Abstract
The application of advanced technologies to the study of little-known species is a necessary step in generating effective conservation strategies. Despite the biological importance of the small carnivore guild, a paucity of data exists in terms of the spatial ecology of these species, largely due to logistical constraints of large and bulky collar units. This study reports the first known satellite collaring of a viverrid, the Malay civet (Viverra tangalunga), in Sabah, Malaysian Borneo. Stationary tests of two generations of 65–70 g e-obs GmbH ‘Collar 1A’ units recorded high fix success rates and good accuracy and precision under semi-open canopy. From October 2013–August 2015, nine adult V. tangalunga were fit with e-obs collars recording hourly nocturnal GPS locations. Collars were successfully deployed for 27–187 days. Field GPS fix success varied from 22 to 88.3 %, with the study documenting a total GPS success of 58.1 % across all individuals. Despite this large in-field performance range, the quality and quantity of data collected by these units surpass that of previous VHF studies on Asian viverrids, collecting on average a 16-fold increase in locations per collaring day. The successful application of satellite technology to these little-known carnivores carries significant biological and conservation implications, and it is recommended that satellite collars are a viable technology to conduct detailed and well-designed ecological studies of Viverridae species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Global biodiversity loss is progressing at increasingly alarming rates (Schipper et al. 2008; Barnosky et al. 2011; Ceballos et al. 2015; Ripple et al. 2015). To mitigate further loss, effective conservation management plans are critical, which in turn require in-depth understandings of species’ biological requirements (Margules and Pressey 2000; Chetkiewicz et al. 2006; Cooke 2008). Documenting and quantifying factors crucial to species survival are the central aims of applied wildlife research, and the design of innovative research tools facilitates the achievement of these goals.
Remote tracking technologies in wildlife studies have revolutionized scientific understanding of animal behavioural patterns and processes (Cooke et al. 2004; Ropert-Coudert and Wilson 2005; Fuller and Fuller 2012). The application of radio telemetry as very high frequency (VHF) collar transmitters allowed for the first remote documentation of animal movements in the 1960s (Craighead et al. 1963). While revolutionary, VHF tracking often requires significant field effort for sparse and relatively inaccurate data (Recio et al. 2011a; Gitzen et al. 2013). Furthermore, the collection, applicability, and quality of VHF data are limited by intrinsic biases such as observer presence, site remoteness, weather, and specific animal behaviours (Fuller et al. 2005; Recio et al. 2011b).
In response to these limitations, satellite-based tracking technologies, such as the global positioning system (GPS), were first applied to wildlife in the 1970s (Craighead et al. 1972). This development meant the collection of larger, more consistent, fine-scaled and accurate datasets (Rodgers 2001; Kochanny et al. 2002). Satellite tracking minimizes logistical effort and eliminates the influence of observer presence on recorded behavioural patterns, generating datasets otherwise unobtainable by VHF tracking and more relevant to conservation actions (Hebblewhite and Haydon 2010). Satellite collars have established habitat utilisation and preferences for wary and remote species (Amstrup et al. 2004; Simcharoen et al. 2014), uncovered areas of previously unknown reproductive significance in widely migrating species (Lindsell et al. 2009; Schofield et al. 2009; Hays et al. 2014), and discovered novel cryptic behaviours (Davis et al. 1999; Bandeira de Melo et al. 2007; Lührs and Kappeler 2013).
However, the universal application of GPS to wildlife tracking is still limited by technological constraints, as historically, both transmitters and batteries have been bulky and large (>400 g, some up to 2.2 kg) (Rodgers 2001). Most terrestrial GPS studies have focused on mammals >7 kg, so spatial research on small and medium-sized species relies on VHF transmitters (Blackie 2010; Cagnacci et al. 2010). Owing to recent advancements in both battery longevity and the miniaturization of GPS component design, long-term satellite technologies are being applied to increasingly smaller mammalian species, such as the ocelot Leopardus pardalis (Haines et al. 2006), European hedgehog Erinaceus europaeus (Recio et al. 2011c), fossa Cryptoprocta ferox (Lührs and Kappeler 2013), fisher Martes pennanti (Brown et al. 2012), and brushtail possum Trichosurus vulpecula (Blackie 2010; Dennis et al. 2010).
The Viverridae family (Order Carnivora) comprises 34 species in 14 genera, the majority of which weigh <8 kg (Jennings and Veron 2009). There exists a significant paucity of data concerning even basic ecological information of this family (Schreiber et al. 1989), and five viverrid species are not yet represented in peer-reviewed literature (Brooke et al. 2014 ). What studies do occur are dominated by camera trap deployments (Wilting et al. 2010; Jennings et al. 2015), survey transects (Heydon and Bulloh 1996; Iseborn et al. 2010), and a handful of VHF studies (Joshi et al. 1995; Grassman 1998; Grassman et al. 2005a; Berhanu et al. 2013; Camps and Alldredge 2013; Nakashima et al. 2013). Viverrids are threatened by habitat loss and hunting, but also by lack of scientific information regarding rudimentary survival parameters (Schipper et al. 2008; Brooke et al. 2014). This knowledge gap poses a substantial threat to the effective conservation and management of these species.
Therefore, this study sought to demonstrate the applicability, performance, and value of satellite tracking a model viverrid, the Malay civet (Viverra tangalunga). Weighing between 3–7 kg, V. tangalunga is a predominantly solitary and terrestrial small carnivore found throughout the Sundaic region of Southeast Asia (Payne and Francis 1985). Although several VHF studies have evaluated the spatial ecology of V. tangalunga (Nozaki et al. 1994; Colón 2002; Jennings et al. 2006, 2010), this study represents the first known application of GPS collars to the Viverridae family, and ultimately aimed to demonstrate the scientific and conservation value of GPS technology deployments on small carnivores.
Methods
Study site
The Lower Kinabatangan Floodplain is located in eastern Sabah, Malaysian Borneo (approximate range: 5°18′N to 5°42′N and 117°54′E to 118°33′E). The climate is humid tropical with temperatures ranging from 21 to 34 °C (Ancrenaz et al. 2004). This study was based in the Lower Kinabatangan Wildlife Sanctuary (LKWS), a 270 km2 area of protected secondary forest flanking the Kinabatangan River (Ancrenaz et al. 2004; Goossens et al. 2005). Comprised of ten riparian lots of varying degrees of disturbance history, the sanctuary contains a mixture of dry lowland, semi-inundated, and swamp forests interspersed with small grasslands (Abram et al. 2014).
Trapping and immobilization
Trapping periods spanned October 2013–August 2015. Small carnivores were trapped using locally constructed, specially designed box treadle traps (110 cm × 35 cm × 40 cm). Traps were set by 18h00, checked between 07h00 and 08h00, and closed during the day to avoid non-target diurnal captures.
Upon capture, animals were administered anesthesia by a qualified veterinarian. Animals were either sedated with Tiletamine/Zolazepam (Zoletil™, Virbac Laboratories, Carros, France), or a mixture of Ketamine (Narketan™, Vétoquinol UK Limited, Buckingham, UK), Xylazine (Ilium Xylazil™, Troy Laboratories PTY Limited, Glendenning, Australia) and Tiletamine/Zolazepam. In the case of the latter drug combination, the reversal agent Yohimbine (Reverzine™, Bomac Pty Limited, Hornsby, Australia) was administered once sampling was complete.
GPS collaring and tracking
GPS collars (Collar 1A and second-generation Collar 1A, e-obs GmbH, Grünwald, Germany) weighed 65–70 g, or between 1.3–1.4 % of average adult civet body weight, well within the restrictions of the American Mammal Society for the ethical collaring of animals (Sikes and Gannon 2011). Each tag contained a GPS microchip, either a 2300 or 2500 mAh battery, UHF radiotransmitter, a tri-axial accelerometer, and an antenna. The collar was constructed to degrade through a thin section of leather near the fastening, and efforts were made to retrieve the collar following the cessation of data collection.
Civets are nocturnal, and utilize day bed resting sites in dense forest or grassy areas (Colón 2002; Jennings et al. 2006). To conserve tag battery life and obtain the highest resolution of relevant GPS fixes, collars were set to record 13 hourly GPS points from 18h00 until 06h00. Collars were programmed with a 150 s ‘GPS timeout’, whereby the unit would cease searching for available satellites if no successful fix was acquired within this limit. For each successful GPS point, the collar recorded longitude, latitude, date, time, time to first fix, battery voltage, temperature, speed estimate, and heading.
Due to the small size of the tags, a remote ultra-high frequency (UHF) download was required to access GPS and activity sensor data. A hand-held device (BaseStation II, e-obs GmbH) needed to be within transmittable range of a tagged individual, following which a high-speed wireless radio-link would be established and logged data downloaded to the BaseStation.
Collars emitted a UHF radio signal for two and a half hours daily to allow for tracking. Collared individuals were tracked using a UHF 7E 868 MHz Yagiantenna (e-obs GmbH), an AOR AR8200 handscanner radio receiver (AOR Ltd, Tokyo, Japan), and the BaseStation. For most data download events, animals were tracked via the UHF pinger until adequate tag proximity (~50–150 m) was reached for data download to commence.
Data extraction occurred through the use of DataDecoder software v5_1s6 and v7_1 (e-obs GmbH), which converted encrypted BaseStation data into a desired file format. These were imported into MS Excel and ArcGIS 10.1 for subsequent analysis.
Collar stationary performance
Prior to deployment, three first-generation and four second-generation collars were tested at a stationary reference point under semi-closed canopy for a 13-h nocturnal period. Collar performance was determined by proportion of successful fix attempts. Accuracy was measured as Euclidean distance between collar fixes and the unit’s true location, as recorded by a GPSmap 62 GPS (Garmin Ltd., Kansas, USA). The precision of fixes was evaluated by measuring the Euclidean inter-point distances between each collar’s hourly fixes. Each collar’s maximum linear error was determined as the greatest inter-point distance, representing effective point spread, collected throughout this trial period.
Analysis
All spatial analyses were carried out with ArcGIS software (Version 10.1, ESRI, Inc., Redlands, CA). In stationary accuracy tests, data were log transformed for normality, and a pooled, two-tailed, two-sample t test was used to evaluate if linear error differed by collar generation. A Mann–Whitney U-test determined whether linear inter-point differences differed by collar generation. Maximum inter-point distances between generations were tested by a pooled, two-tailed, two-sample t-test. For field-deployed collar data, in-field performance values were pooled regardless of collar generation. In order to quantify tracking data resolution per collaring effort, a mean value of location fixes per active collaring day was calculated for this study and other viverrid VHF research. The total number of successfully recorded locations was divided by the maximum active collaring days for each animal in each study, and a two-tailed, two-sample t-test compared the log-transformed mean fix values between GPS and VHF studies.
Results
Trapping
From October 2013–August 2015, 731 trap nights (night × number of active traps) were conducted throughout the LKWS, resulting in 43 small carnivore captures (5.9 % trapping success rate). Of these, 27 unique Malay civets were captured, and nine of these individuals collared (eight males, one female).
Collar stationary performance
Of the collars tested, fix success was high and location error slight, with second-generation collars outperforming first in most parameters. First-generation units demonstrated a mean (±SE) fix success of 87.2 % ± 5.13 (n = 39), while second-generation collars obtained all attempted fixes (n = 50).
In terms of collar accuracy, mean (±SE) Euclidean distance error between GPS fixes and true location was 21.0 m ± 2.60 for first-generation units (n = 34). Second-generation collars displayed a significantly lower mean horizontal distance error of 12.3 m ± 1.87, nearly half as large as that of the first-generation units (n = 50) (t = 3.9225, d.f. = 82, P < 0.001). Collar precision improved between first- (n = 177) and second-generation units (n = 288), with each collecting median inter-point distances of 19.65 m and 12.67 m, respectively (U = 33749, P < 0.01e-6). However, the mean (±SE) maximum linear point spread of first-generation collars [75.9 m ± 17.7 (n = 3)] did not significantly vary from that recorded by second-generation units [53.3 m ± 14.7 (n = 4), t = 0.9926, d.f. = 5, P = 0.3665].
Collar field performance
From October 2013–August 2015, nine Malay civets (eight males and one female) were fit with GPS collars. Data were successfully retrieved from seven of these nine individuals (Table 1). Four animals were recaptured following completion of the data collection period, and no change was documented in body condition or weight, although minor hair loss was noted.
In total, 8,450 GPS fixes were attempted by all collars, with 4906 successfully obtaining latitude and longitude, a total fix success of 58.1 %, with individual collar performances ranging from 22.0 to 88.3 %. Collar longevity reached a maximum of 187 days before battery exhaustion. The shortest complete deployment occurred when a collar was prematurely shed after 27 nights of data collection.
Collars collected a mean (±SE) 8.1 ± 1.7 fix locations per active collaring day, representing a significantly greater maximum data resolution than of that collected by VHF studies on Asian viverrids (Table 2; t = 11.604, d.f. = 36, P < 0.01e-10).
Discussion
The recent downsizing of satellite tracking technologies has allowed a greater diversity of species to be collared with the aim of providing accurate and high-resolution spatiotemporal data. This study demonstrated the successful preliminary performance of small satellite collars, both in stationary tests and deployed upon a novel small carnivore, the Malay civet.
Stationary test performance parameters of both first- and second-generation collars were comparable to the high fix success rates, accuracy, and precision values of other small satellite unit performance studies (Cain et al. 2005; Jiang et al. 2008; Dennis et al. 2010; LaPoint et al. 2013). The error associated with triangulation-based VHF tracking can be upwards of 200 m, so this slight GPS error is of little comparative consequence (Grassman et al. 2005b; Bartolommei et al. 2012; Nakashima et al. 2013). These stationary trials demonstrate the much higher quality of GPS compared to VHF data in small carnivore research.
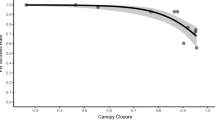
This study reports the first known satellite collaring of a Viverridae species. Overall fix success rate was similar to the performances of other small, field-deployed GPS units (Haines et al. 2006; Blackie 2010; Recio et al. 2010; Brown et al. 2012). Large variability of GPS fix success rates between tracked individuals (22.0–88.3 %) is a commonly documented trend in satellite tracking, and could be attributed to the specific behaviours of collared animals (Blackie 2010; Mattisson et al. 2010; Recio et al. 2011a). Collar-bearing civets utilised a range of microhabitat types, with some individuals (Males 3, 4, 6–8) residing in areas of greater undergrowth and canopy densities than others (Males 1, 2). These environmental factors can influence fix success rates, and should be taken into consideration when designing and analyzing studies on tropical terrestrial species (Rempel et al. 1995; Mattisson et al. 2010; Gitzen et al. 2013).
The volume and fine-scale spatiotemporal resolution of data collected by satellite collars exceeded that of Asian VHF-based Viverridae studies, with satellite units collecting, on average, more than 16-times more locations per sampling day than traditional radio-telemetry methods. This increased data resolution allows for more biologically rigorous questions to be answered on wary and cryptic species in demanding field conditions, without the negative effects of VHF observer bias, and for significantly less effort per resultant fix. Positive results from this first civet GPS collaring, along with the high quality and resolution of stationary trial data, confirms this method as a strong alternative to traditional VHF telemetry tracking for small carnivore research.
Despite these successful first deployments, it is of value to note a design limitation with this brand of satellite units. There is currently no standardized internal calibration value of GPS fix quality in these collars, such as a dilution of precision (DOP), a common component of most unit design. These values facilitate the development of post hoc data screening protocols in order to increase the biological relevancy of acquired GPS data (Lewis et al. 2007; Frair et al. 2010).
Study designs must carefully consider the objectives of their research before deciding upon the deployment of satellite units, as several shortcomings must be overcome before the technology becomes a standard method in small carnivore research (Cooke 2008). While collars utilised in this study are currently the smallest known mammalian long-term GPS units, 65 g is still much too large for certain species. For those species large enough to bear a tag, battery life can be an issue, such that long-term behavioural patterns are unable to be determined. The balance between data resolution and study longevity must be considered when planning studies (Kochanny et al. 2002; Land et al. 2006; Tomkiewicz et al. 2010).
Furthermore, GPS hardware is currently more expensive than VHF transmitters (Rodgers 2001). This high initial cost can pose a logistical barrier to projects, and researchers must not sacrifice statistical power in the blind pursuit of advanced techniques. Many scientists discuss that for certain research questions, analytical rigor is strengthened more so by an increase in the number of individuals tracked than by the number of locations per animal (Hebblewhite and Haydon 2010). Researchers must then carefully consider their specific study aims, budget, focal species, and working conditions to strike a balance between the deployment of several expensive GPS units vs. a larger number of VHF collars, as this study demonstrated. Additionally, when GPS collar failures do occur, they are considerably more costly in both value and relative data loss than VHF failures. A large amount of GPS collar failures have been documented in field-deployed scenarios (Blake et al. 2001; Johnson et al. 2002; Gau et al. 2004; Hebblewhite et al. 2007; Blackie 2010). In this study alone, 22 % (n = 2) of deployed GPS collars failed. Research must then be further prepared to accommodate the costs, both monetary and scientific, of such occurrences with robust study designs.
Despite these considerations, the resolution and quality of GPS data remain irrefutable advantages of the technology. This study highlights the value of GPS collars for small carnivore research due to the data accuracy, quantity and quality otherwise unobtainable in this study area with alternative methods such as VHF or camera trap surveys. The successful application of GPS collars to V. tangalunga generated a large dataset of fine-scale spatiotemporal information useful for identifying landscape features important for the persistence of small tropical carnivores. This information can begin to fill the knowledge gaps currently afflicting the guild, such that informed and effective conservation management plans might be drafted. Given a well-crafted study and sufficient funds, small carnivore scientists can utilize satellite technologies to explore the next frontier in understanding and conserving this unique guild.
References
Abram NK, Xofix P, Tzanopoulos J, MacMillan DC, Ancrenaz M, Chung R, Peter L, Ong R, Lackman I, Goossens B, Ambu L, Knight AT (2014) Synergies for improving oil palm production and forest conservation in floodplain landscapes. PLoS One 9:e95388. doi:10.1371/journal.pone.0095388
Amstrup SC, Mcdonald TL, Durner GM (2004) Using satellite radiotelemetry data to delineate and manage wildlife populations. Wildl Soc Bull 32:661–679. doi: 10.2193/0091-7648(2004)032[0661:USRDTD]2.0.CO;2
Ancrenaz M, Goossens B, Gimenez O, Sawang A, Lackman-Ancrenaz I (2004) Determination of ape distribution and population size using ground and aerial surveys: a case study with orang-utans in lower Kinabatangan, Sabah, Malaysia. Anim Conserv 7:375–385. doi:10.1017/S136794300400157X
Bandeira de Melo LF, Lima Sábato MA, Vaz Magni EM, Young RJ, Coelho CM (2007) Secret lives of maned wolves (Chrysocyon brachyurus Illiger 1815): as revealed by GPS tracking collars. J Zool 271:27–36. doi:10.1111/j.1469-7998.2006.00176.x
Barnosky AD et al (2011) Has the Earth’s sixth mass extinction already arrived? Nature 471:51–57. doi:10.1038/nature09678
Bartolommei P, Francucci S, Pezzo F (2012) Accuracy of conventional radio telemetry estimates: a practical procedure of measurement. Hystrix 23:12–18. doi:10.4404/hystrix-23.2-6376
Berhanu A, Bekele A, Balakrishnan M (2013) Home range and movement patterns of African Civet Civettictis civetta in Wondo Genet, Ethiopia. Small Carnivore Conserv 48:83–86
Blackie HM (2010) Comparative performance of three brands of lightweight global positioning system collars. J Wildl Manage 74:1911–1916. doi:10.2193/2009-412
Blake S, Douglas-Hamilton I, Karesh WB (2001) GPS telemetry of forest elephants in Central Africa: results of a preliminary study. Afr J Ecol 39:178–186. doi:10.1046/j.1365-2028.2001.00296.x
Brooke ZM, Bielby J, Nambiar K, Carbone C (2014) Correlates of research effort in carnivores: body size, range size and diet matter. PLoS One 9:e93195. doi:10.1371/journal.pone.0093195
Brown DD, LaPoint S, Kays R, Heidrich W, Kümmeth F, Wikelski M (2012) Accelerometer-informed GPS telemetry: reducing the trade-off between resolution and longevity. Wildl Soc Bull 36:139–146. doi:10.1002/wsb.111
Cagnacci F, Boitani L, Powell RA, Boyce MS (2010) Animal ecology meets GPS-based radiotelemetry: a perfect storm of opportunities and challenges. Phil Trans R Soc B 365:2157–2162. doi:10.1098/rstb.2010.0107
Cain JWI, Krausman PR, Jansen BD, Morgart JR (2005) Influence of topography and GPS fix interval on GPS collar performance. Wildl Soc Bull 33:926–934. doi: 10.2193/0091-7648(2005)33[926:IOTAGF]2.0.CO;2
Camps D, Alldredge JR (2013) Multi-scale habitat use and selection of common genet Genetta genetta (Viverridae, Carnivora) in a Mediterranean environment. Mammalia 77:285–295. doi:10.1515/mammalia-2012-0023
Ceballos G, Ehrlich PR, Barnosky AD, Garcia A, Pringle RM, Palmer TM (2015) Accelerated modern human-induced species losses: entering the sixth mass extinction. Sci Adv 1:e1400253. doi:10.1126/sciadv.1400253
Chetkiewicz CB, St. Clair CC, Boyce MS (2006) Corridors for conservation: integrating pattern and process. Annu Rev Ecol Evol Syst 37:317–342. doi:10.1146/annurev.ecolsys.37.091305.110050
Colón CP (2002) Ranging behaviour and activity of the Malay civet (Viverra tangalunga) in a logged and an unlogged forest in Danum Valley, East Malaysia. J Zool 257:473–485. doi:10.1017/S0952836902001073
Cooke SJ (2008) Biotelemetry and biologging in endangered species research and animal conservation: relevance to regional, national, and IUCN Red List threat assessments. Endanger Species Res 4:165–185. doi:10.3354/esr00063
Cooke SJ, Hinch SG, Wikelski M, Andres RD, Kuchel LJ, Wolcott TG, Butler PJ (2004) Biotelemetry: a mechanistic approach to ecology. Trends Ecol Evol 19:334–343. doi:10.1016/j.tree.2004.04.003
Craighead FC, Craighead JJ, Davies RS (1963) Radiotracking grizzly bears. In: Slater LE (ed) Biotelemetry: the use of telemetry in animal behavior and physiology in relation to ecological problems. Macmillan, New York, pp 133–148
Craighead FC, Craighead JJ, Cote CE, Buechner HK (1972) Satellite and ground radio tracking of elk. In: Galler S et al (eds) Animal orientation and navigation. NASA, Washington DC, pp 99–111
Davis RW, Fuiman LA, Williams TM, Collier SO, Hagey WP, Kanatous SB, Kohin S, Horning M (1999) Hunting behavior of a marine mammal beneath the Antarctic fast ice. Science 283:993–996. doi:10.1126/science.283.5404.993
Dennis TE, Chen WC, Koefoed IM, Lacoursiere CJ, Walker MM, Laube P, Forrer P (2010) Performance characteristics of small global-positioning-system tracking collars for terrestrial animals. Wildl Biol Pract 6:14–31. doi:10.2461/wbp.2010.6.2
Frair JL, Fieberg J, Hebblewhite M, Cagnacci F, DeCesare NJ, Pedrotti L (2010) Resolving issues of imprecise and habitat-biased locations in ecological analyses using GPS telemetry data. Phil Trans R Soc B 365:2187–2200. doi:10.1098/rstb.2010.0084
Fuller MR, Fuller TK (2012) Radio-telemetry equipment and applications for carnivores. In: Boitani L, Powell RA (eds) Carnivore ecology and conservation: a handbook of techniques. Oxford University Press, Oxford, pp 152–168
Fuller MR, Millspaugh JJ, Church KE, Kenward RE (2005) Wildlife radio telemetry. In: Braun CE (ed) Techniques for wildlife investigations and management, 6th edn. The Wildlife Society, Bethesda, pp 377–417
Gau RJ, Mulders R, Ciarnello LM, Heard DC, Chetkiewicz CL, Boyce MS, Munro R, Stenhouse G, Chruszcz B, Gibeau ML, Milakovic B, Parker KL (2004) Uncontrolled field performance of Televilt GPS-SimplexTM collars on grizzly bears in western and northern Canada. Wildl Soc Bull 32:693–701. doi: 10.2193/0091-7648(2004)032[0693:UFPOTG]2.0.CO;2
Gitzen RA, Belant JL, Millspaugh JJ, Wong ST, Hearn AJ, Ross J (2013) Effective use of radiotelemetry for studying tropical carnivores. Raff Bull Zool 28:67–83
Goossens B, Chikhi L, Jalil MF, Ancrenaz M, Lackman-Ancrenaz I, Mohamed M, Andau P, Bruford MW (2005) Patterns of genetic diversity and migration in increasingly fragmented and declining orang-utan (Pongo pygmaeus) populations from Sabah, Malaysia. Mol Ecol 14:441–456. doi:10.1111/j.1365-294X.2004.02421.x
Grassman LI (1998) Movements and fruit selection of two Paradoxurinae species in a dry evergreen forest in Southem Thailand. Small Carnivore Conserv 19:25–29
Grassman LI, Tewes ME, Silvy NJ (2005a) Ranging, habitat use and activity patterns of binturong Arctictis binturong and yellow-throated marten Martes flavigula in north-central Thailand. Wildl Biol 11:49–57. doi: 10.2981/0909-6396(2005)11[49:RHUAAP]2.0.CO;2URL:
Grassman LI, Tewes ME, Silvy NJ, Kreetiyutanont K (2005b) Spatial organization and diet of the leopard cat (Prionailurus bengalensis) in north-central Thailand. J Zool 266:45–54. doi:10.1017/S095283690500659X
Haines AM, Grassman LI, Tewes ME, Janečka JE (2006) First ocelot (Leopardus pardalis) monitored with GPS telemetry. Eur J Wildl Res 52:216–218. doi:10.1007/s10344-006-0043-5
Hays GC, Mortimer JA, Ierodiaconou D, Estaban N (2014) Use of long-distance migration patterns of an endangered species to inform conservation planning for the world’s largest marine protected area. Conserv Biol 28:1636–1644. doi:10.1111/cobi.12325
Hebblewhite M, Haydon DT (2010) Distinguishing technology from biology: a critical review of the use of GPS telemetry data in ecology. Phil Trans R Soc B 365:2303–2312. doi:10.1098/rstb.2010.0087
Hebblewhite M, Percy M, Merrill EH (2007) Are all global positioning system collars created equal? Correcting habitat-induced bias using three brands in the Central Canadian Rockies. J Wildl Manage 71:2026–2033. doi:10.2193/2006-238
Heydon MJ, Bulloh P (1996) The impact of selective logging on sympatric civet species in Borneo. Oryx 30:31–36. doi:10.1017/S0030605300021360
Iseborn T, Rogers LD, Rawson B, Nekaris KAI (2010) Sightings of common palm civets Paradoxurus hermaphroditus and of other civet species at Phnom Samkos Wildlife Sanctuary and Veun Sai-Siem Pang Conservation Area, Cambodia. Small Carnivore Conserv 46:26–29
Jennings AP, Veron G (2009) Family viverridae (Civets, Genets, and Oyans). In: Wilson DE, Mittermeier RA (eds) Handbook of mammals of the world, vol 1., carnivoresLynx Edicions, Barcelona, pp 174–232
Jennings AP, Seymour AS, Dunstone N (2006) Ranging behaviour, spatial organization and activity of the Malay civet (Viverra tangalunga) on Buton Island, Sulawesi. J Zool 268:63–71. doi:10.1111/j.1469-7998.2005.00023.x
Jennings AP, Zubaid A, Veron G (2010) Ranging behaviour, activity, habitat use, and morphology of the Malay civet (Viverra tangalunga) on Peninsular Malaysia and comparison with studies on Borneo and Sulawesi. Mamm Biol 75:437–446. doi:10.1016/j.mambio.2009.10.002
Jennings AP, Naim M, Advento AD, Aryawan AAK, Ps S, Caliman J, Verwilghen A, Veron G (2015) Diversity and occupancy of small carnivores within oil palm plantations in central Sumatra, Indonesia. Mamm Res 60:181–188. doi:10.1007/s13364-015-0217-1
Jiang Z, Sugita M, Kitahara M, Takatsuki S, Goto T, Yoshida Y (2008) Effects of habitat feature, antenna position, movement, and fix interval on GPS radio collar performance in Mount Fuji, Central Japan. Ecol Res 23:581–588. doi:10.1007/s11284-007-0412-x
Johnson CJ, Heard DC, Parker KL (2002) Expectations and realities of GPS animal location collars: results of three years in the field. Wildl Biol 8:153–159
Joshi AR, Smith JLD, Cuthbert FJ (1995) Influence of food distribution and predation pressure on spacing behavior in palm civets. J Mamm 76:1205–1212. doi:10.2307/1382613
Kochanny CO, Delguidice GD, Fieberg J (2002) Comparing global positioning system and very high frequency telemetry home ranges of white-tailed deer. J Wildl Manage 73:779–787. doi:10.2193/2008-394
Land ED, Shindle DB, Kawula RJ, Benson JF, Lotz MA, Onorato DP (2006) Florida panther habitat selection analysis of concurrent GPS and VHF telemetry data. J Wildl Manage 72:633–639. doi:10.2193/2007-136
LaPoint S, Gallery P, Wikelski M, Kays R (2013) Animal behavior, cost-based corridor models, and real corridors. Landsc Ecol 28:1615–1630. doi:10.1007/s10980-013-9910-0
Lewis JS, Rachlow JL, Garton EO, Vierling LA (2007) Effects of habitat on GPS collar performance: using data screening to reduce location error. J Appl Ecol 44:663–671. doi:10.1111/j.1365-2664.2007.01286.x
Lindsell JA, Serra G, Peške L, Abdullah MS, Qaim GA, Kanani A, Wondafrash M (2009) Satellite tracking reveals the migration route and wintering area of the Middle East population of Critically Endangered northern bald ibis Geronticus eremita. Oryx 43:329–335. doi:10.1017/S0030605309001963
Lührs ML, Kappeler PM (2013) Simultaneous GPS tracking reveals male associations in a solitary carnivore. Behav Ecol Sociobiol 67:1731–1743. doi:10.1007/s00265-013-1581-y
Margules CR, Pressey RL (2000) Systematic conservation planning. Nature 405:243–253. doi:10.1038/35012251
Mattisson J, Andren H, Persson J, Segerstrom P (2010) Effects of species behavior on global positioning system collar fix rates. J Wildl Manage 74:557–563. doi:10.2193/2009-157
Nakashima Y, Nakabayashi M, Sukor JA (2013) Space use, habitat selection, and day-beds of the common palm civet (Paradoxurus hermaphroditus) in human-modified habitats in Sabah, Borneo. J Mamm 94:1169–1178. doi:10.1644/12-MAMM-A-140.1
Nozaki E, Azuma S, Sasaki H, Torri H (1994) Home range of the Malay civet (Viverra tangalunga) in Teluk Kaba area, Kutai National Park, East Kalimantan, Indonesia. Kyoto Univ Over Rep Spec Num 85–94
Payne J, Francis CM (1985) A field guide to the mammals of Borneo. The Sabah Society, Kota Kinabalu
Recio MR, Mathieu R, Maloney R, Seddon PJ (2010) First results of feral cats (Felis catus) monitored with GPS collars in New Zealand. N Z J Ecol 34:288–296
Recio MR, Mathieu R, Denys P, Sirguey P, Seddon PJ (2011a) Lightweight GPS-tags, one giant leap for wildlife tracking? An assessment approach. PLoS One 6:e28225. doi:10.1371/journal.pone.0028225
Recio MR, Mathieu R, Maloney R, Seddon PJ (2011b) Cost comparison between GPS- and VHF-based telemetry: case study of feral cats. N Z J Ecol 35:114–117
Recio MR, Mathieu R, Seddon PJ (2011c) Design of a GPS backpack to track European hedgehogs Erinaceus europaeus. Eur J Wildl Res 57:1175–1178. doi:10.1007/s10344-011-0530-1
Rempel RS, Rodgers AR, Abraham KF (1995) Performance of a GPS animal location system under boreal forest canopy. J Wildl Manage 59:543–551
Ripple WJ et al (2015) Collapse of the world’s largest herbivores. Sci Adv 1:e1400103. doi:10.1126/sciadv.1400103
Rodgers AR (2001) Tracking animals with GPS: The first 10 years. Pages 1–10 in Proceedings of Tracking Animals with GPS, 12 March–13 March 2001, Aberdeen, Scotland
Ropert-Coudert Y, Wilson RP (2005) Trends and perspectives in animal-attached remote sensing. Front Ecol Environ 3:437–444. doi:10.2307/3868660
Schipper J et al (2008) The status of the world’s land and marine mammals: diversity, threat, and knowledge. Science 322:225–230. doi:10.1126/science.1165115
Schofield G, Lilley MKS, Bishop CM, Brown P, Katselidis KA, Dimopoulos P, Pantis JD, Hays GC (2009) Conservation hotspots: implications of intense spatial area use by breeding male and female loggerheads at the Mediterranean’s largest rookery. Endanger Species Res 10:191–202. doi:10.3354/esr00137
Schreiber A, Wirth R, Riffel M, Van Rompaey H (1989) Weasels, civets, mongooses, and their relatives: an action plan for the conservation of mustelids and viverrids. IUCN, Gland
Sikes RS, Gannon WL (2011) Guidelines of the American Society of Mammalogists for the use of wild mammals in research. J Mamm 92:235–253. doi:10.1644/10-MAMM-F-355.1
Simcharoen A, Savini T, Gale GA, Simcharoen S, Duangchantrasiri S, Pakpien S, Smith JLD (2014) Female tiger Panthera tigris home range size and prey abundance: important metrics for management. Oryx 48:370–377. doi:10.1017/S0030605312001408
Tomkiewicz SM, Fuller MR, Kie JG, Bates KK (2010) Global positioning system and associated technologies in animal behaviour and ecological research. Phil Trans R Soc B 365:2163–2176. doi:10.1098/rstb.2010.0090
Wilting A, Samejima H, Mohamed A (2010) Diversity of Bornean viverrids and other small carnivores in Deramakot. Small Carnivore Conserv 42:10–13
Acknowledgments
We thank the Sabah Wildlife Department and the Sabah Biodiversity Centre for issuing the research permits necessary to conduct this study. We are grateful to the funding provided by Sime Darby Foundation, Houston Zoo, Phoenix Zoo, and Danau Girang Field Centre. A hearty thank you goes out to L. Evans, S. Edwards, and S. Joscelyne for their assistance on the project. Further thanks go to A. Jennings, G. Veron, W. Heidrich, and F. Kümmeth for their knowledge, guidance and continued support.
Compliance with ethical standards
This study was approved by and conducted under the jurisdiction of Sabah Wildlife Department and with the clearance of the Sabah Biodiversity Centre.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Evans, M.N., Guerrero-Sanchez, S., Bakar, M.S.A. et al. First known satellite collaring of a viverrid species: preliminary performance and implications of GPS tracking Malay civets (Viverra tangalunga). Ecol Res 31, 475–481 (2016). https://doi.org/10.1007/s11284-016-1338-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-016-1338-y