Abstract
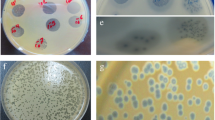
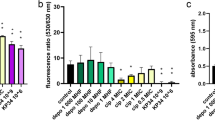
The ability of most opportunistic bacteria to form biofilms, coupled with antimicrobial resistance, hinder the efforts to control widespread infections, resulting in high risks of negative outcomes and economic costs. Endolysins are promising compounds that efficiently combat bacteria, including multidrug-resistant strains and biofilms, without a low probability of subsequent emergence of stable endolysin-resistant phenotypes. However, the details of antibiofilm effects of these enzymes are poorly understood. To elucidate the interactions of bacteriophage endolysins LysAm24, LysAp22, LysECD7, and LysSi3 with bacterial films formed by Gram-negative species, we estimated their composition and assessed the endolysins’ effects on the most abundant exopolymers in vitro. The obtained data suggests a pronounced efficiency of these lysins against biofilms with high (Klebsiella pneumoniae) and low (Acinetobacter baumannii) matrix contents, or dual-species biofilms, resulting in at least a twofold loss of the biomass. These peptidoglycan hydrolases interacted diversely with protective compounds of biofilms such as extracellular DNA and polyanionic carbohydrates, indicating a spectrum of biofilm-disrupting effects for bacteriolytic phage enzymes. Specifically, we detected disruption of acid exopolysaccharides by LysAp22, strong DNA-binding capacity of LysAm24, both of these interactions for LysECD7, and neither of them for LysSi3.
Graphical abstract







Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information. Additionally, the raw data are available from the authors upon reasonable request.
References
Ahmad I, Nygren E, Khalid F, Myint SL, Uhlin BE (2020) A Cyclic-di-GMP signaling network regulates biofilm formation and surface associated motility of Acinetobacter baumannii 17978. Sci Rep 10:1–11. https://doi.org/10.1038/s41598-020-58522-5
Antonova NP, Vasina DV, Lendel AM, Usachev EV, Makarov VV, Gintsburg AL, Tkachuk AP, Gushchin VA (2019) Broad bactericidal activity of the myoviridae bacteriophage lysins LysAm 24, LysECD7, and LysSi3 against gram-negative ESKAPE pathogens. Viruses 11:284. https://doi.org/10.3390/v11030284
Arroyo-Moreno S, Cummings M, Corcoran DB, Coffey A, McCarthy RR (2022) Identification and characterization of novel endolysins targeting Gardnerella vaginalis biofilms to treat bacterial vaginosis. npj Biofilms Microbiomes 8:29. https://doi.org/10.1038/s41522-022-00285-0
Batoni G, Maisetta G, Esin S (2016) Antimicrobial peptides and their interaction with biofilms of medically relevant bacteria. Biochim Biophys Acta, Biomembr 1858:1044–1060. https://doi.org/10.1016/j.bbamem.2015.10.013
Chiang WC, Nilsson M, Jensen PØ, Høiby N, Nielsen TE, Givskov M, Tolker-Nielsen T (2013) Extracellular DNA shields against aminoglycosides in Pseudomonas aeruginosa Biofilms. Antimicrob Agents Chemother 57:2352–2361. https://doi.org/10.1128/AAC.00001-13
Choi AHK, Slamti L, Avci FY, Pier GB, Maira-Litrán T (2009) The pgaABCD locus of Acinetobacter baumannii encodes the production of poly-β-1-6-N-acetylglucosamine, which is critical for biofilm formation. J Bacteriol 191:5953–5963. https://doi.org/10.1128/JB.00647-09
Das T, Sehar S, Koop L, Wong YK, Ahmed S, Siddiqui KS, Manefield M (2014) Influence of calcium in extracellular DNA mediated bacterial aggregation and biofilm formation. PLoS ONE 9:e91935. https://doi.org/10.1371/journal.pone.0091935
De Gregorio E, Del Franco M, Martinucci M, Roscetto E, Zarrilli R, Di Nocera PP (2015) Biofilm-associated proteins: news from Acinetobacter. BMC Genomics 16:1–14. https://doi.org/10.1186/s12864-015-2136-6
Donlan RM, Costerton JW (2002) Biofilms: microbial life on surfaces. Emerg Infect Dis 8:881–890. https://doi.org/10.3201/eid0809.020063
Eriksson E, Liu PY, Schultz GS, Martins-Green MM, Tanaka R, Weir D, Gould LJ, Armstrong DG, Gibbons GW, Wolcott R (2022) Chronic wounds: treatment consensus. Wound Rep Reg 30:156–171. https://doi.org/10.1111/wrr.12994
Fernández L, González S, Campelo AB, Martínez B, Rodríguez A, García P (2017) Downregulation of autolysin-encoding genes by phage-derived lytic proteins inhibits biofilm formation in Staphylococcus aureus. Antimicrob Agents Chemother 61:e02724-e2816. https://doi.org/10.1128/AAC.02724-16
Flemming H-C, Wingender J, Szewzyk U, Steinberg P, Rice SA, Kjelleberg S (2016) Biofilms: an emergent form of bacterial life. Nat Rev Microbiol 14:563–575. https://doi.org/10.1038/nrmicro.2016.94
Freeman DJ, Falkiner FR, Keane CT (1989) New method for detecting slime production by coagulase negative staphylococci. J Clin Pathol 42:872–874. https://doi.org/10.1136/jcp.42.8.872
Giacobbe DR, Mikulska M, Viscoli C (2018) Recent advances in the pharmacological management of infections due to multidrug-resistant gram-negative bacteria. Expert Rev Clin Pharmacol 11:1219–1236. https://doi.org/10.1080/17512433.2018.1549487
Gontijo MTP, Vidigal PMP, Lopez MES, Brocchi M (2021) Bacteriophages that infect gram-negative bacteria as source of signal-arrest-release motif lysins. Res Microbiol 172:103794. https://doi.org/10.1016/j.resmic.2020.103794
Grishin AV, Karyagina AS, Vasina DV, Vasina IV, Gushchin VA, Lunin VG (2020) Resistance to peptidoglycan-degrading enzymes. Crit Rev Microbiol 46:703–726. https://doi.org/10.1080/1040841X.2020.1825333
Gutiérrez D, Ruas-Madiedo P, Martínez B, Rodríguez A, García P (2014) Effective removal of staphylococcal biofilms by the endolysin LysH5. PLoS ONE 9:e107307. https://doi.org/10.1371/journal.pone.0107307
Heselpoth RD, Swift SM, Linden SB, Mitchell MS, Nelson DC (2021) Enzybiotics: endolysins and bacteriocins. In: Harper DR, Abedon ST, Burrowes BH, McConville ML (eds) Bacteriophages: biology, technology, therapy. Springer International Publishing, Cham, pp 989–1030
Hughes RB, Smith AC (2013) Capsule stain protocols. Am J Microbiol 13:1–12
Irazoki O, Hernandez SB, Cava F (2019) Peptidoglycan muropeptides: release, perception, and functions as signaling molecules. Front Microbiol 10:500. https://doi.org/10.3389/fmicb.2019.00500
Iwashkiw JA, Seper A, Weber BS, Scott NE, Vinogradov E, Stratilo C, Reiz B, Cordwell SJ, Whittal R, Schild S, Feldman MF (2012) Identification of a general O-linked protein glycosylation system in Acinetobacter baumannii and its role in virulence and biofilm formation. PLoS Pathog 8:e1002758. https://doi.org/10.1371/journal.ppat.1002758
Karau MJ, Mandrekar J, Lehoux D, Schuch R, Cassino C, Patel R (2023) In vitro activity of exebacase against methicillin-resistant Staphylococcus aureus biofilms on orthopedic Kirschner wires. BMC Res Notes 16:209. https://doi.org/10.1186/s13104-023-06468-y
Karygianni L, Ren Z, Koo H, Thurnheer T (2020) Biofilm matrixome: extracellular components in structured microbial communities. Trends Microbiol 28:668–681. https://doi.org/10.1016/j.tim.2020.03.016
Koo H, Allan RN, Howlin RP, Stoodley P, Hall-Stoodley L (2017) Targeting microbial biofilms: current and prospective therapeutic strategies. Nat Rev Microbiol 15:740–755. https://doi.org/10.1038/nrmicro.2017.99
Liu L, Jia X, Zhao X, Li T, Luo Z, Deng R, Peng B, Mao D, Liu H, Zheng Q (2023) In vitro PCR verification that lysozyme inhibits nucleic acid replication and transcription. Sci Rep 13:6383. https://doi.org/10.1038/s41598-023-33228-6
Mann EE, Wozniak DJ (2012) Pseudomonas biofilm matrix composition and niche biology. FEMS Microbiol Rev 36:893–916. https://doi.org/10.1111/j.1574-6976.2011.00322.x
Minas K, McEwan NR, Newbold CJ, Scott KP (2011) Optimization of a high-throughput CTAB-based protocol for the extraction of qPCR-grade DNA from rumen fluid, plant and bacterial pure cultures. FEMS Microbiol Lett 325:162–169. https://doi.org/10.1111/j.1574-6968.2011.02424.x
Nesse LL, Osland AM, Mo SS, Sekse C, Slettemeås JS, Bruvoll AEE, Urdahl AM, Vestby LK (2020) Biofilm forming properties of quinolone resistant Escherichia coli from the broiler production chain and their dynamics in mixed biofilms. BMC Microbiol 20:46. https://doi.org/10.1186/s12866-020-01730-w
Omar A, Wright JB, Schultz G, Burrell R, Nadworny P (2017) Microbial biofilms and chronic wounds. Microorganisms 5:1–15. https://doi.org/10.3390/microorganisms5010009
Ostapska H, Howell PL, Sheppard DC (2018) Deacetylated microbial biofilm exopolysaccharides: it pays to be positive. PLoS Pathog 14:e1007411. https://doi.org/10.1371/journal.ppat.1007411
Paczosa MK, Mecsas J (2016) Klebsiella pneumoniae: going on the offense with a strong defense. Microbiol Mol Biol Rev 80:629–661. https://doi.org/10.1128/mmbr.00078-15
Pompilio A, Scribano D, Sarshar M, Di Bonaventura G, Palamara AT, Ambrosi C (2021) Gram-negative bacteria holding together in a biofilm: the Acinetobacter baumannii way. Microorganisms 9:1353. https://doi.org/10.3390/microorganisms9071353
Prasad NK, Seiple IB, Cirz RT, Rosenberg OS (2022) Leaks in the pipeline: a failure analysis of gram-negative antibiotic development from 2010 to 2020. Antimicrob Agents Chemother 66:e00054-e122. https://doi.org/10.1128/aac.00054-22
Puchtler H, Sweat F, Levine M (1962) On the binding of Congo red by amyloid. J Histochem Cytochem 10:355–364. https://doi.org/10.1177/10.3.355
Römling U, Sierralta WD, Eriksson K, Normark S (1998) Multicellular and aggregative behaviour of Salmonella typhimurium strains is controlled by mutations in the agfD promoter. Mol Microbiol 28:249–264. https://doi.org/10.1046/j.1365-2958.1998.00791.x
Said KB, Alsolami A, Alshammari F, Alreshidi FS, Fathuldeen A, Alrashid F, Bashir AI, Osman S, Aboras R, Alshammari A, Alshammari T, Alharbi SF (2022) Selective COVID-19 coinfections in diabetic patients with concomitant cardiovascular comorbidities are associated with increased mortality. Pathogens 11:508. https://doi.org/10.3390/pathogens11050508
Scott JE, Dorling J (1965) Differential staining of acid glycosaminoglycans (mucopolysaccharides) by Alcian blue in salt solutions. Histochemie 5:221–233. https://doi.org/10.1007/BF00306130
Singh JK, Adams FG, Brown MH (2019) Diversity and function of capsular polysaccharide in Acinetobacter baumannii. Front Microbiol 9:3301. https://doi.org/10.3389/fmicb.2018.03301
Stepanović S, Vuković D, Hola V, Di Bonaventura G, Djukić S, Ćirković I, Ruzicka F (2007) Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS 115:891–899. https://doi.org/10.1111/j.1600-0463.2007.apm_630.x
Stepnaya OA, Ryazanova LP, Krupyanko VI, Kulaev IS (2001) Influence of acidic exopolysaccharide of Xanthomonas campestris IBPM 124 on the kinetic parameters of extracellular bacteriolytic enzymes. Biochemistry 66:662–666. https://doi.org/10.1023/A:1010263432155
Stewart PS (2015) Antimicrobial tolerance in biofilms. Microbiol Spectr 3:269–285. https://doi.org/10.1128/microbiolspec.mb-0010-2014
Tetz GV, Artemenko NK, Tetz VV (2009) Effect of DNase and antibiotics on biofilm characteristics. Antimicrob Agents Chemother 53:1204–1209. https://doi.org/10.1128/AAC.00471-08
Vasina DV, Antonova NP, Grigoriev IV, Yakimakha VS, Lendel AM, Nikiforova MA, Pochtovyi AA, Remizov TA, Usachev EV, Shevlyagina NV, Zhukhovitsky VG, Fursov MV, Potapov VD, Vorobev AM, Aleshkin AV, Laishevtsev AI, Makarov VV, Yudin SM, Tkachuk AP, Gushchin VA (2021) Discovering the potentials of four phage endolysins to combat gram-negative infections. Front Microbiol 12:748718. https://doi.org/10.3389/fmicb.2021.748718
Wohlfarth JC, Feldmüller M, Schneller A, Kilcher S, Burkolter M, Meile S, Pilhofer M, Schuppler M, Loessner MJ (2023) L-form conversion in gram-positive bacteria enables escape from phage infection. Nat Microbiol 8:387–399. https://doi.org/10.1038/s41564-022-01317-3
Acknowledgements
Not applicable.
Funding
The study was supported by the Russian Science Foundation (RSF), grant No. 23–74-10027, https://rscf.ru/project/23-74-10027/.
Author information
Authors and Affiliations
Contributions
Conceptualization: Daria V. Vasina; Methodology: Anastasiya M. Lendel; Formal analysis and investigation: Anastasiya M. Lendel, Nataliia P. Antonova, Igor V. Grigoriev, Evgeny V. Usachev; Writing—original draft preparation: Anastasiya M. Lendel; Writing—review and editing: Nataliia P. Antonova, Igor V. Grigoriev, Daria V. Vasina; Funding acquisition: Daria V. Vasina; Resources and project administration: Vladimir A. Gushchin; Supervision: Daria V. Vasina. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no financial or proprietary interests in any material discussed in this article.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Authors information
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lendel, A.M., Antonova, N.P., Grigoriev, I.V. et al. Biofilm-disrupting effects of phage endolysins LysAm24, LysAp22, LysECD7, and LysSi3: breakdown the matrix. World J Microbiol Biotechnol 40, 186 (2024). https://doi.org/10.1007/s11274-024-03999-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-024-03999-9