dorsal horn neurons in these regions (also see [36]). Recordings from lamina V neurons demonstrated that P2X receptor agonists produced a prolonged increase of monosynaptic transmission to the majority of lamina V neurons (Figure 2; [23, 24]). This finding suggests the wide expression of P2X receptors at the central terminals of these afferent fibers (Figure 3). These afferent fibers have been found to be Aδ afferent fibers and insensitive to capsaicin (Figures 2 and 3), a noxious stimulant that has been commonly used to identify nociceptive afferent fibers. Pharmacological studies have suggested that the P2X receptors expressed on these capsaicin-insensitive Ad afferent terminals were not P2X3 or P2X2 + 3 subtypes but were more likely to be P2X1 + 5 or P2X4 + 6 subtypes [10, 38]. One potential function of this P2X purinergic sensory pathway may be to transmit sensory information of mechanical stimuli, as based on sensory physiology of Aδ-afferent fibers that innervates lamina V of the spinal cord dorsal horn. Currently, it is unclear whether this P2X purinergic sensory pathway is directly involved in nociceptive transmission due to its lack of capsaicin sensitivity. However, nociceptive afferent fibers can also be capsaicin-insensitive. Interestingly, we have found that this ATP-sensitive/capsaicin-insensitive P2X purinergic pathway has convergence and temporal summation with a capsaicin-sensitive input that is polysynaptically transmitted to lamina V neurons (Figure 3; [24]). This convergence may suggest that this ATP-sensitive/capsaicin-insensitive P2X purinergic pathway has interaction with nociceptive input.
Effects of αβmeATP on the frequency of miniature excitatory postsynaptic currents recorded from lamina V dorsal horn neurons. A–B) Spinal cord slice preparation viewed under IR-DIC microscope. Lamina regions were identified under 10× objective (A). A part of a patch electrode is also seen in panel A. The electrode tip is inside tissue about 70 µM from the surface, and its lamina location is indicated by a box. A neuron in the box region can be seen under 40× objective (B, center of the field). The patch electrode is to the left side. C) A trace (top) shows mEPSCs recorded from a lamina V neuron before and following application of 100 µM αβmeATP. Bottom traces show, at an expanded time scale, the mEPSCs before (left three traces) and following (right three traces) αβmeATP application. D) Histogram shows the time course and degree of the increases in mEPSC frequency after 100 µM αβmeATP. It also shows, in the same recording, 2 µM capsaicin did not have effect on mEPSCs. Modified and reprinted (with permission) from Nakatsuka et al. [38]. Copyright by the Society for Neuroscience.
P2X-purinergic sensory pathways to the dorsal horn of the spinal cord. The schematic diagram illustrates three P2X-purinergic sensory pathways to laminas I, II, and V of the dorsal horn. The pathway to lamina II is the afferent fibers expressing both P2X3 and VR1 receptors. The P2X receptor subtypes of two other pathways had pharmacological properties distinct from P2X3 containing receptors.
In contrast to lamina V, many lamina II neurons are monosynaptically contacted by P2X3-expressing afferent fibers ([38], Figure 3). This result is consistent with immunochemistry of P2X3 subunit distribution in lamina II. These P2X3-expressing afferent fibers also express VR1 receptors [47, 51] and are sensitive to capsaicin. Thus, this P2X3 sensory pathway may be directly involved in transmitting noxious signals. Consistent with this idea, previous studies have shown the nociceptive function of P2X3 [35, 52–54]. The ATP-sensitive/capsaicin-sensitive pathway to lamina II was shown to converge to lamina V neurons through polysynaptic transmission ([24], Figure 3). This may mediate the spatial and temporal sensory summation and subsequent hyperactivity in deep laminae, a potential mechanism of hyperalgesia.
Another important region for pain transmission, processing, and the development of pathological pain is lamina I of the dorsal horn. Due to technical difficulties in performing electrophysiological recordings in lamina I region, there is no report on whether P2X-mediated sensory signals are transmitted to and processed in this important nociceptive region. Our recent experiments performed in lamina I show that some afferent fibers innervating lamina I neurons also express P2X receptors at their central sites (Figure 3, unpublished result, presented at the Purines 2004 meeting). These afferent fibers are capsaicin-sensitive fibers, suggesting that this is a nociceptive P2X purinergic pathway to the spinal cord dorsal horn. These P2X receptors are less likely to be P2X3 containing subtypes based on the restricted lamina IIi distribution of P2X3-expression afferent central terminals [47, 51]. It would be interesting to identify the P2X subtype or subtypes and to see whether it can be a selective target for the control of pain conditions.
Concluding remarks
The map of P2X purinergic sensory input to different laminas of the spinal cord dorsal horn helps in understanding the sensory functions of P2X receptors. Under pathological conditions, the destination of P2X purinergic sensory input in different laminas may be altered due to an aberrant expression of P2X receptors on primary afferent fibers [34, 55]. This can be a potential mechanism by which P2X receptors are involved in abnormal sensations such as mechanical allodynia.
Abbreviations
- ATP:
-
adenosine 5′-triphosphate
- DH:
-
dorsal horn
- DRG:
-
dorsal root ganglion
- EPSCs:
-
excitatory postsynaptic currents
- mEPSCs:
-
miniature excitatory postsynaptic currents
- VR1:
-
vanilloid receptor subtype 1
References
BS Khakh G Burnstock C Kennedy et al. (2001) ArticleTitleInternational union of pharmacology. XXIV. Current status of the nomenclature and properties of P2X receptors and their subunits Pharmacol Rev 53 107–118 Occurrence Handle11171941 Occurrence Handle1:CAS:528:DC%2BD3MXhsFWqtb8%3D
RA North (2002) ArticleTitleMolecular physiology of P2X receptors Physiol Rev 82 1013–1067 Occurrence Handle12270951 Occurrence Handle1:CAS:528:DC%2BD38Xot1Ggu78%3D
G Collo RA North E Kawashima et al. (1996) ArticleTitleCloning of P2X5 and P2X6 receptors and the distribution and properties of an extended family of ATP-gated ion channels J Neurosci 16 2495–2507 Occurrence Handle8786426 Occurrence Handle1:CAS:528:DyaK28XitFKhtbc%3D
Z Xiang X Bo G Burnstock (1998) ArticleTitleLocalization of ATP-gated P2X receptor immunoreactivity in rat sensory and sympathetic ganglia Neurosci Lett 256 105–108 Occurrence Handle10.1016/S0304-3940(98)00774-5 Occurrence Handle9853714 Occurrence Handle1:CAS:528:DyaK1cXmvFWlt7g%3D
EC Burgard W Niforatos T Biesen Particlevan et al. (1999) ArticleTitleP2X receptor-mediated ionic currents in dorsal root ganglion neurons J Neurophysiol 82 1590–1598 Occurrence Handle10482772 Occurrence Handle1:CAS:528:DyaK1MXmsVKku74%3D
S Ueno M Tsuda T Iwanaga et al. (1999) ArticleTitleCell type-specific ATP-activated responses in rat dorsal root ganglion neurons Br J Pharmacol 126 429–436 Occurrence Handle10.1038/sj.bjp.0702319 Occurrence Handle10077235 Occurrence Handle1:CAS:528:DyaK1MXht1ers7g%3D
C Li RW Peoples TH Lanthorn et al. (1999) ArticleTitleDistinct ATP-activated currents in different types of neurons dissociated from rat dorsal root ganglion Neurosci Lett 263 57–60 Occurrence Handle10.1016/S0304-3940(99)00114-7 Occurrence Handle10218910 Occurrence Handle1:CAS:528:DyaK1MXhvVClt7s%3D
JC Petruska BY Cooper RD Johnson et al. (2000) ArticleTitleDistribution patterns of different P2X receptor phenotypes in acutely dissociated dorsal root ganglion neurons of adult rats Exp Brain Res 134 126–132 Occurrence Handle10.1007/s002210000414 Occurrence Handle11026733 Occurrence Handle1:CAS:528:DC%2BD3cXnsVers7k%3D
JC Petruska N Mena T Nakatsuka et al. (2000) ArticleTitleP2X1 receptor subunit immunoreactivity and ATP-evoked fast currents in adult rat dorsal root ganglion neurons NeuroReport 11 3589–3592 Occurrence Handle10.1097/00001756-200011090-00037 Occurrence Handle11095524 Occurrence Handle1:CAS:528:DC%2BD3cXosFKnurs%3D
K Tsuzuki A Ase P Seguela et al. (2003) ArticleTitleTNP-ATP-resistant P2X ionic current on the central terminals and somata of rat primary sensory neurons J Neurophysiol 89 3235–3242 Occurrence Handle10.1152/jn.01171.2002 Occurrence Handle12783957 Occurrence Handle1:CAS:528:DC%2BD3sXlsFaju78%3D
C Lewis S Neidhart C Holy et al. (1995) ArticleTitleCoexpression of P2X2 and P2X3 receptor subunits can account for ATP-gated currents in sensory neurons Nature 377 432–435 Occurrence Handle10.1038/377432a0 Occurrence Handle7566120 Occurrence Handle1:CAS:528:DyaK2MXos1Slsr8%3D
SP Cook L Vulchanova KM Hargreaves et al. (1997) ArticleTitleDistinct ATP receptors on pain-sensing and stretch-sensing neurons Nature 387 505–508 Occurrence Handle10.1038/387505a0 Occurrence Handle9168113 Occurrence Handle1:CAS:528:DyaK2sXjsF2gurs%3D
KM Radford C Virginio A Surprenant et al. (1997) ArticleTitleBaculovirus expression provides direct evidence for heteromeric assembly of P2X2 and P2X3 receptors J Neurosci 17 6529–6533 Occurrence Handle9254665 Occurrence Handle1:CAS:528:DyaK2sXlslejurs%3D
M Liu BF King PM Dunn et al. (2001) ArticleTitleCoexpression of P2X3 and P2X2 receptor subunits in varying amounts generates heterogeneous populations of P2X receptors that evoke a spectrum of agonist responses comparable to that seen in sensory neurons J Pharmacol Exp Ther 296 1043–1050 Occurrence Handle11181939 Occurrence Handle1:CAS:528:DC%2BD3MXhtlOksLg%3D
EC Burgard W Niforatos T Biesen Particlevan et al. (2000) ArticleTitleCompetitive antagonism of recombinant P2X2/3 receptors by 2′,3′-O-(2,4,6-trinitrophenyl) adenosine 5′-triphosphate (TNP-ATP) Mol Pharmacol 58 1502–1510 Occurrence Handle11093790 Occurrence Handle1:CAS:528:DC%2BD3cXovFeqsLY%3D
GY Xu LY Huang (2002) ArticleTitlePeripheral inflammation sensitizes P2X receptor-mediated responses in rat dorsal root ganglion neurons J Neurosci 22 93–102 Occurrence Handle11756492 Occurrence Handle1:CAS:528:DC%2BD38XislWnsg%3D%3D
OA Krishtal SM Marchenko VI Pidoplichko (1983) ArticleTitleReceptor for ATP in the membrane of mammalian sensory neurones Neurosci Lett 35 41–45 Occurrence Handle10.1016/0304-3940(83)90524-4 Occurrence Handle6302606 Occurrence Handle1:CAS:528:DyaL3sXht1Shur8%3D
CE Jahr TM Jessell (1983) ArticleTitleATP excites a subpopulation of rat dorsal horn neurones Nature 304 730–733 Occurrence Handle10.1038/304730a0 Occurrence Handle6888539 Occurrence Handle1:CAS:528:DyaL3sXltFKktbo%3D
CC Chen AN Akopian L Siviltotti et al. (1995) ArticleTitleA P2X purinoceptor expressed by a subset of sensory neurons Nature 377 428–431 Occurrence Handle10.1038/377428a0 Occurrence Handle7566119 Occurrence Handle1:CAS:528:DyaK2MXos1Slsr4%3D
JG Gu AB MacDermott (1997) ArticleTitleActivation of ATP P2X receptors elicits glutamate release from sensory neuron synapses Nature 389 749–753 Occurrence Handle10.1038/39639 Occurrence Handle9338789 Occurrence Handle1:CAS:528:DyaK2sXmvVSnsbs%3D
DA Cockayne SG Hamilton QM Zhu et al. (2000) ArticleTitleUrinary bladder hyporeflexia and reduced pain-related behaviour in P2X3-deficient mice Nature 407 1011–1015 Occurrence Handle10.1038/35039519 Occurrence Handle11069181 Occurrence Handle1:CAS:528:DC%2BD3cXnvVaqsLk%3D
V Souslova P Cesare D Nebenuis-Oosthuizen et al. (2000) ArticleTitleWarm-coding deficits and aberrant inflammatory pain in mice lacking P2X3 receptors Nature 407 1015–1017 Occurrence Handle10.1038/35039526 Occurrence Handle11069182 Occurrence Handle1:CAS:528:DC%2BD3cXnvVaqsbc%3D
T Nakatsuka JG Gu (2001) ArticleTitleATP P2X receptor-mediated enhancement of glutamate release and evoked EPSCs in dorsal horn neurons of the rat spinal cord J Neurosci 21 6522–6531 Occurrence Handle11517241 Occurrence Handle1:CAS:528:DC%2BD3MXmt1Wjurw%3D
T Nakatsuka H Furue M Yoshimura et al. (2002) ArticleTitleActivation of central terminal vanilloid receptor-1 receptors and alpha beta-methylene-ATP-sensitive P2X receptors reveals a converged synaptic activity onto the deep dorsal horn neurons of the spinal cord J Neurosci 22 1228–1237 Occurrence Handle11850450 Occurrence Handle1:CAS:528:DC%2BD38XhsFeksrc%3D
G Burnstock (1996) ArticleTitleA unifying purinergic hypothesis for the initiation of pain Lancet 347 1604–1605 Occurrence Handle10.1016/S0140-6736(96)91082-X Occurrence Handle8667873 Occurrence Handle1:STN:280:DyaK283ktV2nsQ%3D%3D
G Burnstock (2000) ArticleTitleP2X receptors in sensory neurones Br J Anaesth 84 476–488 Occurrence Handle10823099 Occurrence Handle1:CAS:528:DC%2BD3cXivFWkurc%3D
D Irnich DJ Tracey J Polten et al. (2002) ArticleTitleATP stimulates peripheral axons in human, rat and mouse–differential involvement of A(2B) adenosine and P2X purinergic receptors Neuroscience 110 123–129 Occurrence Handle10.1016/S0306-4522(01)00556-5 Occurrence Handle11882377 Occurrence Handle1:CAS:528:DC%2BD38XhslSkur0%3D
J Sawynok A Reid (1997) ArticleTitlePeripheral adenosine 5′-triphosphate enhances nociception in the formalin test via activation of a purinergic P2X receptor Eur J Pharmacol 330 115–121 Occurrence Handle10.1016/S0014-2999(97)01001-7 Occurrence Handle9253943 Occurrence Handle1:CAS:528:DyaK2sXkt1Sht7o%3D
E Dowd DS McQueen IP Chessell et al. (1998) ArticleTitleP2X receptor-mediated excitation of nociceptive afferents in the normal and arthritic rat knee joint Br J Pharmacol 125 341–346 Occurrence Handle10.1038/sj.bjp.0702080 Occurrence Handle9786507 Occurrence Handle1:CAS:528:DyaK1cXmtlClurg%3D
M Tsuda S Koizumi A Kita et al. (2000) ArticleTitleMechanical allodynia caused by intraplantar injection of P2X receptor agonist in rats: Involvement of heteromeric P2X2/3 receptor signaling in capsaicin-insensitive primary afferent neurons J Neurosci 20 RC90 Occurrence Handle10899177 Occurrence Handle1:STN:280:DC%2BD387os1Onuw%3D%3D
G Wynn W Rong Z Xiang et al. (2003) ArticleTitleBurnstock G. Purinergic mechanisms contribute to mechanosensory transduction in the rat colorectum Gastroenterology 125 1398–1409 Occurrence Handle10.1016/j.gastro.2003.07.008 Occurrence Handle14598256 Occurrence Handle1:CAS:528:DC%2BD3sXpslyru70%3D
HV Tempest AK Dixon WH Turner et al. (2004) ArticleTitleP2X and P2X receptor expression in human bladder urothelium and changes in interstitial cystitis BJU Int 93 1344–1348 Occurrence Handle10.1111/j.1464-410X.2004.04858.x Occurrence Handle15180635 Occurrence Handle1:CAS:528:DC%2BD2cXmt1ertLY%3D
SD Novakovic LC Kassotakis IB Oglesby et al. (1999) ArticleTitleImmunocytochemical localization of P2X3 purinoceptors in sensory neurons in naive rats and following neuropathic injury Pain 80 273–282 Occurrence Handle10.1016/S0304-3959(98)00225-5 Occurrence Handle10204740 Occurrence Handle1:CAS:528:DyaK1MXht1Oks7s%3D
K Tsuzuki E Kondo T Fukuoka et al. (2001) ArticleTitleDifferential regulation of P2X(3) mRNA expression by peripheral nerve injury in intact and injured neurons in the rat sensory ganglia Pain 91 351–360 Occurrence Handle10.1016/S0304-3959(00)00456-5 Occurrence Handle11275393 Occurrence Handle1:CAS:528:DC%2BD3MXitF2qtrg%3D
S McGaraughty CT Wismer CZ Zhu et al. (2003) ArticleTitleEffects of A-317491, a novel and selective P2X3/P2X2/3 receptor antagonist, on neuropathic, inflammatory and chemogenic nociception following intrathecal and intraplantar administration Br J Pharmacol 140 1381–1388 Occurrence Handle10.1038/sj.bjp.0705574 Occurrence Handle14623769 Occurrence Handle1:CAS:528:DC%2BD2cXhsV2rug%3D%3D
JG Gu (2003) ArticleTitleP2X receptor-mediated modulation of sensory transmission to the spinal cord dorsal horn Neuroscientist 9 370–378 Occurrence Handle10.1177/1073858403252788 Occurrence Handle14580121 Occurrence Handle1:CAS:528:DC%2BD3sXoslajsbk%3D
RE Fyffe ER Perl (1984) ArticleTitleIs ATP a central synaptic mediator for certain primary afferent fibers from mammalian skin? Proc Natl Acad Sci USA 81 6890–6893 Occurrence Handle10.1073/pnas.81.21.6890 Occurrence Handle6593733 Occurrence Handle1:CAS:528:DyaL2MXhvVaisg%3D%3D
T Nakatsuka K Tsuzuki J Ling et al. (2003) ArticleTitleDistinct roles of P2X receptors in modulating glutamate release at different primary sensory synapses in the dorsal horn of rat spinal cord J Neurophysiol 89 3243–3252 Occurrence Handle10.1152/jn.01172.2002 Occurrence Handle12783958 Occurrence Handle1:CAS:528:DC%2BD3sXlsFaju7w%3D
BA Chizh P Illes (2001) ArticleTitleP2X receptors and nociception Pharmacol Rev 53 553–568 Occurrence Handle11734618 Occurrence Handle1:CAS:528:DC%2BD38XltVOlug%3D%3D
B Rexed (1952) ArticleTitleThe cytoarchitectonic organization of the spinal cord in the rat J Comp Neurol 94 415–466 Occurrence Handle10.1002/cne.900960303
WD Willis SuffixJr RE Coggeshall (1991) Sensory Mechanisms of the Spinal Cord Plenum Press New York
F Cervero (1995) ArticleTitleWhat is a nociceptor-specific (class 3) cell? Pain 62 123–125 Occurrence Handle10.1016/0304-3959(94)00233-5 Occurrence Handle7478702 Occurrence Handle1:STN:280:DyaK28%2FnsF2hsA%3D%3D
R Dubner GJ Bennett (1983) ArticleTitleSpinal and trigeminal mechanisms of nociception Annu Rev Neurosci 6 381–18 Occurrence Handle10.1146/annurev.ne.06.030183.002121
S Mense (1993) ArticleTitleNociception from skeletal muscle in relation to clinical muscle pain Pain 54 241–289 Occurrence Handle10.1016/0304-3959(93)90027-M Occurrence Handle8233542 Occurrence Handle1:STN:280:DyaK2c%2FltFartQ%3D%3D
HG Schaible BD Grubb (1993) ArticleTitleAfferent and spinal mechanisms of joint pain Pain 55 5–54 Occurrence Handle10.1016/0304-3959(93)90183-P Occurrence Handle8278210 Occurrence Handle1:CAS:528:DyaK2cXitFertLc%3D
TJ Ness GF Gebhart (1990) ArticleTitleVisceral pain: A review of experimental studies Pain 41 167–234 Occurrence Handle10.1016/0304-3959(90)90021-5 Occurrence Handle2195438 Occurrence Handle1:STN:280:DyaK3czhtVGnug%3D%3D
L Vulchanova MS Riedl SJ Shuster et al. (1998) ArticleTitleP2X3 is expressed by DRG neurons that terminate in inner lamina. II Eur J Neurosci 10 3470–3478 Occurrence Handle10.1046/j.1460-9568.1998.00355.x Occurrence Handle9824460 Occurrence Handle1:STN:280:DyaK1M%2Fks1yntA%3D%3D
EJ Bradbury G Burnstock SB McMahon (1998) ArticleTitleThe expression of P2X3 purinoreceptors in sensory neurons: Effects of axotomy and glial-derived neurotrophic factor Mol Cell Neurosci 12 256–268 Occurrence Handle10.1006/mcne.1998.0719 Occurrence Handle9828090 Occurrence Handle1:CAS:528:DyaK1cXnsVeqtr0%3D
A Guo L Vulchanova J Wang et al. (1999) ArticleTitleImmunocytochemical localization of the vanilloid receptor 1 (VR1): Relationship to neuropeptides, the P2X3 purinoceptor and IB4 binding sites Eur J Neurosci 11 946–958 Occurrence Handle10.1046/j.1460-9568.1999.00503.x Occurrence Handle10103088 Occurrence Handle1:STN:280:DyaK1M3gtlehsw%3D%3D
L Vulchanova U Arvidsson M Riedl et al. (1996) ArticleTitleDifferential distribution of two ATP-gated channels (P2X receptors) determined by immunocytochemistry Proc Natl Acad Sci USA 93 8063–8067 Occurrence Handle10.1073/pnas.93.15.8063 Occurrence Handle8755603 Occurrence Handle1:CAS:528:DyaK28XksFSru7c%3D
L Vulchanova MS Riedl SJ Shuster et al. (1997) ArticleTitleImmunohistochemical study of the P2X2 and P2X3 receptor subunits in rat and monkey sensory neurons and their central terminals Neuropharmacology 36 1229–1242 Occurrence Handle10.1016/S0028-3908(97)00126-3 Occurrence Handle9364478 Occurrence Handle1:CAS:528:DyaK2sXnt1emu7o%3D
P Honore K Kage J Mikusa et al. (2002) ArticleTitleAnalgesic profile of intrathecal P2X(3) antisense oligonucleotide treatment in chronic inflammatory and neuropathic pain states in rats Pain 99 11–19 Occurrence Handle10.1016/S0304-3959(02)00032-5 Occurrence Handle12237180 Occurrence Handle1:CAS:528:DC%2BD38XntVWrtL4%3D
G Dorn S Patel G Wotherspoon et al. (2004) ArticleTitlesiRNA relieves chronic neuropathic pain Nucleic Acids Res 32 e49 Occurrence Handle10.1093/nar/gnh044 Occurrence Handle15026538 Occurrence Handle1:CAS:528:DC%2BD2cXisF2ksL8%3D
RA North (2004) ArticleTitleP2X3 receptors and peripheral pain mechanisms J Physiol 554 301–308 Occurrence Handle10.1113/jphysiol.2003.048587 Occurrence Handle12832496 Occurrence Handle1:CAS:528:DC%2BD2cXhtFGrtbc%3D
MS Ramer EJ Bradbury SB McMahon (2001) ArticleTitleNerve growth factor induces P2X(3) expression in sensory neurons J Neurochem 77 864–875 Occurrence Handle10.1046/j.1471-4159.2001.00288.x Occurrence Handle11331415 Occurrence Handle1:CAS:528:DC%2BD3MXjs1Wlsbg%3D
Acknowledgement
The authors thank Dr T. Nakatsuka, K. Tsuzuki, M. Chen, and other members in the laboratory for their hard work on the P2X project. This work was supported by a NIH grant NS38254 and an ONR N00014-01-1-0188 to J.G.G.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Gu, J.G., Heft, M.W. P2X receptor-mediated purinergic sensory pathways to the spinal cord dorsal horn. Purinergic Signalling 1, 11–16 (2004). https://doi.org/10.1007/s11302-004-4743-6
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11302-004-4743-6