Abstract
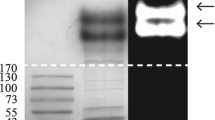
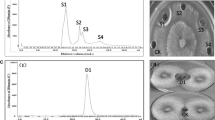
Public concern for food safety and environmental issues and the increase in fungicide-resistant pathogen have enhanced the interest in developing alternative methods to fungicides to control postharvest fruit decay. In this study, a bacterial strain isolated from stale potato vermicelli was identified as Bacillus pumilus HN-10 based on morphological characteristics and 16S rRNA gene sequence analysis. Furthermore, two novel cationic antifungal peptides named P-1 and P-2 were purified from B. pumilus HN-10 using macroporous adsorbent resin AB-8, Sephadex G-100 chromatography, and reversed-phase high-performance liquid chromatography. The primary structure of P-1 and P-2, which were proved to be novel antifungal peptides by BLAST search in NCBI database, was PLSSPATLNSR and GGSGGGSSGGSIGGR with a molecular weight of 1142.28 and 1149.14 Da, respectively, as indicated by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Both P-1 and P-2 exhibited strong antifungal activity against Trichothecium roseum with minimum inhibitory concentrations starting from 1 μg/mL. The two novel antifungal peptides were stable below 80 °C for 2 h, but lost their activity in 15 min at 121 °C. In addition, they were resistant to the proteolytic action of pepsin, trypsin, and papain, and stable within a wide range of pH (2.0–12.0). These results showed that P-1 and P-2 are novel cationic antifungal peptides with specific activity against T. roseum.






Similar content being viewed by others
References
Brogden KA (2005) Antimicrobial peptides: pore formers or metabolic inhbitors in bacteria? Nat Rev Microbiol 3(3):238–250. https://doi.org/10.1038/nrmicro1098
Chan DI, Prenner EJ, Vogel HJ (2006) Tryptophan- and arginine-rich antimicrobial peptides: structures and mechanisms of action. BBA-Biomembranes 1758:1184–1202. https://doi.org/10.1016/j.bbamem.2006.04.006
Chen YX, Guarnieri MT, Vasil AI, Vasil ML, Mant CT, Hodges RS (2007) Role of peptide hydrophobicity in the mechanism of action of α-Helical antimicrobial peptides. Antimicrob Agents Ch 51(4):1398–1406. https://doi.org/10.1128/AAC.00925-06
Dutta SR, Gauri SS, Ghosh T, Halder SK, DasMohapatra PK, Mondal KC, Ghosh AK (2017) Elucidation of structural and functional integration of a novel antimicrobial peptide from Antheraea mylitta. Bioorg Med Chem Lett 27(8):1686–1692. https://doi.org/10.1016/j.bmcl.2017.03.003
Ge YH, Deng HW, Bi Y, Li CY, Liu YY, Dong BY (2015) Postharvest ASM dipping and DPI pre-treatment regulated reactive oxygen species metabolism in muskmelon (Cucumis melo L.) fruit. Postharvest Biol Technol 99:160–167. https://doi.org/10.1016/j.postharvbio.2014.09.001
Hancock RE (2001) Cationic peptides effectors in innate immunity and novel antimicrobials. Lancet Infect Dis 1(3):156–164. https://doi.org/10.1016/s1473-3099(01)00092-5
Huang YB, Huang JF, Chen YX (2010) Alpha-helical cationic antimicrobial peptides: relationships of structure and function. Protein Cell 1(2):143–152. https://doi.org/10.1007/s13238-010-0004-3
Huang XQ, Zhang N, Yong XY, Yang XG, Shen QR (2012) Biocontrol of Rhizoctonia solani damping-off disease in cucumber with Bacillus pumilus SQR-N43. Microbiol Res 167(3):135–143. https://doi.org/10.1016/j.micres.2011.06.002
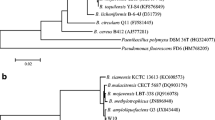
Huang YQ, Yun JM, Zhang WW, Ai DY, Qi QY, Yao B (2017) Effect of Bacillus pumilus NH-10 on antagonistic activity and cell structure of Trichothecium roseum. J Food Sci Biotechnol 36(7):689–692 (in Chinese)
Kayalvizhi N, Gunasekaran P (2010) Purification and characterization of a novel broad-spectrum bacteriocin from Bacillus licheniformis MKU3. Biotechnol Bioproc E 15(2):365–370. https://doi.org/10.1007/s12257-009-0164-2
Leaes FL, Velho RV, Gomes Caldas DG, Ritter AC, Tsai SM, Brandelli A (2016) Expression of essential genes for biosynthesis of antimicrobial peptides of Bacillus is modulated by inactivated cells of target microorganisms. Res Microbiol 167:83–89. https://doi.org/10.1016/j.resmic.2015.10.005
Lee DG, Kim HN, Park Y, Kim HK, Choi BH, Choi CH, Hahm KS (2002) Design of novel analogue peptides with potent antibiotic activity based on the antimicrobial peptide, HP(2–20), derived from N-terminus of Helicobacter pylori ribosomal protein L1. BBA 1598:185–194
Li WH, Bi Y, Ge YH, Li YC, Wang JJ, Wang Y (2012) Effects of postharvest sodium silicate treatment on pink rot disease and oxidative stress-antioxidative system in muskmelon fruit. Eur Food Res Technol 234(1):137–145. https://doi.org/10.1007/s00217-011-1611-9
Li SM, Hao LL, Bao WG, Zhang P, Su D, Cheng YY, Nie LY, Wang G, Hou F, Yang Y (2016) A novel short anionic antimicrobial peptide isolated from the skin of Xenopus laevis with broad antimicrobial activity and inhibitory activity against breast cancer cell. Arch Microbiol 198(5):473–482. https://doi.org/10.1007/s00203-016-1206-8
Marc O, Philippe J (2007) Bacillus lipopeptides: versatile weapons for plant disease bio control. Trends Microbiol 16(3):115–125. https://doi.org/10.1016/j.tim.2007.12.009
Miao JY, Guo HX, Ou YW, Liu G, Fang X, Liao ZL, Ke C, Chen YJ, Zhao LC, Cao Y (2014) Purification and characterization of bacteriocin F1, a novel bacteriocin produced by Lactobacillus paracasei subsp. tolerans FX-6 from Tibetan kefir, a traditional fermented milk from Tibet, China. Food Control 42:48–53. https://doi.org/10.1016/j.foodcont.2014.01.041
Midura-Nowaczek K, Markowska A (2014) Antimicrobial peptides and their analogs: searching for new potential therapeutics. Perspect Medicin Chem 6:73–80. https://doi.org/10.4137/PMC.S13215
Migliolo L, Felício MR, Cardoso MH, Silva ON, Xavier ME, Nolasco DO, Oliveira AS, Roca-Subira I, Estape JV, Teixeira LD, Freitas SM, Otero-Gonzalez AJ, Gonçalves S, Santos NC, Franco OL (2016) Structural and functional evaluation of the palindromic alanine-rich antimicrobial peptide Pa-MAP2. BBA-Biomembrane 1858(7):1488–1498. https://doi.org/10.1016/j.bbamem.2016.04.003
Miyoshi N, Saito T, Ohmura T, Kuroda K, Suita K, Ihara K, Isogai E (2016) Functional structure and antimicrobial activity of persulcatusin, an antimicrobial peptide from the hard tick Ixodes persulcatus. Parasites Vectors 9:85. https://doi.org/10.1186/s13071-016-1360-5
Mookherjee N, Hancock REW (2007) Cationic host defence peptides: innate immune regulatory peptides as a novel approach for treating infections. Cell Mol Life Sci 64:922–933. https://doi.org/10.1007/s00018-007-6475-6
Niu LL, Bi Y, Bai XD, Zhang SG, Xue HL, Li YC, Wang Y, Calderón-Urrea A (2016) Damage to Trichothecium roseum caused by sodium silicate is independent from pH. Can J Microbiol 62(2):161–172. https://doi.org/10.1139/cjm-2015-0657
Nunes CA (2012) Biological control of postharvest diseases of fruit. Eur J Plant Pathol 133(1):181–196. https://doi.org/10.1007/s10658-011-9919-7
Onaizi SA, Leong SSJ (2011) Tethering antimicrobial peptides: current status and potential challenges. Biotechnol Adv 29(1):67–74. https://doi.org/10.1016/j.biotechadv.2010.08.012
Plácido A, Bragança I, Marani M, A AR, Vasconcelos AG, Batziou K, Domingues VF, Eaton P, Leite JRSAA., Delerue-Matos C (2017) Antibacterial activity of novel peptide derived from Cry1Ab16 toxin and development of LbL films for foodborne pathogens control. Mat Sci Eng C-Mater 75(1):503–509. https://doi.org/10.1016/j.msec.2017.02.027
Rahman MS, Choi YH, Choi YS, Yoo JC (2017) Glycin‑rich antimicrobial peptide YD1 from B. amyloliquefaciens, induced morphological alteration in and showed affinity for plasmid DNA of E. coli. AMB Expr 7(8):1–11. https://doi.org/10.1186/s13568-016-0315-8
Regmi S, Choi YH, Choi YS, Kim MR, Yoo JC (2017) Antimicrobial peptide isolated from Bacillus amyloliquefaciens K14 revitalizes its use in combinatorial drug therapy. Folia Microbio 62(2):127–138. https://doi.org/10.1007/s12223-016-0479-2
Rishad KS, Rebello S, Shabanamol PS, Jisha MS (2017) Biocontrol potential of Halotolerant bacterial chitinase from high yielding novel Bacillus Pumilus MCB-7 autochthonous to mangrove ecosystem. Pestic Biochem Phys 137:36–41. https://doi.org/10.1016/j.pestbp.2016.09.005
Shali A, Ghasemi S, Ahmadian G, Ranjbar G, Dehestani A, Khalesi N, Motallebi E, Vahed M (2010) Bacillus pumilus SG2 chitinases induced and regulated by chitin, show inhibitory activity against Fusarium graminearum and Bipolaris sorokiniana. Phytoparasitica 38(2):141–147. https://doi.org/10.1007/s12600-009-0078-8
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599. https://doi.org/10.1093/molbev/msm092
Tang YM, Xue HL, Bi Y, Li YC, Wang Yi, Zhao Y, Shen KP (2014) A method of analysis for T-2 toxin and neosolaniol by UPLC-MS/MS in apple fruit inoculated with Trichothecium roseum. Food Addit Contam Part A 32(4):480–487. https://doi.org/10.1080/19440049.2014.968884
Thompson AH, Bjourson AJ, Orr DF, Shaw C, McClean S (2007) A combined mass spectrometric and cDNA sequencing approach to the isolation and characterization of novel antimicrobial peptides from the skin secretions of Phyllomedusa hypochondrialis azurea. Peptides 28(7):1331–1343. https://doi.org/10.1016/j.peptides.2007.05.001
Wang G, Yu MZ, Dong F, Shi JR, Xu JH (2017) Esterase activity inspired selection and characterization of zearalenone degrading bacteria Bacillus pumilus ES-21. Food Control 77:57–64. https://doi.org/10.1016/j.foodcont.2017.01.021
Acknowledgements
This study was funded by the financial support of the Nature Science Foundation of China (NSFC) (No. 31360405), and the Project of Gansu Major Science and Technology Plans (No. 2016GS09742).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Yan, H., Yun, J., Ai, D. et al. Two novel cationic antifungal peptides isolated from Bacillus pumilus HN-10 and their inhibitory activity against Trichothecium roseum . World J Microbiol Biotechnol 34, 21 (2018). https://doi.org/10.1007/s11274-017-2392-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-017-2392-5