Abstract
Climate change is affecting the composition and functioning of ecosystems across the globe. Mountain ecosystems are particularly sensitive to climate warming since their biota is generally limited by low temperatures. Cryptogams such as lichens and bryophytes are important for the biodiversity and functioning of these ecosystems, but have not often been incorporated in vegetation resurvey studies. Hence, we lack a good understanding of how vascular plants, lichens and bryophytes respond interactively to climate warming in alpine communities. Here we quantified long-term changes in species richness, cover, composition and thermophilization (i.e. the increasing dominance of warm-adapted species) of vascular plants, lichens and bryophytes on four summits at Dovrefjell, Norway. These summits are situated along an elevational gradient from the low alpine to high alpine zone and were surveyed for all species in 2001, 2008 and 2015. During the 15-year period, a decline in lichen richness and increase in bryophyte richness was detected, whereas no change in vascular plant richness was found. Dwarf-shrub abundance progressively increased at the expense of lichens, and thermophilization was most pronounced for vascular plants, but occurred only on the lowest summits and northern aspects. Lichens showed less thermophilization and, for the bryophytes, no significant thermophilization was found. Although recent climate change may have primarily caused the observed changes in vegetation, combined effects with non-climatic factors (e.g. grazing and trampling) are likely important as well. At a larger scale, alpine vegetation shifts could have a profound impact on biosphere functioning with feedbacks to the global climate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biological consequences of climate change are increasingly evident across a wide range of ecosystems. Advancing phenology as well as distribution range shifts towards higher latitudes and elevations have already been observed (Parmesan and Yohe 2003; Neilson et al. 2005; Schwartz et al. 2006; Wolkovich et al. 2012; De Frenne et al. 2013). In addition, climate change is expected to shift plant communities towards increasing dominance of warm-adapted species and loss of cold-adapted species (a process described as thermophilization; Bertrand et al. 2011; Gottfried et al. 2012; De Frenne et al. 2013).
Tundra ecosystems are considered to be particularly susceptible to global warming as they are generally limited by low temperatures (Klanderud and Birks 2003; Walker et al. 2006; Elmendorf et al. 2015). Hence, these ecosystems can also be used as indicators for the impacts of climate change (Grabherr et al. 2010; Malanson et al. 2011), and they provide an excellent base for detection of its early-warning signals (Wolf et al. 2012). Over the past decades, widespread implications of warming on tundra vegetation have been reported (Post et al. 2009; Mayor et al. 2017), including an upslope migration and increasing species richness of vascular plants (Pauli et al. 2007) and a progressive shrub expansion (Myers-Smith et al. 2011).
Current climate-impact studies in tundra environments, but also in other ecosystems, are strongly biased towards vascular plants (Corlett and Westcott 2013; He et al. 2016). Non-vascular cryptogams such as lichens and bryophytes are generally neglected or considered as a group, and data on a species level are often lacking (Jägerbrand and Alatalo 2015). At higher latitudes/elevations, however, the relative abundance of cryptogams increases significantly compared to vascular plants (Wielgolaski et al. 1981; Jägerbrand and Alatalo 2015; Mateo et al. 2016), and they often make up the majority of species and aboveground biomass (Matveyeva and Chernov 2000; Hassel et al. 2012). Hence, cryptogams play a crucial role in the functioning of tundra ecosystems. For instance, some lichen and bryophyte species (living in association with cyanobacteria) are responsible for fixing external nitrogen (N) (Solheim et al. 1996; Nash 2008). In addition, lichens are known for their key role in nutrient cycling and vascular plant seedling establishment and, along with other cryptogams, they account for considerable contributions to the global carbon (C) sink (Kershaw 1985; Malmer et al. 1994; Van Breemen 1995; Nash 2008; Lang et al. 2009; Asplund and Wardle 2016). According to Elbert et al. (2012), cryptogams take up approximately 3.9 Pg C on an annual basis, which corresponds to about 7% of the net primary productivity by terrestrial vegetation. Macrolichens also serve as an important winter staple food for grazing mammals such as reindeer or caribou (Rangifer tarandus) (Boertje 1984; Danell et al. 1994), whereas bryophytes contribute to the preservation of the permafrost layer through their temperature-isolating capacity (Porada et al. 2016).
In addition to their importance for ecosystem functioning, lichens and bryophytes also interact intensively with vascular plants in tundra communities (e.g. Longton 1988; Dorrepaal et al. 2006; Cornelissen et al. 2007; Casanova-Katny et al. 2014). For instance, vascular plants have a profound influence on cryptogam performance and abundance (Cornelissen et al. 2001; Jägerbrand et al. 2012). Vice versa, lichens and bryophytes both facilitate and compete with vascular plants, and can therefore affect natural vegetation succession (Sedia and Ehrenfeld 2003; Dorrepaal et al. 2006; Cornelissen et al. 2007). Moreover, climate warming expected to amplify the aboveground productivity of vascular plants, which could enable faster-growing plant species to outcompete lichens and bryophytes (Cornelissen et al. 2001), and may be the cause of the observed decline in cryptogam biomass and/or abundance in subarctic and mid-arctic ecosystems (Virtanen et al. 2003; Walker et al. 2006; Elmendorf et al. 2012).
Considering the importance of cryptogams in tundra ecosystems, more studies are required that include the impact of climate change on lichens and bryophytes at the species level, in relation to vascular plants. This would significantly improve our understanding of how these fragile communities as well as ecosystem functioning are affected by warming, and can in turn support the development of urgently needed conservation strategies for alpine habitats and their biota. Besides, the indicator value of tundra ecosystems could be enhanced if cryptogams are considered, given that they represent different functional groups with different microhabitat requirements and distribution patterns across elevations (Grytnes et al. 2006). Finally, also long-term monitoring studies are essential when studying climate-change impacts on tundra plant communities, as these often bear the imprint of past climatic changes (Johansen et al. 2016). Especially for lichens and bryophytes, which are generally slow-growing (Ahti 1977), long-term observation records are of major importance, but these are to date still scarce.
In order to obtain more insight into the ecological effects of global warming on high-mountain ecosystems, we performed a long-term vegetation resurvey study at Dovrefjell National Park, Norway. Vegetation was monitored on four mountain summits in 2001, 2008 and 2015. Each year, plot-scale and summit-wide surveys were conducted to investigate whether species richness, cover, community composition and thermophilization of vascular plants, lichens and bryophytes changed over time and whether these changes can be attributed to the ongoing changes in climatic conditions. To the best of our knowledge, this is also the first study to quantify thermophilization of vascular plants, lichens and bryophytes together. We specifically tested the following hypotheses: (H1) Warming of the study area contributes to increased vascular plant species richness and cover of some species, especially shrubs and dwarf shrubs, (H2) the diversity and cover of lichens and bryophytes have declined over time, and (H3) the plant community composition has shifted to more dominance of warm-adapted species (i.e. thermophilization).
Materials and methods
Study site
The study area is located in the Dovrefjell-Sunndalsfjella National Park, Norway. The plots are part of the GLORIA network (www.gloria.ac.at), and the ‘Multi-Summit Approach’ (Pauli et al. 2015) was used to select four summits, representing an elevational gradient of vegetation patterns from the treeline ecotone up to the vascular plant limit: Vesle Armodshøkollen (62°15′40.5″N, 9°39′55.4″E, 1161 m a.s.l.), Veslekolla (62°18′23.6″N, 9°27′25.2″E, 1418 m a.s.l.), Kolla (62°17′30.5″N, 9°29′12.7″E, 1651 m a.s.l.) and Storkinn (62°20′48.2″N, 9°26′27.1″E, 1845 m a.s.l.) (Fig. S1). These summits are situated in the low alpine zone, the transition zone between the low and the middle alpine zone, the transition zone between the middle and the high alpine zone, and the high alpine zone, respectively.
The 1418 m-site and 1845 m-site are located on strongly metamorphosed sandstone. The 1651 m-site predominantly consist of gneiss, granite and anorthosite, and the 1161 m-site is composed of greenstone and amphibolite (Bretten et al. 1994; Syverhuset, 2009; Geological Survey of Norway 2015). The vegetation on the summits mainly consists of plant communities belonging to the Loiseleurio-Arctostaphylio alliance (on the exposed ridges), the Phyllodoco-Vaccinion and Nardo-Carcion bigelowii alliance (on the lee sides of the summits) and the Cassiopo-Salicion herbaceae alliance (in the late-melting snow beds) (Elven et al. 1996).
Vegetation surveys
For each summit, a standard procedure was followed to set up the equipment and establish the monitoring site (cf. Pauli et al. 2015; see also ESM 1 in Electronic Supplementary Material [ESM]). In summary, each summit was divided into eight summit area sections: two (an upper and a lower) for each slope direction (aspect; corresponding to the four main cardinal directions). The upper sections were situated between the highest summit point (HSP) and 5-m contour line, while the lower sections were installed between the 5-m and 10-m contour line. Finally, four 3 × 3 m2 quadrat clusters were positioned on each summit, one in every upper section, at 5 height meters below the HSP. These clusters consisted of nine 1-m2 quadrats, whereof the four corner quadrats were inventoried for occurrence and visual cover of all vascular plant, lichen and bryophyte species, measured as a percentage of the quadrat covered. In addition to the detailed vegetation analysis in the 1-m2 quadrats, all vascular plant species were inventoried in each summit area section, and their percentage cover was estimated visually using five abundance classes (Table S1 in ESM).
Macroclimate and microclimate
The macroclimate temperature and precipitation data for the period 1900–2014 were obtained from Fokstugu weather station (data available at http://www.yr.no), which is located at 973 m a.s.l. and about 23 km southwest of the study area. To quantify the microclimate on each study summit and each aspect, the soil temperature was recorded at an hourly basis between 2001 and 2015, using miniature data loggers (GEO-Precision M-Log5 W logger). Each summit contained four temperature loggers, one in every middle quadrat of the 3 × 3 m2 quadrat clusters for each cardinal direction and buried in the substrate at a depth of about 10 cm below the soil surface.
Data analyses
All statistical analyses were performed in R version 3.2.2 (R Core Team 2015). Main effects were tested using a significance level of 5%, while a significance level of 1% was used for interaction effects. Temporal changes in the macroclimate air temperature and precipitation were analysed with linear regression, both for the period 1900–2014 and 2001–2014. To test whether the microclimate soil temperature of all study summits combined changed significantly over time during the periods 2001–2008, 2008–2015 and 2001–2015, two linear mixed-effect models (LMM) were composed for each period using the nlme-package (Pinheiro et al. 2017). In the first model, ‘summit’ and ‘aspect’ (nested within ‘summit’) were defined as fixed effects, and the mean annual soil temperature as response variable. A random effect term ‘sensor’ was also included to account for temporal autocorrelation in the temperature measurements of the same data loggers. In the second model, a continuous variable ‘year’ was added to the fixed effects. The two models were subsequently compared with a χ 2-test. This procedure was repeated for the annual mean summer (June, July, August) and winter (December, January, February) soil temperature at the study summits. The soil temperature data of the different summits and aspects were also compared in a pairwise way with a Tukey multiple comparison test using the multcomp-package (Torsten et al. 2008).
Next, a LMM was used to investigate whether the cover percentage of individual species and growth forms (i.e. dwarf shrubs, forbs, graminoids, lichens and bryophytes) changed significantly in the quadrats of the four summits between 2001 and 2015. In this model, ‘year of sampling’ was included as a fixed effect, whereas ‘summit’ and ‘aspect’ (nested within ‘summit’) were defined as random factors to account for the hierarchical structure of the data and spatial autocorrelation between populations of the same summit or mountainside. A similar LMM was used to investigate the relationship between the cover changes of lichens and dwarf shrubs in the quadrats. Here we defined the change in lichen cover between 2001 and 2015 as response variable, while the corresponding change in dwarf-shrub cover was defined as a fixed effect. ‘Summit’ and ‘aspect’ (nested within ‘summit’) were again included as random factor.
To test how the species richness of vascular plants, lichens and bryophytes changed over time, an ANOVA test was performed with ‘summit’, ‘aspect’ (nested within ‘summit’), ‘year of sampling’ and their interactions as fixed effects, and the number of species per quadrat of each organism group as response variable. In addition, dissimilarity indices were calculated for vascular plants, lichens and bryophytes, and evaluated for the periods 2001–2008, 2008–2015 and 2001–2015, to quantify the corresponding β-diversity. We used the modified Gower dissimilarity index (Gower 1987), which quantifies both species turnover and nestedness, and can be interpreted as the average change in orders of magnitude per species between two samples of the same plot at different points in time (Anderson et al. 2006). Differences in dissimilarity among the summits and aspects were assessed for each period with ANOVA, specifying ‘summit’ and ‘aspect’ (nested within ‘summit’) as a fixed effects and the dissimilarity index as response variable. Both species richness and dissimilarity indices were calculated with the vegan-package (Oksanen et al. 2017).
Finally, in order to assess the effect of climate warming on the composition of alpine plant, lichen and bryophyte communities at Dovrefjell, thermophilization was quantified. This takes into account the realized position of a plant species along an environmental gradient and quantifies thus the increasing dominance of warmth-loving species. Here the difference in elevation was considered to consistently represent a thermal gradient. All recorded plant species in the quadrats of the summits received an elevational rank (Table S2 in ESM), which was originally based on the species’ lower and upper margin as well as its distribution centre. For vascular plants, these ranks were derived from Gottfried et al. (2012), while for lichens and bryophytes, these are based on own compiled expert data and local floras (Nyholm 1969; Damsholt 2002; Hallingbäck et al. 2006, 2008; Thell and Moberg 2011; Ahti et al. 2013; Hedenäs et al. 2014). All ranks are mentioned in Tables S3–S5 in ESM. Subsequently, the thermic vegetation indicator (S) per quadrat and survey year (i.e. 2001, 2008 and 2015) was calculated as the community-weighted mean of the species’ elevational ranks, weighed by their cover values. This was determined for vascular plants, lichens and bryophytes separately. The thermophilization indicator (D) was then calculated for each time period (i.e. 2001–2008, 2008–2015 and 2001–2015) as the shift over time (recent minus old survey, positive values thus denote an increasing thermophilization) in the thermic vegetation indicator (following Gottfried et al. 2012). A Wilcoxon singed rank test was used to test whether the thermophilization indicators differed significantly from zero, while the effects of ‘summit’ and ‘aspect’ (nested within the variable ‘summit’) were investigated with ANOVA as described above. Due to uncertain identification of some of the bryophyte species in 2008 (as, for instance, indicated by the total number of species, see Discussion), we decided not to include the thermophilization indicators of bryophytes for the periods involving the year 2008.
Results
Macroclimate and microclimate
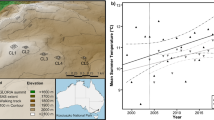
The macroclimate data showed that the mean annual temperature was −0.1 °C for the reference period 1961–1990 (Fig. 1a). Mean July temperatures were 9.8 °C, while mean January temperatures were −8.8 °C (Fig. S2 in ESM). The mean annual temperature increased significantly between 1900 and 2014 (Linear regression, F = 8.96, df = 1, 113, P = 0.003, slope = 0.00759 °C year−1) (Fig. 1a). Yet, for the period 2001–2014, no significant change in the mean annual temperature was found (Linear regression, F = 0.075, df = 1, 13, P = 0.789, slope = −0.016 °C year−1) (Fig. 1b). In addition, no significant change was detected for the growing season (average of July) temperature during period 1900–2014 (Linear regression, F = 0.981, df = 1, 113, P = 0.324, slope = 0.00392 °C year−1), nor during the period 2001–2014 (Linear regression, F = 1.15, df = 1, 13, P = 0.303, slope = 0.126 °C year−1). The mean annual precipitation at Fokstugu amounted to 435 mm between 1961 and 1990 (Norwegian Meteorological Institute, 2015) and increased significantly between 1900 and 2014 (Linear regression, F = 17, df = 1, 113, P < 0.001, slope = 0.157% year−1) (Fig. 1c). Especially in spring (March, April, May) (Linear regression, F = 6.83, df = 1, 113, P = 0.010, slope = 0.188% year−1) and autumn (September, October, November) (Linear regression, F = 9.02, df = 1, 113, P = 0.003, slope = 0.208% year−1), significant increases in the mean precipitation were found (Fig. S3 in ESM).
Macroclimate and microclimate of the study area. a Anomaly of the mean annual temperature relative to the baseline period of 1961–1990 at the weather station of Fokstugu between 1900 and 2014. b Mean annual (black), annual mean summer (June, July, August; dark grey) and annual mean winter (December, January, February; light grey) temperature at the weather station of Fokstugu between 2001 and 2014. c Percentage anomaly of the mean annual precipitation relative to the baseline period of 1961–1990 at the weather station of Fokstugu between 1900 and 2014. d Mean annual (black), annual mean summer (June, July, August; dark grey) and annual mean winter (December, January, February; light grey) soil temperatures averaged for all temperature loggers in our plots throughout the period 2001–2015. The dashed lines show the long-term linear trend, whereas the dark grey lines in (a) and (c) represent a 10-year running average
In spite of the long-term increase in macroclimate air temperatures, the microclimate data showed a surprising significant decrease in the mean annual soil temperature across the four summits during the period 2001–2015 (χ 2-test, df = 1, χ 2 = 25.9, P < 0.001) (Fig. 1d). However, we noticed a significant increase in mean annual soil temperature between 2001 and 2008 (χ 2-test, df = 1, χ 2 = 8.22, P = 0.004) and a significant decrease between 2008 and 2015 (χ 2-test, df = 1, χ 2 = 10.9, P < 0.001). Significant differences in the mean soil temperature were noticeable between the different summits and between the southern vs. northern and southern vs. western aspects (Fig. S4–S7, Table S6 in ESM). For the annual mean summer temperature, a marginally significant negative trend was observed between 2001 and 2015 (χ 2-test, df = 1, χ 2 = 3.75, P = 0.053), whereas for the annual mean winter temperature no significant change was found (χ 2-test, df = 1, χ 2 = 1.61, P = 0.204) (Fig. 1d).
Total number of species and response of individual species
The total number of vascular plant species of all summit areas combined increased from 67 to 73 between 2001 and 2015. For the lichens, a decrease in the total species number of the quadrats was found from 39 to 33, whereas for the bryophytes, a steady-state situation of 20 species was observed between 2001 and 2015. Note that a significantly lower total amount of bryophyte species was recorded in 2008 (Table S7 in ESM).
The vascular plant species Comastoma tenellum, Corallorhiza trifida, Erigeron sp., Geranium sylvaticum, Luzula confusa, Silene acaulis and Pinus sylvestris were recorded for the first time in the summit area sections of our study summits in 2015. However, Draba fladnizensis, Euphrasia sp., Poa arctica, Salix lapponum and Silene uralensis disappeared from the summits.
The total cover of dwarf shrubs, forbs and graminoids in the quadrats increased significantly between 2001 and 2015, from 9.72 to 18.2% (LMM, df = 1, 175, F = 9.92, P = 0.001), 0.099 to 0.296% (LMM, df = 1, 175, F = 4.37, P = 0.0305) and 0.339 to 1.31% (LMM, df = 1, 175, F = 32.1, P < 0.001), respectively. The total cover of lichens in the quadrats, however, decreased significantly over time from 24.1 to 17.7% (LMM, df = 1, 175, F = 5.56, P = 0.0134). The total cover of bryophytes did not change over time (decreased from 2.32 to 1.93%, but LMM, df = 1, 175, F = 0.216, P = 0.612) (Fig. 2a). Moreover, we found a significant negative correlation between the cover changes of lichens and dwarf shrubs in the quadrats between 2001 and 2015 (LMM, df = 1, 175, F = 43.9, P < 0.001) (Fig. 2b).
Temporal cover changes. a Percentage cover of dwarf shrubs (S), forbs (F), graminoids (G), lichens (L) and bryophytes (B) in all 1-m2 quadrats of the four study summits combined for 2001, 2008 and 2015. Results are shown as mean ± standard error of mean (SEM). Different letters indicate significant differences among the sampling years (Linear mixed-effect model [LMM], P < 0.05). b Relationship between the cover changes of lichens and dwarf shrubs in the 1-m2 quadrats of the four study summits between 2001 and 2015 (LMM, P < 0.001, pseudo-R 2 = 0.747)
Four dwarf shrub species (Betula nana, Empetrum nigrum, Vaccinium uliginosum and Vaccinium vitis-idaea) and three graminoid species (Festuca ovina, Juncus trifidus and Luzula arcuata) showed a significant cover increase over time in the quadrats. For lichen species, a decreasing cover percentage was observed for Allantoparmelia alpicola, Cladonia arbuscula, Cladonia bellidiflora, Cladonia macrophylla, Cladonia pleurota, Coelocaulon aculeatum, Ochrolechia sp., Pseudephebe sp. and Umbilicaria sp., whereas an increasing cover was found for Bryocaulon divergens, Sphaerophorus globulus and Thamnolia vermicularis. The bryophyte species Anthelia sp., Lophozia cf. ventricosa, Pogonatum sp., Pohlia sp. and Polytrichum juniperinum showed an increasing cover (Table 1).
α-diversity: vascular plants in quadrats
For the vascular plants in the quadrats, no significant change in the number of species was found between 2001 and 2015. The vascular plant richness differed, however, significantly between the summits and the aspects of each summit (Fig. 3a and Fig. S8, Table S8 in ESM). For instance, Fig. 3a shows that the mean number of species decreased with elevation, ranging from about 7.85 ± 0.34 (mean ± standard error of mean [SEM]) species per 1-m2 quadrat on the lowest summit to only 1.11 ± 0.11 species per 1-m2 quadrat on the highest summit. Yet, no significant summit by year interaction was found.
Temporal changes in vegetation per elevation (m). Species richness for vascular plants (a), lichens (b) and bryophytes (c) in all 1-m2 quadrats at the different elevations for 2001, 2008 and 2015. Modified Gower dissimilarity index for vascular plants (d), lichens (e) and bryophytes (f) in all 1-m2 quadrats at the different elevations between 2001–2008, 2008–2015 and 2001–2015. Thermophilization indicator for vascular plants (g), lichens (h) and bryophytes (i) in all 1-m2 quadrats at the different elevations between 2001–2008, 2008–2015 and 2001–2015. For the bryophytes, we decided not to include the thermophilization indicators for the periods involving the year 2008 due to uncertain identification of some species. Results are shown as mean ± standard error of mean (SEM). Different letters indicate significant differences among the sampling years/periods (ANOVA, P < 0.05)
α-diversity: lichens and bryophytes in quadrats
For the lichen richness, we observed a significant decrease over time. Lichen richness also differed significantly between the summits and the aspects of each summit, but no significant summit by year interaction was found (Fig. 3b and Fig. S8, Table S8 in ESM). Figure 3b, for instance, shows a higher number of lichen species on the summits of intermediate elevations. For the bryophytes, a significant increase in the number of species over time was observed. Bryophyte richness also differed significantly between the summits and aspects, but again no significant summit by year interaction was detected (Fig. 3c and Fig. S8, Table S8 in ESM).
β-diversity: modified Gower dissimilarity indices for the quadrats
The modified Gower dissimilarity indices of vascular plants decreased with increasing elevation in all three time periods. However, only for the period 2008–2015, a significant effect of aspect was observed (Fig. 3d and Fig. S8, Table S9 in ESM). Also for the lichens, a significant decrease in dissimilarity with elevation was found, but the effect of aspect was only significant for 2001–2008 (Fig. 3e and Fig. S8, Table S9 in ESM). For the bryophytes, a significantly higher dissimilarity was observed at the 1418 m-site during the periods 2001–2008 and 2008–2015. The effect of aspect was only significant for 2001–2008 and marginally significant for 2001–2015 (Fig. 3f and Fig. S8, Table S9 in ESM).
Thermophilization
Significantly positive thermophilization of vascular plants was found at the 1161 m-site (Wilcoxon signed rank test, P = 0.006) and the 1418 m-site (Wilcoxon signed rank test, P = 0.025) between 2001 and 2015, while the thermophilization at the 1651 m-site and 1845 m-site was negligibly small (Fig. 3g). Moreover, a significant effect of summit and aspect on the thermophilization was noticed for the periods 2001–2008 and 2001–2015, with especially higher values on the lower summits and northern aspects. For the period 2008–2015, however, we found no significant effect of the summit and only a marginally significant effect of the aspect (Fig. 3g and Fig. S8, Table S10 in ESM).
For the thermophilization of lichens, positive values were detected for 2008–2015 (Wilcoxon signed rank test, P = 0.002), while negative values were found for 2001–2008 (Wilcoxon signed rank test, P < 0.001). No significant effects of elevations were found, except for the period 2008–2015, where the thermophilization increased with elevation. No significant effects of the aspect were observed for the three periods (Fig. 3h and Fig. S8, Table S10 in ESM). Finally, the thermophilization of bryophytes was significantly positive for the 1451 m-site (Wilcoxon signed rank test, P = 0.005), but showed no significant differences between the other summits and aspects (Fig. 3i and Fig. S8, Table S10 in ESM).
Discussion
Temporal changes in climatic conditions
Air temperatures at Fokstugu weather station increased significantly by 0.87 °C between 1900 and 2014. However, over the past 15 years, the rate of temperature increase at Fokstugu apparently slowed down, and no significant change in air temperature was found for that period. This short-term slowdown in the warming trend (the so-called global warming hiatus between 2000 and 2013) was observed globally, and can primarily be attributed to internal variability associated with El Niño Southern Oscillation (ENSO) (Kosaka and Xie 2013). In the coming decades, significant warming trends will likely continue due to the dominant long-term warming effect of atmospheric greenhouse gases (Schmidt et al. 2014). Precipitation at Fokstugu increased significantly by 18% over the period 1900–2014, especially in spring and autumn. These long-term climatic changes in the study area could have caused significant changes in vegetation patterns and community dynamics (Theurillat and Guisan 2001).
The mean annual soil temperature in our plots increased significantly between 2001 and 2008, whereas a significant decrease was observed between 2008 and 2015. Overall, a small decrease in the mean annual soil temperature was noticed between 2001 and 2015. This observation is consistent with the macroclimate temperature record in this period. Alternatively, this cooling trend observed through our soil temperature measurements might be related to the significant increase in dwarf-shrub cover, since shrub encroachment is known to induce shading and soil cooling in summer (Myers-Smith et al. 2011), although similar trends were also observed on the higher summits where dwarf-shrub cover is generally low or absent.
Changes in species richness with elevation
Vascular plant species richness decreased significantly towards higher elevations (see also Bruun et al. 2006; Grytnes et al. 2006; McCain and Grytnes 2010). This is most likely a consequence of the increasingly harsh environmental conditions on the higher summits (for instance, demonstrated by the difference in soil temperature of about −3.5 °C between the lowest and highest study summit). Yet, another possible explanation for the higher species richness on the lowest summit could be the difference in bedrock composition. The 1161 m-site mainly consists of calcareous bedrock (Michelsen et al. 2011), which is known to be high in nutrients and could, therefore, support a higher productivity and species richness of vascular plants (Rey Benayas and Scheiner 2002; Virtanen et al. 2003). Lichen species richness showed no clear trend along the elevational gradient. This is in line with Grytnes et al. (2006), who also failed to find a trend in lichen species richness above the treeline in western Norway. For the bryophytes, a slight increase in species richness with elevation was found. This corresponds with Bruun et al. (2006), who also noticed an increasing species richness of bryophytes and liverworts along an elevational gradient in northern Fennoscandia.
Temporal changes in species richness and abundance in the quadrats
Vascular plants
Even though the total number of vascular plant species increased on our study summits between 2001 and 2015, no significant change in vascular plant richness was found in the quadrats. Most probably, this can be explained by the absence of a significant warming trend in our study area during that period, in combination with the exceptionally cold spring of 2015. Nevertheless, Michelsen et al. (2011) found a significant increase of vascular plant species richness on the lowest summit at Dovrefjell between 2001 and 2008. This observation was consistent with previous studies, indicating an upward distribution shift and increased species richness of alpine vegetation in several Scandinavian (e.g. Klanderud and Birks 2003; Kullman 2007; Felde et al. 2012; Kapfer et al. 2012) as well as other European mountain areas (e.g. Grabherr et al. 1994; Pauli et al. 2007; Parolo and Rossi 2008; Holzinger et al. 2008; Lenoir et al. 2008; Pauli et al. 2012). For instance, Holzinger et al. (2008) found an increase in vascular plant richness of about 11% per decade in the alpine-nival ecotone of several summits in the Alps over 120 years. Significant changes in species richness were also observed over shorter periods: Pauli et al. (2007) reported that the mean number of species per square meter increased by 11.8% at Mount Schrankogel, Austria, during the period 1994–2004.
We observed a significant increase in the cover of some vascular plant species at a relatively constant rate between 2001 and 2015, especially for dwarf shrubs (B. nana, E. nigrum, V. uliginosum and V. vitis-idaea) and graminoids (F. ovina, J. trifidus and L. arcuata), but almost exclusively on the lowest summit. Encroachment by shrubs is noticed throughout many alpine and (sub)arctic tundra ecosystems (e.g. Klanderud and Birks 2003; Elmendorf et al. 2012; Grytnes et al. 2014), and can be attributed to the warming-induced increase in the length of the snow-free period and the length (and temperature) of the growing season, supporting colonization into former snow-bed communities. Klanderud and Birks (2003), for instance, found a temporal increase in the cover of dwarf shrubs and species typical of exposed ridges (e.g. J. trifidus) in Jotunheimen, Norway. The woody species B. nana showed the most substantial change in cover percentage at Dovrefjell, with an increase of about +3% over the 15-year period (more specifically, the species increased by +1.7% in 2001–2008 and +1.3% in 2008–2015). This rate is very similar to the one reported by Cannone et al. (2007), who found an average increase in the cover of shrubs and dwarf shrubs of approximately 1.9% per decade in the Alps, with the highest expansion rates in the alpine vegetation belt.
Although summer warming was not observed on our study summits during the 15-year sampling period, we argue that the observed increase in shrub abundance is likely a long-lasting response to climatic changes in the past two decades, and can therefore mainly be attributed to the long-term warming trend. This hypothesis is supported by several authors (e.g. Körner 2003; de Witte and Stöcklin 2010; Gottfried et al. 2012), who indicated that climate-induced responses of alpine plants are mainly the result of long-term climatic changes, rather than short-term oscillations. At a larger scale, shrub encroachment could change the entire ecosystem structure and alter nutrient cycling, energy fluxes, microclimate, snow cover patterns, ecological interactions and ecosystem services (Tylianakis et al. 2008; Myers-Smith et al. 2011). These changes can in turn either mitigate (e.g. through increased shading and cooling effects) or amplify (e.g. by reducing the albedo effect, leading to higher air temperatures and a more rapid snowmelt) warming in the tundra (Chapin et al. 2005; Wookey et al. 2009).
Lichens and bryophytes
For the lichens, we noticed a significant decrease in species richness and cover over time (see also Chapin et al. 1995; Cornelissen et al. 2001; Virtanen et al. 2003; Elmendorf et al. 2012). This change can likely be attributed to the increased competition with vascular plants, especially shrubs and dwarf shrubs (for instance, indicated by the highly significant negative correlation between dwarf-shrub and lichen abundance). More specifically, the increase in vascular plant biomass and canopy cover is expected to reduce light penetration to the ground surface, allowing a competition-driven loss of shade-intolerant lichen species in the longer run (Cornelissen et al. 2001; Virtanen et al. 2003; Walker et al. 2006). In addition to shade, increased litter production from vascular plants could have an adverse impact on lichens as well (Cornelissen et al. 2001). For instance, according to Chapin et al. (1995), the observed decline in lichen species at Toolik Lake, Alaska, could be attributed to increased amounts of B. nana litter. Particularly on the lower summits, where the shrub expansion was most apparent, severe reductions in lichen richness and abundance were observed. In these milder ecosystems, indirect biotic factors such as competition and facilitation might play a more decisive role, whereas, at higher elevations, lichen survival is most likely controlled by direct macroclimatic factors (Cornelissen et al. 2001).
For the bryophytes, an increase in species richness and cover was found between 2001 and 2015, especially on the lower summits. Previous studies found contrasting effects of climate change on bryophytes in tundra ecosystems (e.g. Klanderud and Totland 2005; Hassel et al. 2012; Jägerbrand et al. 2012; Lang et al. 2012). This was largely attributed to an inconsistent response of bryophytes to shading effects (Alatalo 1998; Marschall and Proctor 2004; Jägerbrand and During 2005). According to Hassel et al. (2012) and Jägerbrand et al. (2012), however, bryophyte cover and species richness will potentially decline in a warmer climate due to increased shading and litter production by vascular plants. Given the sharp increase in dwarf-shrub cover, we expected a similar decrease in bryophyte abundance on our lower study summits. Nevertheless, we argue that the increased levels of precipitation (see Fig. 1c) could have favoured bryophytes and vascular plants over lichens. To illustrate, lichens are much more abundant in the drier Norwegian inlands compared to the more humid coastlines, whereas the opposite pattern is observed for bryophyte species (Artskart 2017).
Lichens, but also bryophytes, play a pivotal role in nutrient cycling of tundra ecosystems and act both as insulators and filters of water and nutrients (Nash 2008; Porada et al. 2016). A progressive loss of lichen species could, therefore, lead to a significant alteration of the soil temperature and moisture regime, active layer depth and soil decomposition, which could in turn cause an extensive thawing of permafrost and release of soil carbon with several positive feedbacks to climate warming (Rinke et al. 2008; Schuur et al. 2015). Moreover, as an important winter forage, a reduction in the surface coverage of lichens could adversely affect local reindeer and caribou populations (Joly et al. 2009).
Thermophilization
Vascular plants
Our results indicate the occurrence of thermophilization, albeit mainly for vascular plants on the lower summits between 2001 and 2008. This observation is in line with Gottfried et al. (2012), reporting a thermophilization of summit vegetation across Europe, and can likely be attributed to the increase in soil temperature at our plot sites during that period as well as the years before sampling. The fact that the thermophilization primarily occurred on the lower summits, could be related to the larger species cover changes as well as the more prominent invasion of lowland-species in the low-alpine zone (cf. Alexander et al. 2016). This is also supported by the larger dissimilarity indices of vascular plants on the lower summits, indicating a higher species turnover. Yet, the nutrient-rich soils of the lowest summit could also partly explain the larger shift in species composition, compared to the higher summits with relatively infertile soils. Grime et al. (2008) showed that plant communities on low-fertile soils can prove more resistance to climate change than those on more fertile soils. Furthermore, the thermophilization of vascular plants was more pronounced on the northern aspects. Northern-facing slopes generally contain more cold-adapted species due to lower daily temperatures. Hence, the potential for thermophilization effects may be enhanced when these cold-adapted species are gradually outcompeted by thermophilic species, though colonisations did not occur more common at northern than at other aspects on boreal summits (Winkler et al. 2016).
At a larger spatiotemporal scale, this transformation of alpine vegetation could be associated with a progressive decline of cold-mountain habitat biota (Gottfried et al. 2012). For instance, a considerable decline in cold-adapted alpine plant species has already been noticed at their lower margins in the Alps (Pauli et al. 2007). Moreover, Arctic and alpine ecosystems generally comprise a broad spectrum of plant growth forms, and changes in the dominance among these forms could induce a substantial change in the structural characteristics and functional properties of these ecosystems (Wookey et al. 2009).
Lichens and bryophytes
Lichens showed less thermophilization than vascular plants, even though cryptogams commonly have a higher dispersal capacity (Lenoir et al. 2012) which, in turn, would improve their ability to track the upwards shifting isotherms (Fuller et al. 2015). Hence, lichen species would likely experience a greater upward distribution shift than vascular plants, which might enhance thermophilization. However, we speculate that an increased competition with vascular plants and/or bryophytes could have counteracted the upward shift and increasing dominance of thermophilic lichen species on the lower study summits. Moreover, lichens are marked by slow growth rates and thus natural succession (Cornelissen et al. 2001), and might show a delayed response to changing environmental conditions. Therefore, a possible lag effect in the response of lichens to increasing temperatures from a previous period (e.g. warming in the years prior to the surveys; cf. Dullinger et al. 2012; Gottfried et al. 2012) cannot be ruled out. Finally, for the bryophytes, no thermophilization was noticed on the 1161 m-site and 1651 m-site between 2001 and 2015. Most probably, this can be attributed to the low turnover of bryophyte species during the sampling period. On the 1418 m-site, however, a somewhat higher turnover of bryophytes was observed, which could explain the positive thermophilization.
Another important reason for the lacking thermophilization of lichens and bryophytes could be their wider elevational distribution ranges, compared to most vascular species (Vittoz et al. 2010a), with a lower percentage of exclusively alpine to high alpine species (cf. Tables S3–S5). Due to the lower degree of thermally distinct niches of cryptogam species, detection of thermophilization is more difficult. Moreover, a designation of elevational ranks for lichens and bryophytes was not possible in a number of cases, since taxa could only be identified to the genus level. Hence, no weight of their thermal preferences could be included in the analysis, which also weakens the validity of the respective thermophilization results. In general, however, cryptogams functionally deviate from vascular plants in occupying different habitat niches, where light-limitation may be a prevailing factor for lichens near the treeline, and suitable microhabitats are presumably more important for bryophytes than thermally defined elevation belts (Bruun et al. 2006; Grytnes et al. 2006).
Other possible drivers of changes in vegetation
Grazing by sheep and reindeer is increasing throughout Norway and the number of herbivores increased by approximately 6% in Dovrefjell during the last decades (Syverhuset 2009). Grazing has the potential to buffer climate-induced changes in plant diversity as it can hamper the increase in species richness and abundance at high elevations (Pajunen et al. 2012; Speed et al. 2013; Barrio et al 2016). The absence of an increasing trend in vascular plant species richness on our study summits could, therefore, also be related to the increasing number of grazing mammals. Olofsson et al. (2009) found that herbivory adversely affected the cover of shrubs like B. nana at several tundra sites in northern Fennoscandia. Given the significant increase of dwarf-shrub cover in our study area, it seems unlikely that grazing had a similar buffering effect on the changes in vegetation. Nevertheless, Vistnes and Nellemann (2008) reported that some graminoid species (e.g. J. trifidus) positively responded to a higher grazing intensity and sometimes even established at heavily grazed sites. The increase in herbivores, especially musk oxen, in the western part of the study site might therefore also have caused the significant increase in cover of J. trifidus at the 1418 m-site. Furthermore, grazing and trampling by reindeer generally leads to a decline in lichen abundance (Helle and Aspi 1983; Jägerbrand and Alatalo 2015), and could partly explain the significant decrease in lichen diversity at Dovrefjell. Bryophytes, however, tend to benefit from grazing (Väre et al. 1996; Austrheim and Eriksson 2001; Takala et al. 2014). For instance, Helle and Aspi (1983) observed an increase in small-bodied mosses (e.g. Polytrichum sp.) with heavy grazing in Finnish heathlands. In addition, Takala et al. (2014) reported that grazing promoted bryophyte species richness in Finnish semi-natural grasslands, which might also have been the case at Dovrefjell.
Potential observer bias
Resurveys are essential when studying shifts in community composition over time, even though they often involve sources of unwanted variability (Verheyen et al. 2017). We therefore delimited the permanently marked plots during the resurvey campaigns. Sources of variability arising from deviating recording seasons (Van Calster et al. 2008) and possible impacts caused by trampling effects from the observers (Semboli et al. 2014) should have been low in our study, as we carefully followed the standard protocol, attempted to avoid disturbance in and around the permanent plots, and recording was always conducted during the mid-growing season, when the majority of species should be identifiable. We always worked in teams of three observers, with partly the same observers throughout all field campaigns. This has facilitated a consistent procedure, also concerning species identification and use of plant names, which could be an important source of bias (Jansen and Dengler 2010). Nevertheless, closely related species may always be confused (Gray and Azuma 2005), thus we critically checked potential cases. Furthermore, problems with species identification and detectability are generally more common for cryptogams than for vascular plants (Archaux et al. 2008; Vittoz et al. 2010b). This was also obvious in our study, where a significantly lower number of bryophyte species was recorded in 2008, compared to the other two sampling years and almost 30% of both bryophyte and lichen taxa only could be identified to the genus level.
Pseudo-changes may also arise from observer variation during species recording and cover estimation (Vittoz and Guisan 2007; Milberg et al. 2008). Moreover, rare and small individuals, especially seedlings, are more easily overlooked or misidentified. Thus, cover estimation may be less consistent compared to more abundant species (Klimeš et al. 2001; Vittoz and Guisan 2007), and temporal cover changes should be treated with care for species having a low cover. A constant plot size, their delimitation with measuring tapes and the occasional use of a transparent template showing the area of small cover percentages, were strongly facilitating cover estimation in our study. Combined with our precautions to minimize bias in change detection, we assume that the observed significant temporal changes in community composition and species cover were not substantially caused by observational errors.
Conclusions
Vascular plants, lichens and bryophytes showed different response patterns to climatic changes as well as other biotic and abiotic changes. Even though warming in our plots was only observed between 2001 and 2008, but not between 2008 and 2015, we conclude that most of the observed changes in vegetation at Dovrefjell are consistent with long-term climate warming and confirm previous experimental and/or field studies. Yet, since this is an observational study, other external factors (e.g. grazing and trampling) cannot be ruled out. Most likely, the recorded changes in species richness and composition are a combination of recent climate change and plant-herbivore interactions. An exception, however, is the increase in dwarf-shrub abundance, which could be primarily attributed to long-lasting climate change effects. At a larger spatiotemporal scale, progressive changes in the vegetation of alpine environments can have a significant impact on biosphere functioning (e.g. carbon sequestration) with several feedbacks to the global climate.
Change history
29 March 2018
The article, Impact of climate change on alpine vegetation of mountain summits in Norway.
References
Ahti T (1977) Lichens of the boreal coniferous zone. In: Seaward MRD (ed) Lichen ecology. Academic Press, London, pp 145–181
Ahti T, Stenroos S, Moberg R (2013) Parmeliaceae. In: Ahti T, Stenroos S, Moberg R (eds) Nodric lichen flora, museum of evolution. Uppsala University, Uppsala
Alatalo J (1998) Climate change: impacts on structure and biodiversity of subarctic plant communities. Botanical Institute, Systematic Botany. Göteborg University, Göteborg
Alexander JM, Lembrechts JJ, Cavieres LA, Daehler C, Haider S, Kueffer C, Liu G, McDougall K, Milbau A, Pauchard A, Rew LJ, Seipel T (2016) Plant invasions into mountains and alpine ecosystems: current status and future challenges. Alpine Bot 126:89–103. doi:10.1007/s00035-016-0172-8
Anderson MJ, Ellingsen KE, McArdle BH (2006) Multivariate dispersion as a measure of beta diversity. Ecol Lett 9:683–693. doi:10.1111/j.1461-0248.2006.00926.x
Archaux F, Camaret S, Dupouey J-L, Ulrich E, Corcket E, Bourjot L, Brêthes A, Chevalier R, Dobremez J-F, Dumas Y, Dumé G, Forêt M, Forgeard F, Lebret Gallet M, Picard J-F, Richard F, Savoie J-M, Seytre L, Timbal J, Touffet J (2008) Can we reliably estimate species richness with large plots? An assessment through calibration training. Plant Ecol 203:303. doi:10.1007/s11258-008-9551-6
Artskart (2017). Artsdatabanken—a species map service 1.6. https://artskart.artsdatabanken.no/. Accessed 14 Feb 2017
Asplund J, Wardle DA (2016) How lichens impact on terrestrial community and ecosystem properties. Biol Rev. doi:10.1111/brv.12305
Austrheim G, Eriksson O (2001) Plant species diversity and grazing in the Scandinavian mountains: patterns and processes at different spatial scales. Ecography 24:683–695. doi:10.1111/j.1600-0587.2001.tb00530.x
Barrio IC, Hik DS, Jónsdóttir IS, Bueno CG, Mörsdorf MA, Ravolainen VT (2016) Herbivory network: an international, collaborative effort to study herbivory in Arctic and alpine ecosystems. Polar Sci 10:297–302. doi:10.1016/j.polar.2016.03.001
Bertrand R, Lenoir J, Piedallu C, Riofrio-Dillon G, de Ruffray P, Vidal C, Pierrat J-C, Gégout J-C (2011) Changes in plant community composition lag behind climate warming in lowland forests. Nature 479:215–520
Boertje RD (1984) Seasonal diets of the Denali caribou herd, Alaska. Arctic 37:161–165. doi:10.14430/arctic2182
Bretten S, Gjershaug JO, Gjærevoll O, Haugland K, Sande J, Skogland T, Sollid JL, Stabell E, Stenvik LF, Sørbel L (1994) Dovrefjell. Grøndahl Dreyer, Oslo
Bruun HH, Moen J, Virtanen R, Grytnes J-A, Oksanen L, Angerbjörn A (2006) Effects of altitude and topography on species richness of vascular plants, bryophytes and lichens in alpine communities. J Veg Sci 17:37–46. doi:10.1111/j.1654-1103.2006.tb02421.x
Cannone N, Sgorbati S, Guglielmin M (2007) Unexpected impacts of climate change on alpine vegetation. Front Ecol Environ 5:360–364. doi:10.1890/1540-9295(2007)5[360:UIOCCO]2.0.CO;2
Casanova-Katny A, Palfner G, Torres-Mellado GA, Cavieres LA et al (2014) Do Antarctic lichens modify microclimate and facilitate vascular plants in the maritime Antarctic? A comment to Molina-Montenegro et al. (). J Veg Sci 25:601–605. doi:10.1111/jvs.12122
Chapin FS, Shaver GR, Giblin AE, Nadelhoffer KJ, Laundre JA (1995) Responses of Arctic tundra to experimental and observed changes in climate. Ecology 76:694–711. doi:10.2307/1939337
Chapin FS, Sturm M, Serreze MC, McFadden JC, Key JR, Lloyd AH, McGuire AD, Rupp TS, Lynch AH, Schimel JP, Beringer J, Chapman WL, Epstein HE, Euskirchen ES, Hinzman LD, Jia G, Ping CL, Tape KD, Thompson CDC, Walker DA, Welker JM (2005) Role of land-surface changes in arctic summer warming. Science 310:657–660. doi:10.1126/science.1117368
Corlett RT, Westcott DA (2013) Will plant movements keep up with climate change? Trends Ecol Evol 28:482–488. doi:10.1016/j.tree.2013.04.003
Cornelissen JHC, Callaghan TV, Alatalo JM, Michelsen A, Graglia E, Hartley AE, Hik DS, Hobbie SE, Press MC, Robinson CH, Henry GHR, Shaver GR, Phoenix GK, Jones DG, Jonasson S, Chapin FS, Molau U, Neill C, Lee JA, Melillo JM, Sveinbjörnsson B, Aerts R (2001) Global change and arctic ecosystems: is lichen decline a function of increases in vascular plant biomass? J Ecol 89:984–994. doi:10.1046/j.1365-2745.2001.00625.x
Cornelissen JHC, Van Bodegom PM, Aerts R, Callaghan TV, Van Logtestijn RSP, Alatalo J, Stuart Chapin F, Gerdol R, Gudmundsson J, Gwynn-Jones D, Hartley AE, Hik DS, Hofgaard A, Jónsdóttir IS, Karlsson S, Klein JA, Laundre J, Magnusson B, Michelsen A, Molau U, Onipchenko VG, Quested HM, Sandvik SM, Schmidt IK, Shaver GR, Solheim B, Soudzilovskaia NA, Stenström A, Tolvanen A, Totland Ø, Wada N, Welker JM, Zhao X, Brancaleoni L, Brancaleoni L, De Beus MAH, Cooper EJ, Dalen L, Harte J, Hobbie SE, Hoefsloot G, Jägerbrand A, Jonasson S, Lee JA, Lindblad K, Melillo JM, Neill C, Press MC, Rozema J, Zielke M (2007) Global negative vegetation feedback to climate warming responses of leaf litter decomposition rates in cold biomes. Ecol Lett 10:619–627. doi:10.1111/j.1461-0248.2007.01051.x
Damsholt K (2002) Illustrated flora of Nordic liverworts and hornworts. Nordic Bryological Society, Lund
Danell K, Utsi MP, Palo TR, Eriksson O (1994) Food plant selection by reindeer during winter in relation to plant quality. Ecography 17:153–158. doi:10.1111/j.1600-0587.1994.tb00088.x
De Frenne P, Rodríguez-Sánchez F, Coomes DA, Baeten L, Verstraeten G, Vellend M, Bernhardt-Römermann M, Brown CD, Brunet J, Cornelis J, Decocq GM, Dierschke H, Eriksson O, Gilliam FS, Hédl R, Heinken T, Hermy M, Hommel P, Jenkins MA, Kelly DL, Kirby KJ, Mitchell FJG, Naaf T, Newman M, Peterken G, Petřík P, Schultz J, Sonnier G, Van Calster H, Waller DM, Walther G-R, White PS, Woods KD, Wulf M, Graae BJ, Verheyen K (2013) Microclimate moderates plant responses to macroclimate warming. Proc Natl Acad Sci USA 110:18561–18565. doi:10.1073/pnas.1311190110
De Witte LC, Stöcklin J (2010) Longevity of clonal plants: why it matters and how to measure it. Ann Bot Lond 106:859–870. doi:10.1093/aob/mcq191
Dorrepaal E, Aerts R, Cornelissen JHC, Van Logtestijn RSP, Callaghan TV (2006) Sphagnum modifies climate-change impacts on subarctic vascular bog plants. Funct Ecol 20:31–41. doi:10.1111/j.1365-2435.2006.01076.x
Dullinger S, Gattringer A, Thuiller W, Moser D, Zimmermann NE, Guisan A, Willner W, Plutzar C, Leitner M, Mang T, Caccianiga M, Dirnbock T, Ertl S, Fischer A, Lenoir J, Svenning J-C, Psomas A, Schmatz DR, Silc U, Vittoz P, Hulber K (2012) Extinction debt of high-mountain plants under twenty-first-century climate change. Nat Clim Change 2:619–622. doi:10.1038/nclimate1514
Elbert W, Weber B, Burrows S, Steinkamp J, Budel B, Andreae MO, Poschl U (2012) Contribution of cryptogamic covers to the global cycles of carbon and nitrogen. Nat Geosci 5:459–462. doi:10.1038/ngeo1486
Elmendorf SC, Henry GHR, Hollister RD, Björk RG, Bjorkman AD, Callaghan TV, Collier LS, Cooper EJ, Cornelissen JHC, Day TA, Fosaa AM, Gould WA, Grétarsdóttir J, Harte J, Hermanutz L, Hik DS, Hofgaard A, Jarrad F, Jonsdóttir IS, Keuper F, Klanderud K, Klein JA, Koh S, Kudo G, Lang SI, Loewen V, May JL, Mercado J, Michelsen A, Molau U, Myers-Smith IH, Oberbauer SF, Pieper S, Post E, Rixen C, Robinson CH, Schmidt NM, Shaver GR, Stenström A, Tolvanen A, Totland Ø, Troxler T, Wahren CH, Webber PJ, Welker JM, Wookey PA (2012) Global assessment of experimental climate warming on tundra vegetation: heterogeneity over space and time. Ecol Lett 15:164–175. doi:10.1111/j.1461-0248.2011.01716.x
Elmendorf SC, Henry GHR, Hollister RD, Fosaa AM, Gould WA, Hermanutz L, Hofgaard A, Jónsdóttir IS, Jorgenson JC, Lévesque E, Magnusson B, Molau U, Myers-Smith IH, Oberbauer SF, Rixen C, Tweedie CE, Walker MD (2015) Experiment, monitoring, and gradient methods used to infer climate change effects on plant communities yield consistent patterns. Proc Natl Acad Sci USA 112:448–452. doi:10.1073/pnas.1410088112
Elven R, Fremstad E, Hegre H, Nilsen L, Solstad H (1996) Botanical values in the Dovrefjell area. NTNU, pp 1–151
Felde VA, Kapfer J, Grytnes J-A (2012) Upward shift in elevational plant species ranges in Sikkilsdalen, central Norway. Ecography 35:922–932. doi:10.1111/j.1600-0587.2011.07057.x
Fuller E, Brush E, Pinsky ML (2015) The persistence of populations facing climate shifts and harvest. Ecosphere 6:1–16. doi:10.1890/ES14-00533.1
Geological Survey of Norway (2015). https://www.ngu.no/en. Accessed 9 Sep 2015
Gottfried M, Pauli H, Futschik A, Akhalkatsi M, Barančok P, Alonso B, Luis J, Coldea G, Dick J, Erschbamer B, Calzado F, Rosa M, Kazakis G, Krajči J, Larsson P, Mallaun M, Michelsen O, Moiseev D, Moiseev P, Molau U, Merzouki A, Nagy L, Nakhutsrishvili G, Pedersen B, Pelino G, Puscas M, Rossi G, Stanisci A, Theurillat J-P, Tomaselli M, Villar L, Vittoz P, Vogiatzakis I, Grabherr G (2012) Continent-wide response of mountain vegetation to climate change. Nat Clim Change 2:5. doi:10.1038/nclimate1329
Gower JC (1987) Introduction to ordination techniques. In: Legendre P, Legendre L (eds) Developments in numerical ecology. Springer, Berlin, pp 3–48
Grabherr G, Gottfried M, Pauli H (1994) Climate effect on mountain plants. Nature 369:448. doi:10.1038/369448a0
Grabherr G, Gottfried M, Pauli H (2010) Climate change impacts in alpine environments. Geogr Compass 4:1133–1153. doi:10.1111/j.1749-8198.2010.00356.x
Gray AN, Azuma DL (2005) Repeatability and implementation of a forest vegetation indicator. Ecol Indic 5:57–71. doi:10.1016/j.ecolind.2004.09.001
Grime JP, Frindley JD, Askew AP, Thompson K, Hodgson JG, Bennett CR (2008) Long-term resistance to climate change in an infertile grassland. Proc Natl Acad Sci USA 105:10028–10032. doi:10.1073/pnas.0711567105
Grytnes J-A, Heegaard E, Ihlen PG (2006) Species richness of vascular plants, bryophytes, and lichens along an altitudinal gradient in western Norway. Acta Oecol 29:241–246. doi:10.1016/j.actao.2005.10.007
Grytnes J-A, Kapfer J, Jurasinski G, Birks HH, Henriksen H, Klanderud K, Odland A, Ohlson M, Wipf S, Birks HJB (2014) Identifying the driving factors behind observed elevational range shifts on European mountains. Global Ecol Biogeogr 23:876–884. doi:10.1111/geb.12170
Hallingbäck T, Lönnell N, Weibull H, Hedenäs L, van Knorring P (2006) Nationalnyckelen till Sveriges flora och fauna. Bladmossor: Sköldmossor-blåmossor. Bryophyta: Buxbaumia-Leucobryum. ArtDatabanken, SLU, Uppsala
Hallingbäck T, Lönnell N, Weibull H, von Knorring P, Korotynska M, Reisborg C, Birgersson M. (2008) Nationalnyckelen till Sveriges flora och fauna. Bladmossor: Kompaktmossor-kapmossor. Bryophyta: Anoectangium-Orthodontium. ArtDatabanken, SLU, Uppsala
Hassel K, Presto T, Schmidt N (2012) Bryophyte Diversity in high and low arctic Greenland. Establishment of permanent monitoring transects and bryophyte mapping in Zackenberg and Kobbefjord 2009–2010. DCE–Danish Centre for Environment and Energy, pp 42
He X, He KS, Hyvönen J (2016) Will bryophytes survive in a warming world? Perspect Plant Ecol 19:49–60. doi:10.1016/j.ppees.2016.02.005
Hedenäs L, Reisborg C, Hallingbäck T (2014) Nationalnyckeln til Sveriges flora och fauna. Bladmossor: Skirmossor-baronmossor. Bryophyta: Hookeria-Anomodon. Artdatabanken, SLU, Uppsala
Helle T, Aspi J (1983) Effects of winter grazing by reindeer on vegetation. Oikos 40:337–343. doi:10.2307/3544306
Holzinger B, Hülber K, Camenisch M, Grabherr G (2008) Changes in plant species richness over the last century in the eastern Swiss Alps: elevational gradient, bedrock effects and migration rates. Plant Ecol 195:179–196. doi:10.1007/s11258-007-9314-9
Jägerbrand AK, Alatalo JM (2015) Effects of human trampling on abundance and diversity of vascular plants, bryophytes and lichens in alpine heath vegetation, Northern Sweden. SpringerPlus 4:95. doi:10.1186/s40064-015-0876-z
Jägerbrand A, During HJ (2005) Effects of simulated shade on growth, number of branches and biomass in Hylocomium splendens and Racomitrium lanuginosum. Lindbergia 30:117–124. doi:10.1016/j.polar.2012.02.002
Jägerbrand AK, Kudo G, Alatalo JM, Molau U (2012) Effects of neighboring vascular plants on the abundance of bryophytes in different vegetation types. Polar Sci 6:200–208. doi:10.1016/j.polar.2012.02.002
Jansen F, Dengler J (2010) Plant names in vegetation databases—a neglected source of bias. J Veg Sci 21:1179–1186. doi:10.1111/j.1654-1103.2010.01209.x
Johansen L, Wehn S, Hovstad KA (2016) Clonal growth buffers the effect of grazing management on the population growth rate of a perennial grassland herb. Flora 223:11–18. doi:10.1016/j.flora.2016.04.007
Joly K, Jandt RR, Klein DR (2009) Decrease of lichens in Arctic ecosystems: the role of wildfire, caribou, reindeer, competition and climate in north-western Alaska. Polar Res 28:433–442. doi:10.1111/j.1751-8369.2009.00113.x
Kapfer J, Virtanen R, Grytnes J-A (2012) Changes in arctic vegetation on Jan Mayen Island over 19 and 80 years. J Veg Sci 23:771–781. doi:10.1111/j.1654-1103.2012.01395.x
Kershaw KA (1985) The physiological ecology of lichens. Cambridge University Press, Cambridge
Klanderud K, Birks H (2003) Recent increases in species richness and shifts in altitudinal distributions of Norwegian mountain plants. Holocene 13:1–6. doi:10.1191/0959683603hl589ft
Klanderud K, Totland Ø (2005) Simulated climate change altered dominance hierarchies and diversity of an Alpine biodiversity hotspot. Ecology 86:2047–2054. doi:10.1890/04-1563
Klimeš L, Dančak M, Hájek M, Jongepierová I, Kučera T (2001) Scale-dependent biases in species counts in a grassland. J Veg Sci 12:699–704. doi:10.2307/3236910
Körner C (2003) Alpine plant life: functional plant ecology of high mountain, 2nd edn. Springer, Berlin
Kosaka Y, Xie SP (2013) Recent global-warming hiatus tied to equatorial Pacific surface cooling. Nature 501:403–407. doi:10.1038/nature12534
Kullman L (2007) Tree line population monitoring of Pinus sylvestris in the Swedish Scandes, 1973–2005: implications for tree line theory and climate change ecology. J Ecol 95:41–52. doi:10.1111/j.1365-2745.2006.01190.x
Lang SI, Cornelissen JHC, Klahn T, van Logtestijn RSP, Broekman R, Schweikert W, Aerts R (2009) An experimental comparison of chemical traits and litter decomposition rates in a diverse range of subarctic bryophyte, lichen and vascular plant species. J Ecol 97:886–900. doi:10.1111/j.1365-2745.2009.01538.x
Lang SI, Cornelissen JHC, Shaver GR, Ahrens M, Callaghan TV, Molau U, Ter Braak CJF, Hölzer A, Aerts R (2012) Arctic warming on two continents has consistent negative effects on lichen diversity and mixed effects on bryophyte diversity. Glob Change Biol 18:1096–1107. doi:10.1111/j.1365-2486.2011.02570.x
Lenoir J, Gégout JC, Marquet PA, de Ruffray P, Brisse H (2008) A Significant Upward Shift in Plant Species Optimum Elevation During the 20th Century. Science 320:1768
Lenoir J, Virtanen R, Oksanen J, Oksanen L, Luoto M, Grytnes J-A, Svenning J-C (2012) Dispersal ability links to cross-scale species diversity patterns across the Eurasian Arctic tundra. Glob Ecol Biogeogr 21:851–860. doi:10.1111/j.1466-8238.2011.00733.x
Longton RE (1988) The biology of polar bryophytes and lichens. Cambridge University Press, Cambridge
Malanson GP, Rose JP, Schroeder PJ, Fagre DB (2011) Contexts for change in alpine tundra. Phys Geogr 32:97–113. doi:10.2747/0272-3646.32.2.97
Malmer N, Svensson BM, Wallén B (1994) Interactions between Sphagnum mosses and field layer vascular plants in the development of peat-forming systems. Folia Geobot Phytotx 29:483–496. doi:10.1007/BF02883146
Marschall M, Proctor MCF (2004) Are bryophytes shade plants? Photosynthetic light responses and proportions of chlorophyll a, chlorophyll b and total carotenoids. Ann Bott Lond 94:593–603. doi:10.1093/aob/mch178
Mateo RG, Broennimann O, Normand S, Petitpierre B, Araújo MB, Svenning J-C, Baselga A, Fernández-González F, Gómez-Rubio V, Muñoz J, Suarez GM, Luoto M, Guisan A, Vanderpoorten A (2016) The mossy north: an inverse latitudinal diversity gradient in European bryophytes. Sci Rep UK 6:25546. doi:10.1038/srep25546
Matveyeva N, Chernov Y (2000) Biodiversity of terrestrial ecosystems. In: Nutall M, Callaghan TV (eds) The Arctic: environment, people, policy. Hardwood Academic Publishers, New York, pp 233–274
Mayor JR, Sanders NJ, Classen AT, Bardgett RD, Clément J-C, Fajardo A, Lavorel S, Sundqvist MK, Bahn M, Chisholm C, Cieraad E, Ze Gedalof, Grigulis K, Kudo G, Oberski DL, Wardle DA (2017) Elevation alters ecosystem properties across temperate treelines globally. Nature 542:91–95. doi:10.1038/nature21027
McCain CM, Grytnes J-A (2010) Elevational gradients in species richness encyclopidia of life sciences. John Wiley, New York
Michelsen O, Syverhuset AO, Pedersen B, Holten JI (2011) The impact of climate change on recent vegetation changes on Dovrefjell, Norway. Diversity 3:91–111. doi:10.3390/d3010091
Milberg P, Bergstedt J, Fridman J, Odell G, Westerberg L (2008) Observer bias and random variation in vegetation monitoring data. J Veg Sci 19:633–644. doi:10.3170/2008-8-18423
Myers-Smith IH, Forbes CB, Wilmking M, Hallinger M, Lantz T, Blok D, Tape KD, Macias-Fauria M, Sass-Klaassen U, Lévesque E, Boudreau S, Ropars P, Hermanutz L, Trant A, Collier LS, Weijers S, Rozema J, Rayback SA, Schmidt NM, Schaepman-Strub G, Wipf S, Rixen C, Ménard CB, Venn S, Scott G, Andreu-Hayles L, Elmendorf SC, Ravolainen VT, Welker J, Grogan P, Epstein HE, Hik DS (2011) Shrub expansion in tundra ecosystems: dynamics, impacts and research priorities. Environ Res Lett 6:045509. doi:10.1088/1748-9326/6/4/045509
Nash TH (2008) Lichen biology, 2nd edn. Cambridge University Press, Cambridge
Neilson RP, Pitelka LF, Solomon AM, Nathan R, Midgley GF, Fragoso JMV, Lischke H, Thompson K (2005) Forecasting regional to global plant migration in response to climate change. Bioscience 55:749–759. doi:10.1641/0006-3568(2005)055[0749:FRTGPM]2.0.CO;2CO;2
Norwegian Meteorological Institute (2015) Climate in Norway. http://met.no/English. Accessed 9 Sep 2015
Nyholm E (1969) Illustrated moss flora of Fennoscandia: Musci, fasc. 1–6. Natural Science Research Council, Lund
Oksanen J, Blanchet GB, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2017). vegan: Community Ecology Package. R package version 2.4-2. https://CRAN.R-project.org/package=vegan
Olofsson J, Oksanen L, Callaghan T, Hulme PE, Oksanen T, Suominen O (2009) Herbivores inhibit climate-driven shrub expansion on the tundra. Glob Change Biol 15:2681–2693. doi:10.1111/j.1365-2486.2009.01935.x
Pajunen A, Virtanen R, Roininen H (2012) Browsing-mediated shrub canopy changes drive composition and species richness in forest-tundra ecosystems. Oikos 121:1544–1552. doi:10.1111/j.1600-0706.2011.20115.x
Parmesan C, Yohe G (2003) A globally coherent fingerprint of climate change impacts across natural systems. Nature 421:37–42. doi:10.1038/nature01286
Parolo G, Rossi G (2008) Upward migration of vascular plants following a climate warming trend in the Alps. Basic Appl Ecol 9:100–107. doi:10.1016/j.baae.2007.01.005
Pauli H, Gottfried M, Reiter K, Klettner C, Grabherr G (2007) Signals of range expansions and contractions of vascular plants in the high Alps: observations (1994–2004) at the GLORIA master site Schrankogel, Tyrol, Austria. Glob Change Biol 13:147–156. doi:10.1111/j.1365-2486.2006.01282.x
Pauli H, Gottfried M, Dullinger S, Abdaladze O, Akhalkatsi M, Alonso JLB, Coldea G, Dick J, Erschbamer B, Calzado RF, Ghosn D, Holten JI, Kanka R, Kazakis G, Kollár J, Larsson P, Moiseev P, Moiseev D, Molau U, Mesa JM, Nagy L, Pelino G, Puşcaş M, Rossi G, Stanisci A, Syverhuset AO, Theurillat J-P, Tomaselli M, Unterluggauer P, Villar L, Vittoz P, Grabherr G (2012) Recent plant diversity changes on Europe’s mountain summits. Science 336:353. doi:10.1126/science.1219033
Pauli H, Gottfried M, Lamprecht A, Niessner S, Rumpf S, Winkler M, Steinbauer K, Grabherr G (2015) The GLORIA field manual—standard multi-summit approach, supplementary methods and extra approaches. Glob Obs Res Initiat Alp Environ. doi:10.2777/095439
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2017). nlme: linear and nonlinear mixed effects models. R package version 3.1-130. https://CRAN.R-project.org/package=nlme
Porada P, Ekici A, Beer C (2016) Effects of bryophyte and lichen cover on permafrost soil temperature at large scale. Cryosphere 10:2291–2315. doi:10.5194/tc-10-2291-2016
Post E, Forchhammer MC, Bret-Harte MS, Callaghan TV, Christensen TR, Elberling B, Fox AD, Gilg O, Hik DS, Høye TT, Ims RA, Jeppesen E, Klein DR, Madsen J, McGuire AD, Rysgaard S, Schindler DE, Stirling I, Tamstorf MP, Tyler NJC, van der Wal R, Welker J, Wookey PA, Schmidt NM, Aastrup P (2009) Ecological dynamics across the arctic associated with recent climate change. Science 325:1355–1358
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org
Rey Benayas JM, Scheiner SM (2002) Plant diversity, biogeography and environment in Iberia: patterns and possible causal factors. J Veg Sci 13:245–258. doi:10.1111/j.1654-1103.2002.tb02045.x
Rinke A, Kuhry P, Dethloff K (2008) Importance of a soil organic layer for Arctic climate: a sensitivity study with an Arctic RCM. Geophys Res Lett 35. doi:10.1029/2008GL034052
Schmidt GA, Shindell DT, Tsigaridis K (2014) Reconciling warming trends. Nat Geosci 7:158–160. doi:10.1038/ngeo2105
Schuur EAG, McGuire AD, Schadel C, Grosse G, Harden JW, Hayes DJ, Hugelius G, Koven CD, Kuhry P, Lawrence DM, Natali SM, Olefeldt D, Romanovsky VE, Schaefer K, Turetsky MR, Treat CC, Vonk JE (2015) Climate change and the permafrost carbon feedback. Nature 520:171–179. doi:10.1038/nature14338
Schwartz MD, Ahas R, Aasa A (2006) Onset of spring starting earlier across the Northern Hemisphere. Glob Change Biol 12:343–351. doi:10.1111/j.1365-2486.2005.01097.x
Sedia EG, Ehrenfeld JG (2003) Lichens and mosses promote alternate stable plant communities in the New Jersey Pinelands. Oikos 100:447–458. doi:10.1034/j.1600-0706.2003.12058.x
Semboli O, Beina D, Closset-Kopp D, Gourlet-Fleury S, Decocq G (2014) Does long-term monitoring of tropical forests using permanent plots provide unbiased results? Appl Veg Sci 17:737–743. doi:10.1111/avsc.12097
Solheim B, Endal A, Vigstad H (1996) Nitrogen fixation in Arctic vegetation and soils from Svalbard, Norway. Polar Biol 16:35–40. doi:10.1007/BF02388733
Speed JDM, Austrheim G, Mysterud A (2013) The response of plant diversity to grazing varies along an elevational gradient. J Ecol 101:1225–1236. doi:10.1111/1365-2745.12133
Syverhuset AO (2009) Recent changes in temperature and vegetation on Dovrefjell. NTNU—Norwegian University of Science and Technology, Trondheim
Takala T, Tahvanainen T, Kouki J (2014) Grazing promotes bryophyte species richness in seminatural grasslands. Ann Bot Fenn 51:148–160. doi:10.5735/085.053.0102
Thell A, Moberg R (2011) Cladoniaceae. In: Ahti T, Stenroos S, Moberg R (eds) Nodric lichen flora, museum of evolution. Uppsala University, Uppsala
Theurillat J-P, Guisan A (2001) Potential impact of climate change on vegetation in the European Alps: a review. Clim Change 50:77–109. doi:10.1023/A:1010632015572
Torsten H, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363. doi:10.1002/bimj.200810425
Tylianakis JM, Didham RK, Bascompte J, Wardle DA (2008) Global change and species interactions in terrestrial ecosystems. Ecol Lett 11:1351–1363. doi:10.1111/j.1461-0248.2008.01250.x
Van Breemen N (1995) How Sphagnum bogs down other plants. Trends Ecol Evol 10:270–275. doi:10.1016/0169-5347(95)90007-1
Van Calster H, Baeten L, Verheyen K, De Keersmaeker L, Dekeyser S, Rogister JE, Hermy M (2008) Diverging effects of overstorey conversion scenarios on the understorey vegetation in a former coppice-with-standards forest. For Ecol Manag 256:519–528. doi:10.1016/j.foreco.2008.04.042
Väre H, Ohtonen R, Mikkola K (1996) The effect and extent of heavy grazing by reindeer in oligotrophic pine heaths in northeastern Fennoscandia. Ecography 19:245–253. doi:10.1111/j.1600-0587.1996.tb01251.x
Verheyen K, De Frenne P, Baeten L, Waller DM, Hédl R, Perring MP, Blondeel H, Brunet J, Chudomelová M, Decocq G, De Lombaerde E, Depauw L, Dirnböck T, Durak T, Eriksson O, Gilliam FS, Heinken T, Heinrichs S, Hermy M, Jaroszewicz B, Jenkins MA, Johnson SE, Kirby KJ, Kopecký M, Landuyt D, Lenoir J, Li D, Macek M, Maes SL, Máliš F, Mitchell FJG, Naaf T, Peterken G, Petřík P, Reczyńska K, Rogers DA, Schei FH, Schmidt W, Standovár T, Świerkosz K, Ujházy K, Van Calster H, Vellend M, Vild O, Woods K, Wulf M, Bernhardt-Römermann M (2017) Combining biodiversity resurveys across regions to advance global change research. Bioscience 67:73–83. doi:10.1093/biosci/biw150
Virtanen R, Eskelinen A, Gaare E (2003) Long-term changes in Alpine plant communities in Norway and Finland. In: Nagy L, Grabherr G, Körner C, Thompson DBA (eds) Alpine biodiversity in Europe. Springer, Berlin, pp 411–422
Vistnes I, Nellemann C (2008) Reindeer winter grazing in alpine tundra: impacts on ridge community composition in Norway. Arct Antarct Alp Res 40:215–224. doi:10.1657/1523-0430(07-001)[VISTNES]2.0.CO;2
Vittoz P, Guisan A (2007) How reliable is the monitoring of permanent vegetation plots? A test with multiple observers. J Veg Sci 18:413–422. doi:10.1111/j.1654-1103.2007.tb02553.x
Vittoz P, Bayfield N, Brooker R, Elston DA, Duff EI, Theurillat J-P, Guisan A (2010a) Reproducibility of species lists, visual cover estimates and frequency methods for recording high-mountain vegetation. J Veg Sci 21:1035–1047. doi:10.1111/j.1654-1103.2010.01216.x
Vittoz P, Camenisch M, Mayor R, Miserere L, Vust M, Theurillat J-P (2010b) Subalpine-nival gradient of species richness for vascular plants, bryophytes and lichens in the Swiss inner Alps. Bot Helv 120:139–149. doi:10.1007/s00035-010-0079-8
Walker MD, Wahren CH, Hollister RD, Henry GHR, Ahlquist LE, Alatalo JM, Bret-Harte MS, Calef MP, Callaghan TV, Carroll AB, Epstein HE, Jónsdóttir IS, Klein JA, Magnússon B, Molau U, Oberbauer SF, Rewa SP, Robinson CH, Shaver GR, Suding KN, Thompson CC, Tolvanen A, Totland Ø, Turner PL, Tweedie CE, Webber PJ, Wookey PA (2006) Plant community responses to experimental warming across the tundra biome. Proc Natl Acad Sci USA 103:1342–1346. doi:10.1073/pnas.0503198103
Wielgolaski FE, Bliss LC, Svoboda J, Doyle G (1981) Tundra ecosystems: a comparative analysis. Cambridge University Press, Cambridge
Winkler M, Lamprecht A, Steinbauer K, Hülber K, Theurillat J-P, Breiner F, Choler P, Ertl S, Gutiérrez Girón A, Rossi G, Vittoz P, Akhalkatsi M, Bay C, Benito Alonso J-L, Bergström T, Carranza ML, Corcket E, Dick J, Erschbamer B, Fernández Calzado R, Fosaa AM, Gavilán RG, Ghosn D, Gigauri K, Huber D, Kanka R, Kazakis G, Klipp M, Kollar J, Kudernatsch T, Larsson P, Mallaun M, Michelsen O, Moiseev P, Moiseev D, Molau U, Molero Mesa J, Morra di Cella U, Nagy L, Petey M, Pușcaș M, Rixen C, Stanisci A, Suen M, Syverhuset AO, Tomaselli M, Unterluggauer P, Ursu T, Villar L, Gottfried M, Pauli H (2016) The rich sides of mountain summits—a pan-European view on aspect preferences of alpine plants. J Biogeogr 43:2261–2273. doi:10.1111/jbi.12835
Wolf A, Lazzarotto P, Bugmann H (2012) The relative importance of land use and climatic change in Alpine catchments. Clim Change 111:279–300. doi:10.1007/s10584-011-0209-3
Wolkovich EM, Cook BI, Allen JM, Crimmins TM, Betancourt JL, Travers SE, Pau S, Regetz J, Davies TJ, Kraft NJB, Ault TR, Bolmgren K, Mazer SJ, McCabe GJ, McGill BJ, Parmesan C, Salamin N, Schwartz MD, Cleland EE (2012) Warming experiments underpredict plant phenological responses to climate change. Nature 485:494–497. doi:10.1038/nature11014
Wookey PA, Aerts R, Bardgett RD, Baptist F, Bråthen KA, Cornelissen JHC, Gough L, Hartley IP, Hopkins DW, Lavorel S, Shaver GR (2009) Ecosystem feedbacks and cascade processes: understanding their role in the responses of Arctic and alpine ecosystems to environmental change. Glob Change Biol 15:1153–1172. doi:10.1111/j.1365-2486.2008.01801.x
Acknowledgements
The fieldwork and data collection were carried out within the framework of the third vegetation sampling campaign of the Global Observation Research Initiative in Alpine Environments (GLORIA). In 2015, the fieldwork was financed by the Norwegian Environment Agency (Miljødirektoratet), whereas the fieldwork in 2001 and 2008 was financed by the GLORIA-Europe project within the 5th Research, Technical Development and Demonstration Framework Program (FP5) of the European Union and the Norwegian Directorate for Nature Management, respectively. PDF held a fellowship of the Research Foundation – Flanders (FWO). Finally, we thank Kristin Odden Nystuen and three anonymous reviewers for useful comments and constructive suggestions on previous versions of this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original version of this article was revised due to a retrospective Open Access.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Vanneste, T., Michelsen, O., Graae, B.J. et al. Impact of climate change on alpine vegetation of mountain summits in Norway. Ecol Res 32, 579–593 (2017). https://doi.org/10.1007/s11284-017-1472-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-017-1472-1