Abstract
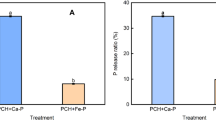
Cells of Azotobacter chroococcum MAL-201 (MTCC 3853) are capable of accumulating the intracellular poly(3-hydroxybutyric acid) [P(3HB)], accounting for 65–71 % of its cell dry weight and also capable of synthesizing the enzyme alkaline phosphatase (APase), when grown in glucose and tricalcium phosphate containing nitrogen-free modified Stockdale medium. The concentration of insoluble phosphate in broth medium was optimized as 0.25 % (w/v) for growth and biosynthesis of APase. However, the suboptimal concentration of phosphate (0.1 %, w/v) appeared as the best suited for accumulation of P(3HB) by the strain. The significant differences were observed in biosynthesis of polymer and APase enzyme under variable phosphate concentrations. Glucose, 3.0 % (w/v) was recorded as the optimum concentration for all of the three parameters. The continuation of APase biosynthesis was observed during the period of significant decline in the cellular content of the polymer in the late phase of growth. In order to study the role of P(3HB), the rate of autodigestion of biopolymer and phosphate solubilization rate (k, mineralization constant) were determined in carbon-free medium under batch cultivation process and the parameters were found to be positively correlated. The maximum phosphate solubilization rate (k = 0.0154) by the strain MAL-201 timed at the 10th hour of incubation when the rate of polymer degradation concomitantly attained its peak corresponding to 87 mg/l/h and then declined gradually. Only a negligible amount of residual polymer remained undigested. These data strongly support the functional role of P(3HB) in response to multinutritional stress condition.




Similar content being viewed by others
References
Anderson AJ, Dawes EA (1990) Occurrence, metabolism, metabolic role, and industrial uses of bacterial polyhydroxyalkanoates. Microbiol Rev 54:450–472
Bernfeld P (1955) Amylases, alpha and beta. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol I. Academic Press, New York, pp 149–158
Chavada NB, Patel R, Vanpuria S, Raval BP, Thakkar PU (2010) A study on isolated diazotrophic (non-symbiotic) bacteria from saline desert soil as a biofertilizer. Int J Pharm Appl Sci 1:52–54
Doi Y, Saesuga A, Kawaguchi Y, Kunioka M (1990) Cyclic nature of poly(3-hydroxyalkanoate) metabolism in Alcaligenes eutrophus. FEMS Micribiol Lett 67:165–170
Goyal V, Chetal S, Nainawatee HS (1984) Water stress alters characteristics of alkaline phosphates in Rhizobium and Azotobacter species. Folia Microbiol 29:228–232
Greenspan EM, Fowler SD (1985) Nile Red: a selective fluorescent stain for intracellular lipid droplets. J Cell Biol 100:965–973
Kadam TA, Gangawane KV (2005) Degradation of forate by Azotobacter isolates. Ind J Biotechnol 4:153–155
Kawaguchi Y, Doi Y (1992) Kinetics and mechanism of synthesis and degradation of poly(3-hydroxybutyrate) in Alcaligenes eutrophus. Macromolecules 25:2324–2329
Lafferty RM, Korasatko B, Korasatko W (1988) Microbiological production of poly-beta-hydroxybutyric acid. In Rehm HJ, Reed G (eds) Biotechnology, 6A Varlagsgesell Schaft Wein Heim, pp 135–176
Law JH, Slepecky RA (1961) Assay of poly-β-hydroxybutyric acid. J Bacteriol 82:33–36
López NL, Floccari ME, Steinbüchel A, García AF, Méndez BS (1994) Effect of poly(3-hydroxybutyrate) (PHB) content on the starvation-survival of bacteria in natural waters. FEMS Microbiol Ecol 16:95–102
Nair IC (2013) Screening for novel bacterial strains capable of accumulating intracellular PHB under phenol stress. Int J Pl Anim Environ Sci 3(2):155–162
Narula N, Kumar V, Bhel R, Deubel A, Gransee A, Merbach W (2000) Effect of phosphate solubilizing Azotobacter chroococcum on N, P, K uptake in P-responsive wheat genotypes grown under greenhouse conditions. J Plant Nutr Soil Sci 163:393–398
Page WJ, Doran JL (1981) Relationship between calcium and uronic acids in the encystment of Azotobacter vinelandii. J Bacteriol 146(1):33–40
Pal Saha S, Paul AK (2005) Intracellular degradation of poly(3-hydroxybutyric acid) by Azotobacter chroococcum MAL-201. Roman Arch Microbiol 64:50–56
Pal Saha S, Patra A, Paul AK (2007) Studies on intracellular degradation of polyhydroxyalkanoic acid-polyethyloene glycol copolymer accumulated by Azotobacter chroococcum MAL-201. J Biotechnol 132:325–330
Pal Saha S, Chakraborty D, Paul AK (2010) On the role of endogenous poly(3-hydroxybutyric acid) in Azotobacter chroococcum MAL-201. J Bot Soc Bengal 64:143–148
Pal S, Manna A, Paul AK (1997) Induction of encystment and polyhydroxybutyric acid production by Azotobacter chroococcum MAL-201. Curr Microbiol 35:327–330
Pal S, Manna A, Paul AK (1998) Nutritional and cultural conditions for production of poly- β-hydroxybutyric acid by Azotobacter chroococcum MAL-201. Folia Microbiol 43:117–181
Pham TH, Webb JS, Rehm BHA (2004) The role of polyhydroxyalkanoate biosynthesis by Pseudomonas aeruginosa in rhamnolipid and alginate production as well as stress tolerance and biofilm formation. Microbiology 150:3405–3413
Ratcliff WC, Kadam SV, Denison RF (2008) Poly-3-hydroxybutyrate (PHB) supports survival and reproduction in starving rhizobia. FEMS Microbiol Ecol 65:391–399
Sadoff HL (1975) Encystment and germination in Azotobacter vinelandii. Bacteriol Rev 39:516–539
Saito T, Takizawa K, Saegusa H (1995) Intracellular poly(3-hydroxybutyrate) depolymerase in Alcaligenes eutrophus. Can J Microbiol 41:187–191
Senior PJ, Beech GA, Ritchie GAF, Dawes EA (1972) The role of oxygen limitation in the formation of poly-β-hydroxybutyric acid during batch and continuous culture of Azotobacter beijerinckii. Biochem J 128:1193–1201
Smith D, Wyss O, Leoposia P (1969) Loss of calcium from Azotobacter cyst. J Basic Microbiol 9:161–166
Stanford G, Smith SJ (1972) Nitrogen mineralization potentials of soils. Soil Sci Soc Am Proc 36:465–472
Stockdale H, Ribbons DW, Dawes EA (1968) Occurance of poly-β-hydroxybutyrate in Azotobacteriaceae. J Bacteriol 95:1798–1803
Walter K, Schütt C (1974) Methods of Enzymatic Analysis, vol II, 2nd edn. Academic Press, New York, pp 860–864
Wang JG, Bakken LR (1998) Screening of soil bacteria for poly-β-hydroxybutyric acid production and its role in the survival of starvation. Microb Ecol 35:94–101
Acknowledgments
This investigation was financially supported by the research grant (PSW/055/10/11) from University Grant Commission, New Delhi, India.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pal Saha, S., Bhattacharyya, S. & Chakraborty, H. Solubilization of tricalcium phosphate by P(3HB) accumulating Azotobacter chroococcum MAL-201. World J Microbiol Biotechnol 30, 1575–1582 (2014). https://doi.org/10.1007/s11274-013-1580-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1580-1