Abstract
Distorted plant diversity patterns due to ungulate herbivory could be explained by changes in community assembly processes, but the effects of ungulate herbivory on plant community assembly remain unclear. Here, we examined the role of deer herbivory in the regulation of the assembly processes of a forest floor plant community by assessing species and functional diversity in over- and no-grazing plots (control and exclosure plots, respectively) in Shiretoko National Park in Japan. Compared with the exclosure plot, vegetation coverage was considerably lower, and species richness and diversity were higher in the control plot. Functional traits associated with competitive ability (leaf area and chlorophyll content) were significantly higher in the exclosure plot. The pattern of functional diversity changed from overdispersion to clustering with an increase in local crowdedness. This trait clustering indicates that the local communities that were free from ungulate disturbance gradually became dominated by some competitively superior plant species, which led to low species diversity and biotic homogenization. In contrast, the reduction in vegetation due to overgrazing by deer resulted in an increase in the relative importance of stochastic assembly processes, which enabled the coexistence of various species, including less competitive ones. Our results emphasize that although deer overabundance is of concern, their complete exclusion has a negative consequence from an ecological perspective. Because deer herbivory is an inherent process that affects the biodiversity of plants on the forest floor, the establishment of fences requires careful consideration to ensure the conservation of ecological processes and their associated biodiversity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In terrestrial ecosystems worldwide, overgrazing by large ungulates (e.g., deer) has created major problems that have impacted the biodiversity, as well as other properties, of the ecosystems that these animals inhabit (Rooney 2001; Côté et al. 2004; Mori et al. 2015). Although herbivory is an inherently consumptive process in forest ecosystems, the overabundance of ungulates has been attributed to human activities, including anthropogenic warming (Forchhammer et al. 1998; Loison et al. 1999), land-use change (Bobek et al. 1984; Roseberry and Woolf 1998), reduced hunting pressure (Brown et al. 2000; Riley et al. 2003), and the loss of keystone predator species (Rooney 2001), and it has been proposed to have adverse effects on vegetation (Côté et al. 2004; Takatsuki 2009). Deer have direct and indirect impacts on the biotic interactions among coexisting plant species (Rooney and Waller 2003). The former includes selective herbivory that is associated with the palatability of plants (Augustine and McNaughton 1998), while the latter is exemplified by changes in habitat conditions due to browsing that can result in gap formation and altered nutrient cycling (Wardle et al. 2002; Bardgett and Wardle 2003). As a result, the structure, composition, and diversity of plant communities can be substantially altered (Russell et al. 2001; Horsley et al. 2003). To safeguard vegetation from overgrazing by ungulates, fences designed to exclude these large herbivores have often been established in various regions, but their effects on local plant diversity have been mixed (Côté et al. 2004; Inatomi et al. 2012). To understand these contradictory results, a rigorous assessment of how intense herbivory alters the structure of biodiversity is necessary, and in doing so, it is worth focusing on the theory of community assembly. However, while most research on the consequences of establishing ungulate fences has primarily quantified the changes in biodiversity patterns, the underlying assembly processes have not been fully assessed. To better guide conservation and management activities in the face of ungulate overabundance, this gap in our knowledge must be urgently addressed.
There are two possible processes of community assembly: deterministic and stochastic (Chase et al. 2011). The deterministic process is based on niche theory (Hutchinson 1959), and when this process is dominant, community composition should depend on local biotic and abiotic environmental conditions. In contrast, when stochastic processes are dominant, community assembly is primarily shaped randomly (Chase et al. 2011). The biotic interactions among species often act strongly at a fine scale (de Bello et al. 2013); thus, these interactions are affected by local factors, such as environmental conditions or disturbances, which have been shown to have various effects on community assembly by altering the relative importance of deterministic and stochastic processes (Chase 2007; Vellend et al. 2007; Chase et al. 2011). This may also be the case for deer herbivory (Begley-Miller et al. 2014; Ohashi and Hoshino 2014).
Recently, there has been a rapid increase in the use of trait-based approaches to reveal community assembly processes at local scales (Kraft et al. 2008, 2014; Angert et al. 2009; Cadotte et al. 2011; Weiher et al. 2011; Götzenberger et al. 2012). Functional traits are any measurable features that potentially affect individual performance or the fitness of organisms (Cadotte et al. 2011). Functional traits determine whether individuals survive or species persist (Lavorel et al. 1997), as well as how they interact with one another, which provide information regarding the strength of competition and the efficiency of consumptive interactions (Davies et al. 2007). When multiple trait dimensions are considered, the ecological differentiation between species may be elucidated because these sets of traits are likely relevant to the ecological tolerance, ecological niche, and competitive ability of a species (Cadotte et al. 2011; Mouillot et al. 2013). Importantly, functional diversity (FD), the dispersion of species in multidimensional trait space, provides information regarding how ecologically similar or dissimilar species are assembled (Petchey and Gaston 2007; Laliberté and Legendre 2010). In theory, a significant departure from random assembly can generate two opposing patterns that are suggestive of deterministic processes, and these patterns can emerge as either low or high FD, which indicate trait clustering or overdispersion, respectively. Under the classical paradigm, ecologically dissimilar species can coexist through niche partitioning, which is based on the theory that the coexistence of functionally similar species is limited by interspecific competition (i.e., limiting similarity; MacArthur and Levins 1967; MacArthur 1972; Chesson 2000). These niche differences can rise from species-specific interactions with resources, specialist consumers, pathogens, mutualists, or temporal environmental heterogeneity (Tilman 1982, 1988; Chesson 2000; Sammul et al. 2006; Chesson and Kuang 2008), and trait overdispersion is thought to be the consequence of this process. In contrast, shared adaptive characteristics among syntopic species promote the co-existence of species with similar traits. Under intense abiotic interactions, traits related to ecological tolerances are important (Cornwell et al. 2006; Grime 2006), but under resource-rich conditions, species with high competitive ability are superior to less competitive ones (Kunstler et al. 2012; Kraft et al. 2014). These processes promote trait clustering (Mayfield and Levine 2010). Likewise, the trait-based approach allows the important mechanisms underlying community assembly (Götzenberger et al. 2012) and the biodiversity response (Mori et al. 2013) to be inferred.
Serious ecological concerns regarding the overabundance of sika deer (Cervus nippon yesoensis) (e.g., overgrazing/browsing) throughout Japan have been frequently expressed (Takatsuki 2009), and these concerns even extend to areas with pristine vegetation that have been designated as protected areas. In Shiretoko National Park (located on the northeastern tip of Hokkaido, Japan), deer overabundance has resulted in numerous ecological issues, including negative impacts on forest understory plant communities (Kaji et al. 2004; Mori et al. 2015). To conserve forest vegetation, deer exclosure fences were established in the early 2000s, and since more than a decade has passed, we expect that the presence and absence of selective deer herbivory has considerably changed the biotic interactions among the plant species in these forests. This is because these areas are now characterized by both an overabundance and an underabundance of deer, which are expected to have impacted the forest understory communities. Here, we aimed to detect the ecological effects of sika deer on the mechanisms of plant species coexistence, as well as the consequences for the patterns of biodiversity, by assessing the differences in community assembly processes between these two ecological extremes (i.e., deer overgrazing and no overgrazing). We envision that this study will inform management treatments and policies that are designed to cope with deer overabundance in the study area and in other regions.
Methods
Study site
This study was conducted in Shiretoko National Park, which is located in northeastern Hokkaido, the northernmost island of Japan (Mori et al. 2016a), and is registered as a United Nations Educational, Scientific, and Cultural Organization World Natural Heritage Site because of its high biodiversity. The mean annual temperature and precipitation at the study site between 2004 and 2014 were 6.5 °C and 1271 mm, respectively (http://www.data.jma.go.jp/obd/stats/etrn/index.php). In this region, the population of sika deer has increased since the late 1980s, and in areas of high deer density, the ground layer of the plant community has been transformed. To address this problem, deer exclosure fences have been constructed in these areas. Horobetsu is one of the most highly populated areas on the Shiretoko Peninsula, and it is located in the northcentral part (44°06′00″N 145°01′42″E) of the peninsula. The forest here is characterized as a mixed coniferous and broad-leaved natural forest that is dominated by Abies sachalinensis, Quercus crispula, and Kalopanax septemlobus, and the forest floor is typically covered with snow from November to May (Kubota 2000). Since around 2000, the density of deer in this area has been as high as approximately 5–10 deer/km, as estimated by light censuses that were conducted in autumn (http://dc.shiretoko-whc.com/). To reduce the deer grazing pressure on the vegetation, exclosure fences were constructed in 2003, and as a result, the coverage of the ground-layer plants inside the boundaries of the fences has increased.
Field data collection
To quantify the effects of deer foraging on the composition of the ground-layer plant community, we established two study plots in the forest. The exclosure plot had fences to protect the vegetation from deer herbivory, and the control plot, which was located outside the boundaries of the fence, was exposed to deer herbivory. The sizes of the exclosure and control plots were 80 m × 120 m (0.96 ha) and 100 m × 100 m (1.00 ha), respectively, and they were located close together so that there was little difference in the species composition of the canopy trees (The Forestry Agency of Japan 2004). These exclosure and control plots were partitioned into 96 and 100 10 m × 10 m subplots, respectively, and at the center of each subplot, we established a 1 m × 1 m quadrat (i.e., there were 96 and 100 quadrats in the exclosure and control plots, respectively). Each plant species smaller than 50 cm in height within each quadrat was taxonomically identified, and its percent cover was estimated by a guided visual observation using a 1 m × 1 m nylon mesh that was divided into 100 10 cm × 10 cm grids. This survey was conducted from late June to early July 2014, the season during which most of the herbaceous species appear on the forest floor after thawing.
To evaluate the effects of biotic and abiotic conditions on plant community structure, we measured several environmental variables, including three soil variables (water content (WC), electrical conductivity (EC), and pH) and four stand structural variables (tree species richness, stem number, total basal area of the trees, and canopy openness), of the 10 m × 10 m subplots. We used the total vegetation ground cover on the forest floor as an indicator of local crowdedness, which can have important consequences for species composition via the prevention of seed colonization and the limitation of light availability at fine scales (Berntson and Wayne 2000; Mori and Takeda 2003), and this metric was arcsine-transformed to improve its normality (Snedecor and Cochran 1967; Sokal and Rohlf 2012). Soil WC (%) and soil EC (mS/cm) values were the means of three repeated random measurements in the 1 m × 1 m quadrats, and they were quantified by inserting a soil moisture meter (DM-18, Takemura Denki, Tokyo, Japan) and an EC tester (soil test HI 98331, Hanna Instruments, Chiba, Japan), respectively, into the top 10 cm of the soil. For soil pH, we collected a soil sample of approximately 0.3 cm3 from a depth of 2 cm in each quadrat, mixed it with the same amount of tap water, and performed the measurement with a pH meter (LAQUA twin B-71X, Horiba, Ltd., Kyoto, Japan). We calculated canopy openness with CanopOn2 (http://takenaka-akio.org/etc./canopon2) using hemispherical photographs taken by a Sony NEX-5 camera (Sony, Tokyo, Japan) mounted with a fish-eye lens (MADOKA, Yasuhara Co., Ltd., Tokyo, Japan) at the center of each subplot. The stand structural variables were sourced from the Forestry Agency of Japan (2014), which measured the size and species identity of all trees taller than 2 m at every 10 m × 10 m subplots.
Trait data collection
We used 10 plant functional traits related to growth, survival, and the response to environmental change (Pérez-Harguindeguy et al. 2013), including leaf area (LA; mm2), specific leaf area (SLA; mm2 g−1), chlorophyll content (measured by the SPAD device unit [see below]), maximum plant height (plant height; cm), leaf phenology, life form, growth form, herbaceous or woody, seed mass (g), and dispersal mode. For LA, SLA, and chlorophyll content, we collected and measured five samples from five individuals of each plant species (25 leaf samples for each species). First, we collected a fully formed adult leaf and measured its chlorophyll content using a Minolta SPAD-502 (Osaka, Japan), and then we immediately scanned the leaf before drying, and calculated the LA from the scanned images using Image-J (Rasband 1997–2008). We used the leaflets for plant species with compound leaves. To calculate the SLA, we dried the leaves for 72 h at 80 °C to obtain their dry weights. The values of the other traits were extracted from the literature and encyclopedias (ESM 1). Trait measurement and sample collection followed the protocols outlined in Pérez-Harguindeguy et al. (2013).
Data analysis
To describe the effect of deer presence/absence on plant communities, we calculated species richness and Shannon’s index values as measures of species diversity (Magurran and McGill 2011). To examine changes in plant community structure, we also determined the Bray–Curtis index, which is one of the most commonly used measures of vegetation compositional dissimilarity (Anderson et al. 2011). To evaluate differences in the species abundance distribution between the two plots, we plotted the frequency of occurrence of each plant species within all quadrats (the frequency of occurrence across all quadrats) by their dominance rank within the meta-community (hereafter, the species dominance-rank curve). We visually checked for differences in the curves and in the lists of dominant species between the two plots.
Furthermore, we calculated the community-weighted mean (CWM) trait values, which are the mean trait values of all species in the community weighted by their relative abundance (Garnier et al. 2004), for five numeric plant traits that are likely related to plant responses to deer herbivory: LA, SLA, chlorophyll content, plant height, and seed mass. It is known that deer selectively graze large LAs of Trillium grandiflorum (Knight 2003), and the SLA is the ratio of leaf area to leaf mass, which is positively correlated with the leaf nitrogen (N) concentration and negatively correlated with leaf longevity (Reich et al. 1997). Chlorophyll is highly correlated with tissue N (Markwell and Blevins 1999). These traits, which are related to the abundances of resources in leaves, are likely affected by deer herbivory. Maximum plant height is also associated with competitive ability (Vojtech et al. 2008) and vulnerability to deer herbivory, and seed mass is related to survival and establishment in the face of environmental hazards (Pérez-Harguindeguy et al. 2013). Although deer herbivory could increase the intraspecific variations in trait values, we used the mean values for each species to calculate the CWM values in this study.
We calculated functional diversity using the functional dispersion (FDis) index that was developed by Laliberté and Legendre (2010). The FDis is the mean distance of all species in the community in multidimensional trait space weighted by their relative abundance, and we selected this index because FDis is independent of species richness, can account for species abundances, and can be used for multiple traits (Laliberté and Legendre 2010). In this study, we calculated FDis using all of the functional traits that we measured (10 traits) to express the ecological differentiation between species (Cadotte et al. 2011; Mouillot et al. 2013). To evaluate the magnitude of deviation from a random assembly, we calculated the standardized effect size (SES) of the FDis, which was defined as (FDisobs − FDisnull)/FDissd, where FDisobs is the observed FDis in actual communities, and FDisnull and FDissd are the mean and standard deviation of FDis values that are calculated by 999 randomizations, respectively. We repeated this procedure for the exclosure and control plots. Randomization was based on the independent swap algorithm (Gotelli and Gotelli 2000), which maintains the species occurrence frequency and sample species richness based on the meta-community. Positive and negative SES values indicate higher (trait overdispersion) and lower (trait clustering) functional diversity, respectively, than expected at random; thus, to examine how biodiversity was structured, we tested for a correlation between the diversity measures (SES of the FDis and Shannon’s diversity) and environmental variables across quadrats. We used a standardized major axis (SMA) regression to describe how these variables were related (Warton et al. 2006; Sokal and Rohlf 2012).
Results
Field sampling
There were a total of 54 species in all of the study sites, of which 35 were found inside the fence (exclosure), and 43 were found outside the fence (control). Twenty-four species were common to both plots. The four dominant species were Dryopteris expansa, Maianthemum dilatatum, Sasa kurilensis, and Toxicodendron orientale, but their coverage was low in the control plot (Fig. S1). In addition, almost all of the other species that were common to both plots had lower coverage in the control plot than in the exclosure plot.
Species diversity
Mean species richness and Shannon’s diversity were significantly higher in the control plot (P < 0.001; t test) (Fig. 1a, b), as was the mean Bray–Curtis dissimilarity value (P < 0.001; t-test) (Fig. 1c). In contrast, the mean ground cover of the vegetation was significantly higher in the exclosure plot (P < 0.001; t-test) (Fig. 1d). The dominance-rank curve exhibited different forms between the two plots (Fig. 2). In the exclosure plot, the dominance of each species decreased sharply as species rank decreased; thus, species were divided into dominant and rare species. In contrast, the control plot exhibited a smooth curve that had more species with intermediate frequencies.
Comparison of a species richness, b Shannon’s diversity index, c Bray–Curtis dissimilarity index, and d coverage (%) values for each quadrat between the exclosure and control plots. The numbers of samples used in a, b, and d are 96 and 100 for the exclosure and control plots, respectively, and in c, they are 4560 and 4950. Significance was assessed with a t-test
Functional diversity
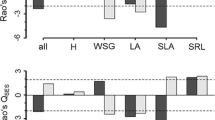
The control plot had a significantly lower LA and chlorophyll content compared with the exclosure plot (Table 1), but other traits (SLA, plant height, and seed mass) were not significantly different. There was no significant difference between the exclosure and control plots in terms of functional diversity (SES) (the mean SES values were −0.09 and 0.09, and the variances were 0.97 and 0.51, respectively).
In terms of the relationship between the SES and the environmental variables, there was a significantly negative correlation between the SES and local crowdedness in the exclosure plot (R = −0.248, P < 0.05) (Table 2; Fig. 3a). However, there were no other significant correlations between the SES and the environmental variables in either plot (Table 2), including local crowdedness in the control plot (R = −0.110, P = 0.285) (Fig. 3b). In the exclosure plot, there was a significantly negative correlation between the environmental variables and Shannon’s diversity (R = −0.317, P < 0.01) (Fig. 4a), but in the control plot, there was no significant correlation between the environmental variables and Shannon’s diversity (R = −0.116, P = 0.254) (Fig. 4b).
Discussion
By focusing on the taxonomic and functional characteristics of plants on the forest floor, we found that deer herbivory played a significant role in shaping local plant communities. Most importantly, using a trait-based approach, we detected changes in the community assembly processes at a local scale, which may explain the mechanisms underlying the patterns of biodiversity.
Taxon- and trait-based evaluation
We compared several metrics of the forest floor plant community between the plots with (exclosure plot) and without (control plot) fences. The significantly lower local crowdedness in the control plot compared with the exclosure plot (Fig. 1d) suggests that deer herbivory played a significant role in suppressing the growth and survival of plants on the forest floor, and the species richness, Shannon’s diversity, and Bray–Curtis dissimilarity values were all higher in the control plot than in the exclosure plot (Fig. 1a–c). Previous reports on the effects of deer herbivory on plant diversity have provided mixed results depending on whether deer primarily consume the dominant species (Côté et al. 2004). In our study site, some dominant species had higher levels of abundance in the exclosure plot than in the control plot (Fig. S1), and the dominance-rank curve of the exclosure plot also showed a more skewed pattern than that of the control plot (Fig. 2). These results indicate that the dominant species with high competitive abilities interfered with the prosperity of the other species in the exclosure plot. Moreover, these results are consistent with the work of Schütz et al. (2003), who reported that a reduction in dominant species due to deer herbivory could enhance plant species richness. Taken together, we suggest that dominance reduction by deer herbivory is one of the important determinants of the maintenance of species diversity, and furthermore, that the lack of herbivory likely decreases the dissimilarity among local assemblages, leading to the homogenization of communities through the disproportionate representation of dominant species. Some studies reported that deer herbivory decreases plant diversity by leaving a limited subset of unpalatable species or, in the extreme case of intensive herbivory, bare soil and little plant cover (Takahashi and Kaji 2001; Rooney et al. 2004; Webster et al. 2005). Although it is difficult to practically compare the impacts of deer over/under-grazing on vegetation across regions, the level of the impacts on our plots may not have been as intense as these extreme cases. While the overabundance of deer has been a concern in the study region (Kaji et al. 2004; The Forestry Agency of Japan 2007, 2012), the intensity of deer herbivory in our study site could be at a level that can, to some extent, maintain plant diversity (as explained by the intermediate disturbance hypothesis; Grime 1973; Connell 1978). In other words, fencing has been affecting the understory plant communities in an adverse way by virtue of unnaturally excluding deer.
To further evaluate the compositional characteristics of forest floor plant communities in response to under- and overgrazing by deer, we used a trait-based approach. Here, we observed higher CWM trait values in LA and chlorophyll content in the exclosure plot than in the control plot (Table 1), which may be due to the selective grazing of large leaves (Knight 2003) and the vulnerability of more nutritious leaves to deer herbivory (Ammer 1996), respectively. Based on the skewed dominance-rank curve, CWM trait values are likely to be highly influenced by dominant species in the exclosure plot, and although the survival of these species cannot be explained by these traits alone, high values of these two traits are thought to be more advantageous under low herbivory pressure. In summary, plants with large leaves and high chlorophyll content are more competitive, but they are also more vulnerable to deer herbivory.
Community assembly processes
Because functional diversity deviated from that expected at random, we examined the community assembly processes at the local scale. In the control plot, the standardized functional diversity values were distributed around zero (Fig. 3b), and there was no significant relationship between standardized functional diversity and the environmental variables (Table 2). These results suggest that deterministic assembly processes, such as interspecific competition or environmental filtering, were weak; that is, stochastic processes predominated in the control plot. When vegetation coverage is reduced by deer, the likelihood of competitive exclusion (i.e., species exclusion by some species with more competitive traits) might decrease, which may increase the opportunities for species establishment (Grime 2006; Mayfield and Levine 2010). The observed pattern in the control plot is consistent with the idea that disturbance promotes random assembly processes (del Moral 2009); when food resources are depleted through selective herbivory, deer are known to graze on plants regardless of their palatability (Waller and Alverson 1997; Takahashi and Kaji 2001; Côté et al. 2004). Therefore, we infer that intensive deer herbivory has reduced vegetation coverage (Fig. 1d), regardless of the species traits at the study site. Stochastic processes often enhance species diversity (e.g., Chase 2010), thereby allowing various species to emerge independently of their traits.
In the exclosure plot, we observed a different pattern of functional diversity along the local crowdedness gradient, i.e., a shift from trait overdispersion to trait clustering (Fig. 3a), which suggests a gradual change in the relative importance of these different assembly processes. The mechanism for trait overdispersion is often interpreted as a consequence of niche partitioning that arises from limiting similarity (Chesson 2000), and in this study, more niches became available to more species as local crowdedness decreased. Niche partitioning promotes the coexistence of various species, thereby resulting in higher species diversity in less crowded localities (Fig. 4a). In terms of trait clustering, a few species became dominant while the majority of species decreased in abundance under fenced (Fig. 2) and crowded conditions, and this could have resulted from intense competition for light and nutrients (Berntson and Wayne 2000). These results suggest that weaker competitors had been excluded by competitively superior species, which led to trait clustering (Mayfield and Levine 2010), and the disappearance of weaker competitors could result in lower species diversity in more crowded localities (Fig. 4a).
In the exclosure plot, only local crowdedness was correlated with the functional diversity patterns of each quadrat; thus, to further understand the mechanisms underlying the functional diversity gradient, we must determine which factors caused the differences in local crowdedness among the localities (i.e., quadrats). Unfortunately, our snapshot datasets did not provide a complete picture, but the monitoring data support our interpretation of the factors that caused the variability in local crowdedness among the localities. According to a report by the Forestry Agency of Japan (2014), the abundance of dominant species has sharply increased since the construction of the fence (Fig. S2), which suggests that crowded localities have continued to expand and that trait clustering is still ongoing.
Implications
Deer overabundance has been caused by human activities, and to mitigate its negative consequences, deer exclosure fences have been constructed around the world with the aim of conserving vegetation. Fences have been reported to be efficient at promoting the regeneration of trees in forests (Shimoda et al. 1994; Nomiya et al. 2003; Kumar et al. 2006), as well as conserving specific species (Fletcher et al. 2001; Tamura et al. 2005) and vegetation ground cover (Kurten and Carson 2015; Tamura 2015). From the perspective of ecosystem-based management, we envision that establishing fences is not an absolute solution, as the importance of considering natural processes has recently been recognized as essential for the conservation of biodiversity and the sustainability of forest ecosystems (Christensen et al. 1996; Lindenmayer and Franklin 2002; Mori 2011). While its impact is often of concern in some situations, deer herbivory is also an inherent process that promotes species coexistence and community assembly through the relaxation of competitive relationships among plant species, and this was reaffirmed by our study. However, our results cannot be generalized because of the lack of spatial and temporal replication, and we were also unable to account for the reproductive status of the individual plants in the community, which could have important consequences for biodiversity conservation. Nonetheless, our results indicate that establishing fences is important if deer herbivory is severely impacting the vegetation. However, to restore and conserve plant diversity, it may be necessary to control the intensity of herbivory by opening/closing the fences, rather than completely eradicating deer herbivory. Therefore, adaptive management and restoration grounded on a mechanistic understanding of ecosystem processes (Mori et al. 2016b) are needed to adequately manage the deer population and its impact on plant diversity.
References
Ammer C (1996) Impact of ungulates on structure and dynamics of natural regeneration of mixed mountain forests in the Bavarian Alps. For Ecol Manage 88:43–53. doi:10.1016/S0378-1127(96)03808-X
Anderson MJ, Crist TO, Chase JM, Vellend M, Inouye BD, Freestone AL, Sanders NJ, Cornell HV, Comita LS, Davies KF, Harrison SP, Kraft NJB, Stegen JC, Swenson NG (2011) Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecol Lett 14:19–28. doi:10.1111/j.1461-0248.2010.01552.x
Angert AL, Huxman TE, Chesson P, Venable DL (2009) Functional tradeoffs determine species coexistence via the storage effect. Proc Natl Acad Sci USA 106:11641–11645. doi:10.1073/pnas.0904512106
Augustine DJ, McNaughton SJ (1998) Ungulate effects on the functional species composition of plant communities: herbivore selectivity and plant tolerance. J Wildl Manage 62:1165–1183. doi:10.2307/3801981
Bardgett RD, Wardle DA (2003) Herbivore-mediated linkages between aboveground and belowground communities. Ecology 84:2258–2268. doi:10.1890/02-0274
Begley-Miller DR, Hipp AL, Brown BH, Hahn M, Rooney TP (2014) White-tailed deer are a biotic filter during community assembly, reducing species and phylogenetic diversity. AoB Plants 6:plu030. doi:10.1093/aobpla/plu030
Berntson GM, Wayne PM (2000) Characterizing the size dependence of resource acquisition within crowded plant populations. Ecology 81:1072–1085. doi:10.1890/0012-9658(2000)081[1072:CTSDOR]2.0.CO;2
Bobek B, Boyce MS, Kosobucka M (1984) Factors affecting red deer (Cervus elaphus) population density in southeastern Poland. J Appl Ecol 21:881–890. doi:10.2307/2405053
Brown TL, Decker DJ, Riley SJ, Enck JW, Lauber TB, Curtis PD, Mattfeld GF (2000) The future of hunting as a mechanism to control white-tailed deer populations. Wildl Soc Bull 28:797–807. doi:10.2307/3783834
Cadotte MW, Carscadden K, Mirotchnick N (2011) Beyond species: functional diversity and the maintenance of ecological processes and services. J Appl Ecol 48:1079–1087. doi:10.1111/j.1365-2664.2011.02048.x
Chase JM (2007) Drought mediates the importance of stochastic community assembly. Proc Natl Acad Sci USA 104:17430–17434. doi:10.1073/pnas.0704350104
Chase JM (2010) Stochastic Community assembly causes higher biodiversity in more productive environments. Science 328:1388–1391. doi:10.1126/science.1187820
Chase JM, Kraft NJB, Smith KG, Vellend M, Inouye BD (2011) Using null models to disentangle variation in community dissimilarity from variation in α-diversity. Ecosphere 2:art24. doi:10.1890/ES10-00117.1
Chesson P (2000) Mechanisms of maintenance of species diversity. Annu Rev Ecol Syst 31:343–366. doi:10.1146/annurev.ecolsys.31.1.343
Chesson P, Kuang JJ (2008) The interaction between predation and competition. Nature 456:235–238. doi:10.1038/nature07248
Christensen NL, Bartuska AM, Brown JH, Carpenter S, D-Antonio C, Francis R, Franklin JF, MacMahon JA, Noss RF, Parsons DJ, Peterson CH, Turner MG, Woodmansee RG (1996) The report of the ecological society of America Committee on the scientific basis for ecosystem management. Ecol Appl 6:665–691. doi:10.2307/2269460
Connell JH (1978) Diversity in tropical rain forests and coral reefs. Science 199:1302–1310
Cornwell WK, Schwilk DW, Ackerly DD (2006) A trait-based test for habitat filtering: convex hull volume. Ecology 87:1465–1471. doi:10.1890/0012-9658(2006)87[1465:ATTFHF]2.0.CO;2
Côté SD, Rooney TP, Tremblay J-P, Dussault C, Waller DM (2004) Ecological impacts of deer overabundance. Annu Rev Ecol Evol Syst 35:113–147. doi:10.1146/annurev.ecolsys.35.021103.105725
Davies TJ, Meiri S, Barraclough TG, Gittleman JL (2007) Species coexistence and character divergence across carnivores. Ecol Lett 10:146–152. doi:10.1111/j.1461-0248.2006.01005.x
De Bello F, Vandewalle M, Reitalu T, Lepš J, Prentice HC, Lavorel S, Sykes MT (2013) Evidence for scale- and disturbance-dependent trait assembly patterns in dry semi-natural grasslands. J Ecol 101:1237–1244. doi:10.1111/1365-2745.12139
Del Moral R (2009) Increasing deterministic control of primary succession on Mount St. Helens, Washington. J Veg Sci 20:1145–1154. doi:10.1111/j.1654-1103.2009.01113.x
Fletcher JD, Shipley LA, McShea WJ, Shumway DL (2001) Wildlife herbivory and rare plants: the effects of white-tailed deer, rodents, and insects on growth and survival of Turk’s cap lily. Biol Conserv 101:229–238. doi:10.1016/S0006-3207(01)00070-2
Forchhammer MC, Stenseth NC, Post E, Langvatn R (1998) Population dynamics of Norwegian red deer: density dependence and climatic variation. Proc R Soc Lond B 265:341–350. doi:10.1098/rspb.1998.0301
Garnier E, Cortez J, Billès G, Navas ML, Roumet C, Debussche M, Laurent G, Blanchard A, Aubry D, Bellmann A, Neill C, Toussaint JP (2004) Plant functional markers capture ecosystem properties during secondary succession. Ecology 85:2630–2637. doi:10.1890/03-0799
Gotelli NJ, Gotelli NJ (2000) Null model analysis of species co-occurrence patterns. Ecology 81:2606–2621. doi:10.1890/0012-9658(2000)081[2606:NMAOSC]2.0.CO;2
Götzenberger L, de Bello F, Bråthen KA, Davison J, Dubuis A, Guisan A, Lepš J, Lindborg R, Moora M, Pärtel M, Pellissier L, Pottier J, Vittoz P, Zobel K, Zobel M (2012) Ecological assembly rules in plant communities-approaches, patterns and prospects. Biol Rev 87:111–127. doi:10.1111/j.1469-185X.2011.00187.x
Grime JP (1973) Competitive exclusion in herbaceous vegetation. Nature 242:344–347. doi:10.1038/242344a0
Grime JP (2006) Trait convergence and trait divergence in herbaceous plant communities: mechanisms and consequences. J Veg Sci 17:255–260. doi:10.1111/j.1654-1103.2006.tb02444.x
Horsley SB, Stout SL, DeCalesta DS (2003) White-tailed deer impact on the vegetation dynamics of a northern hardwood forest. Ecol Appl 13:98–118. doi:10.1890/1051-0761(2003)013[0098:WTDIOT]2.0.CO;2
Hutchinson GE (1959) Homage to Santa Rosalia, or why are there so many kinds of animals? Am Nat 93:145–159. doi:10.1086/282070
Inatomi Y, Uno H, Takashima Y, Onimaru K, Miyaki M, Kaji K (2012) Changes in the understory vegetation due to the decrease in sika deer density in Akan National Park, eastern Hokkaido, Japan. Jap J Conserv Ecol 17:185–197 (in Japanese with English summary)
Kaji K, Okada H, Yamanaka M, Matsuda H, Yabe T (2004) Irruption of a colonizing sika deer population. J Wildl Manage 68:889–899. doi:10.2193/0022-541X(2004)068[0889:IOACSD]2.0.CO;2
Knight TM (2003) Effects of herbivory and its timing across populations of Trillium grandiflorum (Liliaceae). Am J Bot 90:1207–1214. doi:10.3732/ajb.90.8.1207
Kraft NJB, Valencia R, Ackerly DD (2008) Functional traits and niche-based tree community assembly in an Amazonian forest. Science 322:580–582. doi:10.1126/science.1160662
Kraft NJB, Crutsinger GM, Forrestel EJ, Emery NC (2014) Functional trait differences and the outcome of community assembly: an experimental test with vernal pool annual plants. Oikos 123:1391–1399. doi:10.1111/oik.01311
Kubota Y (2000) Spatial dynamics of regeneration in a conifer/broad-leaved forest in northern Japan. J Veg Sci 11:633–640. doi:10.2307/3236570
Kumar S, Takeda A, Shibata E (2006) Effects of 13-year fencing on browsing by sika deer on seedlings on Mt. Ohdaigahara, central Japan. J For Res 11:337–342. doi:10.1007/s10310-006-0216-9
Kunstler G, Lavergne S, Courbaud B, Thuiller W, Vieilledent G, Zimmermann NE, Kattge J, Coomes DA (2012) Competitive interactions between forest trees are driven by species’ trait hierarchy, not phylogenetic or functional similarity: implications for forest community assembly. Ecol Lett 15:831–840. doi:10.1111/j.1461-0248.2012.01803.x
Kurten EL, Carson WP (2015) Do ground-dwelling vertebrates promote diversity in a neotropical forest? Results from a long-term exclosure experiment. Bioscience 65:862–870. doi:10.1093/biosci/biv110
Laliberté E, Legendre P (2010) A distance-based framework for measuring functional diversity from multiple traits. Ecology 91:299–305. doi:10.1890/08-2244.1
Lavorel S, McIntyre S, Landsberg J, Forbes TDA (1997) Plant functional classifications: from general groups to specific groups based on response to disturbance. Trends Ecol Evol 12:474–478. doi:10.1016/S0169-5347(97)01219-6
Lindenmayer DB, Franklin JF (2002) Conserving forest biodiversity: a comprehensive multiscaled approach. Island Press, Washington
Loison A, Langvatn R, Solberg EJ (1999) Body mass and winter mortality in red deer calves: disentangling sex and climate effects. Ecography 22:20–30. doi:10.1111/j.1600-0587.1999.tb00451.x
MacArthur RH (1972) Geographical ecology. Patterns in the distribution of species. Princeton University Press, Princeton
MacArthur RH, Levins R (1967) The limiting similarity, convergence, and divergence of coexisting species. Am Nat 101:377–385. doi:10.1086/282505
Magurran AE, Mcgill BJ (2011) Biological diversity: frontiers in measurement and assessment, 1st edn. Oxford University Press, Oxford
Markwell J, Blevins D (1999) The Minolta SPAD-502 leaf chlorophyll meter: an exciting new tool for education in the plant sciences. Am Biol Teach 61:672–676. doi:10.2307/4450800
Mayfield MM, Levine JM (2010) Opposing effects of competitive exclusion on the phylogenetic structure of communities. Ecol Lett 13:1085–1093. doi:10.1111/j.1461-0248.2010.01509.x
Mori AS (2011) Ecosystem management based on natural disturbances: hierarchical context and non-equilibrium paradigm. J Appl Ecol 48:280–292. doi:10.1111/j.1365-2664.2010.01956.x
Mori A, Takeda H (2003) Architecture and neighbourhood competition of understorey saplings in a subalpine forest in central Japan. Ecoscience 10:217–224. doi:10.1080/11956860.2003.11682769
Mori AS, Furukawa T, Sasaki T (2013) Response diversity determines the resilience of ecosystems to environmental change. Biol Rev 88:349–364. doi:10.1111/brv.12004
Mori AS, Shiono T, Haraguchi TF, Ota AT, Koide D, Ohgue T, Kitagawa R, Maeshiro R, ToeToeAung Nakamori T, Hagiwara Y, Matsuoka S, Ikeda A, Hishi T, Hobara S, Mizumachi E, Frisch A, Thor G, Fujii S, Osono T, Gustafsson L (2015) Functional redundancy of multiple forest taxa along an elevational gradient: predicting the consequences of non-random species loss. J Biogeogr 42:1383–1396. doi:10.1111/jbi.12514
Mori AS, Isbell F, Fujii S, Makoto K, Matsuoka S, Osono T (2016a) Low multifunctional redundancy of soil fungal diversity at multiple scales. Ecol Lett 249–259. doi:10.1111/ele.12560
Mori AS, Lertzman K, Gustafsson L (2016b) Biodiversity and ecosystem services in forest ecosystems: a research agenda for applied forest ecology. J Appl Ecol. doi:10.1111/1365-2664.12669
Mouillot D, Graham NAJ, Villéger S, Mason NWH, Bellwood DR (2013) A functional approach reveals community responses to disturbances. Trends Ecol Evol 28:167–177. doi:10.1016/j.tree.2012.10.004
Nomiya H, Suzuki W, Kanazashi T, Shibata M, Tanaka H, Nakashizuka T (2003) The response of forest floor vegetation to deer exclusion and disturbance in a riparian deciduous forest, Central Japan. Plant Ecol 164:263–276. doi:10.1023/A:1021294021438
Ohashi H, Hoshino Y (2014) Disturbance by large herbivores alters the relative importance of the ecological processes that influence the assembly pattern in heterogeneous meta-communities. Ecol Evol 4:766–775. doi:10.1002/ece3.987
Pérez-Harguindeguy N, Díaz S, Garnier E, Lavorel S, Poorter H, Jaureguiberry P, Bret-Harte MS, Cornwell WK, Craine JM, Gurvich DE, Urcelay C, Veneklaas EJ, Reich PB, Poorter L, Wright IJ, Ray P, Enrico L, Pausas JG, De Vos AC, Buchmann N, Funes G, Quétier F, Hodgson JG, Thompson K, Morgan HD, Ter Steege H, Van Der Heijden MGA, Sack L, Blonder B, Poschlod P, Vaieretti MV, Conti G, Staver AC, Aquino S, Cornelissen JHC (2013) New handbook for standardised measurement of plant functional traits worldwide. Aust J Bot 61:167–234. doi:10.1071/BT12225
Petchey OL, Gaston KJ (2007) Dendrograms and measuring functional diversity. Oikos 116:1422–1426. doi:10.1111/j.2007.0030-1299.15894.x
Rasband WS (1997–2008) ImageJ. US National Institutes of Health, Bethesda, Maryland, USA
Reich PB, Walters MB, Ellsworth DS (1997) From tropics to tundra: global convergence in plant functioning. Proc Natl Acad Sci USA 94:13730–13734. doi:10.1073/pnas.94.25.13730
Riley SJ, Decker DJ, Enck JW, Curtis PD, Lauber TB, Brown TL (2003) Deer populations up, hunter populations down: implications of interdependence of deer and hunter population dynamics on management. Ecoscience 10:455–461. doi:10.1080/11956860.2003.11682793
Rooney TP (2001) Deer impacts on forest ecosystems: a North American perspective. Forestry 74:201–208. doi:10.1093/forestry/74.3.201
Rooney TP, Waller DM (2003) Direct and indirect effects of white-tailed deer in forest ecosystems. For Ecol Manage 181:165–176. doi:10.1016/S0378-1127(03)00130-0
Rooney TP, Wiegmann SM, Rogers DA, Waller DM (2004) Biotic impoverishment and homogenization in unfragmented forest understory communities. Conserv Biol 18:787–798. doi:10.1111/j.1523-1739.2004.00515.x
Roseberry JL, Woolf A (1998) Habitat-population density relationships for white-tailed deer in Illinois. Wildl Soc Bull 26:252–258
Russell FL, Zippin DB, Fowler NL (2001) Effects of white-tailed deer (Odocoileus virginianus) on plants, plant populations and communities: a review. Am Midl Nat 146:1–26. doi:10.1674/0003-0031(2001)146[0001:EOWTDO]2.0.CO;2
Sammul M, Oksanen L, Mägi M (2006) Regional effects on competition-productivity relationship: a set of field experiments in two distant regions. Oikos 112:138–148. doi:10.1111/j.0030-1299.2006.13378.x
Schütz M, Risch AC, Leuzinger E, Krüsi BO, Achermann G (2003) Impact of herbivory by red deer (Cervus elaphus L.) on patterns and processes in subalpine grasslands in the Swiss National Park. For Ecol Manage 181:177–188. doi:10.1016/S0378-1127(03)00131-2
Shimoda K, Kimura K, Kanzaki M, Yoda K (1994) The regeneration of pioneer tree species under browsing pressure of Sika deer in an evergreen oak forest. Ecol Res 9:85–92. doi:10.1007/BF02347245
Snedecor GW, Cochran WG (1967) Statistical methods, 6th edn. Iowa State University Press, Ames
Sokal RR, Rohlf FJ (2012) Biometry, 4th edn. Freeman and Company, New York
Takahashi H, Kaji K (2001) Fallen leaves and unpalatable plants as alternative foods for sika deer under food limitation. Ecol Res 16:257–262. doi:10.1046/j.1440-1703.2001.00391.x
Takatsuki S (2009) Effects of sika deer on vegetation in Japan: a review. Biol Conserv 142:1922–1929. doi:10.1016/j.biocon.2009.02.011
Tamura A (2015) Potential of soil seed banks in the ecological restoration of overgrazed floor vegetation in a cool-temperate old-growth damp forest in eastern Japan. J For Res 21:43–56. doi:10.1007/s10310-015-0509-y
Tamura A, Irino A, Yamane M, Katsuyama T (2005) Protection of endangered plants from sika deer herbivory using deer-proof fences in the Tanzawa Mountains, central Japan. Jap J Conserv Ecol 10:11–17 (in Japanese with English summary)
The Forestry Agency of Japan, Hokkaido Regional Forest Office (2004) The conservation and regeneration project of forestry ecosystems at Shiretoko: the research report (in Japanese). http://dc.shiretoko-whc.com/data/research/report/h15/H15shinrin-seitaikei.pdf
The Forestry Agency of Japan, Hokkaido Regional Forest Office (2007) The conservation and management program of ezo-shika deer in Shiretoko (in Japanese). http://shiretoko-whc.com/data/management/kanri/shikawg_H19_jikkou.pdf
The Forestry Agency of Japan, Hokkaido Regional Forest Office (2012) The conservation and management program of ezo-shika deer in Shiretoko, second term (in Japanese). http://shiretoko-whc.com/data/management/kanri/ezoshika_kanri.pdf
The Forestry Agency of Japan, Hokkaido Regional Forest Office (2014) The conservation and regeneration project of forestry ecosystems at Shiretoko (broad research) (in Japanese). http://www.rinya.maff.go.jp/hokkaido/policy/business/pr/siritoko_wh/pdf/h25houkokusyo.pdf
Tilman D (1982) Resource competition and community structure. Princeton University Press, Princeton
Tilman D (1988) Plant strategies and the dynamics and structure of plant communities. Princeton University Press, Princeton
Vellend M, Verheyen K, Flinn KM, Jacquemyn H, Kolb A, Van Calster H, Peterken G, Graae BJ, Bellemare J, Honnay O, Brunet J, Wulf M, Gerhardt F, Hermy M (2007) Homogenization of forest plant communities and weakening of species-environment relationships via agricultural land use. J Ecol 95:565–573. doi:10.1111/j.1365-2745.2007.01233.x
Vojtech E, Loreau M, Yachi S, Spehn EM, Hector A (2008) Light partitioning in experimental grass communities. Oikos 117:1351–1361. doi:10.1111/j.2008.0030-1299.16700.x
Waller DM, Alverson WS (1997) The white-tailed deer: a keystone herbivore. Wildl Soc Bull 25:217–226
Wardle DA, Bonner KI, Barker GM (2002) Linkages between plant litter decomposition, litter quality, and vegetation responses to herbivores. Funct Ecol 16:585–595. doi:10.1046/j.1365-2435.2002.00659.x
Warton DI, Wright IJ, Falster DS, Westoby M (2006) Bivariate line-fitting methods for allometry. Biol Rev 81:259–291. doi:10.1017/S1464793106007007
Webster CR, Jenkins MA, Rock JH (2005) Long-term response of spring flora to chronic herbivory and deer exclusion in Great Smoky Mountains National Park, USA. Biol Conserv 125:297–307. doi:10.1016/j.biocon.2005.03.027
Weiher E, Freund D, Bunton T, Stefanski A, Lee T, Bentivenga S (2011) Advances, challenges and a developing synthesis of ecological community assembly theory. Philos Trans R Soc Lond B Biol Sci 366:2403–2413. doi:10.1098/rstb.2011.00
Acknowledgments
We thank Y. Ishikawa, M. Mimura, T. Sasaki, S. Fujii, D. Koide, S. Qian, T. Ohgue, M. Kasahara, and T. Okimura for their contributions to this study. Logistical support for the field study was provided by the Shiretoko Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was supported by the Mitsui and Co., Ltd. Environment Fund and the Japanese Ministry of Education, Culture, and Sports (Grant Number 23770083).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Nishizawa, K., Tatsumi, S., Kitagawa, R. et al. Deer herbivory affects the functional diversity of forest floor plants via changes in competition-mediated assembly rules. Ecol Res 31, 569–578 (2016). https://doi.org/10.1007/s11284-016-1367-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-016-1367-6