Abstract
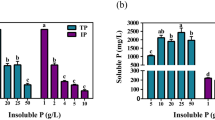
Phosphate-solubilizing fungi (PSF) efficiently dissolve insoluble phosphates through the production of organic acids. This study investigates the mechanisms of organic acid secretion by PSF, specifically Penicillium chrysogenum, under tricalcium phosphate (Ca3(PO4)2, Ca–P) and ferric phosphate (FePO4, Fe–P) conditions. Penicillium chrysogenum exhibited higher phosphorus (P) release efficiency from Ca-P (693.6 mg/L) than from Fe–P (162.6 mg/L). However, Fe–P significantly enhanced oxalic acid (1193.7 mg/L) and citric acid (227.7 mg/L) production by Penicillium chrysogenum compared with Ca–P (905.7 and 3.5 mg/L, respectively). The presence of Fe–P upregulated the expression of genes and activity of enzymes related to the tricarboxylic acid cycle, including pyruvate dehydrogenase and citrate synthase. Additionally, Fe–P upregulated the expression of chitinase and endoglucanase genes, inducing a transformation of Penicillium chrysogenum mycelial morphology from pellet to filamentous. The filamentous morphology exhibited higher efficiency in oxalic acid secretion and P release from Fe–P and Ca–P. Compared with pellet morphology, filamentous morphology enhanced P release capacity by > 40% and > 18% in Ca–P and Fe–P, respectively. This study explored the strategies employed by PSF to improve the dissolution of different insoluble phosphates.







Similar content being viewed by others
Data Availability
Data will be made available on request.
References
Alekseev, K. V., Dubina, M. V., & Komov, V. P. (2017). Molecular-genetic and biochemical characteristics of citrate synthase from the citric-acid producing fungus Aspergillus niger. Applied Biochemistry and Microbiology, 52, 810–817.
Babana, A. H., & Antoun, H. (2006). Effect of Tilemsi phosphate rock-solubilizing microorganisms on phosphorus uptake and yield of field-grown wheat (Triticum aestivum L.) in Mali. Plant and Soil, 287, 51–58.
Behera, B. C. (2020). Citric acid from Aspergillus niger: A comprehensive overview. Critical Reviews in Microbiology, 46, 727–749.
Boot, R. G., Blommaart, E. F., Swart, E., Ghauharali-van der Vlugt, K., Bijl, N., Moe, C., Place, A., & Aerts, J. M. (2001). Identification of a novel acidic mammalian chitinase distinct from chitotriosidase. The Journal of Biological Chemistry, 276, 6770–6778.
Bouzidia, N., Hamdi, B., & Ben Salah, A. (2016). A new non-centrosymmetric microporous fluorinated iron phosphate: Structural elucidation, spectroscopic study and Hirshfeld surface analysis. Chemical Research in Chinese Universities, 32, 519–526.
Busato, J. G., Lima, L. S., Aguiar, N. O., Canellas, L. P., & Olivares, F. L. (2012). Changes in labile phosphorus forms during maturation of vermicompost enriched with phosphorus-solubilizing and diazotrophic bacteria. Bioresource Technology, 110, 390–395.
Canteri, H., & Ghoul, M. (2015). Submerged liquid culture for production of biomass and spores of Penicillium. Food Reviews International, 31, 262–278.
Chen, H. Y. (2019). Why the reactive oxygen species of the fenton reaction switches from oxoiron(IV) species to hydroxyl radical in phosphate buffer solutions? A computational rationale. ACS Omega, 4, 14105–14113.
Clarholm, M., Skyllberg, U., & Rosling, A. (2015). Organic acid induced release of nutrients from metal-stabilized soil organic matter—The unbutton model. Soil Biology and Biochemistry, 84, 168–176.
Cronenberg, C. C. H., Ottengraf, S. P. P., Van den Heuvel, J. C., Pottel, F., Sziele, D., Schügerl, K., & Bellgardt, K. H. (1994). Influence of age and structure of Pencillium chrysogenum pellets on the internal concentration profiles. Bioprocess Engineering, 10, 209–216.
Cwiertny, D. M., Hunter, G. J., Pettibone, J. M., Scherer, M. M., & Grassian, V. H. (2009). Surface chemistry and dissolution of α-FeOOH nanorods and microrods: Environmental implications of size-dependent interactions with oxalate. The Journal of Physical Chemistry C, 113, 2175–2186.
Dai, Z., Zhang, X. N., Nasertorabi, F., Cheng, Q., Pei, H., Louie, S. G., Stevens, R. C., & Zhang, Y. (2018). Facile chemoenzymatic synthesis of a novel stable mimic of NAD+. Chemical Science, 9, 8337–8342.
de Oliveira Mendes, G., Murta, H. M., Valadares, R. V., da Silveira, W. B., da Silva, I. R., & Costa, M. D. (2020). Oxalic acid is more efficient than sulfuric acid for rock phosphate solubilization. Minerals Engineering, 155, 106458.
Dutton, M. V., & Evans, C. S. (1996). Oxalate production by fungi: Its role in pathogenicity and ecology in the soil environment. Canadian Journal of Microbiology, 42, 881–895.
Fischmann, A. J., & Dixon, D. G. (2009). Awaruite (Ni3Fe) as a nickel resource-leaching with ammoniacal–ammonium solution containing citrate and thiosulfate. Hydrometallurgy, 99, 214–224.
Fomina, M., Hillier, S., Charnock, J. M., Melville, K., Alexander, I. J., & Gadd, G. M. (2005). Role of oxalic acid overexcretion in transformations of toxic metal minerals by Beauveria caledonica. Applied and Environmental Microbiology, 71, 371–381.
Gadd, G. M., Bahri-Esfahani, J., Li, Q., Rhee, Y. J., Wei, Z., Fomina, M., & Liang, X. (2014). Oxalate production by fungi: Significance in geomycology, biodeterioration and bioremediation. Fungal Biology Reviews, 28, 36–55.
Hanaka, A., Ozimek, E., Majewska, M., Rysiak, A., & Jaroszuk-Ściseł, J. (2019). Physiological diversity of spitsbergen soil microbial communities suggests their potential as plant growth-promoting bacteria. International Journal of Molecular Sciences, 20, 1207.
Jiang, Y., Tian, J., & Ge, F. (2020). New insight into carboxylic acid metabolisms and pH regulations during insoluble phosphate solubilisation process by Penicillium oxalicum PSF-4. Current Microbiology, 77, 4095–4103.
Koyama, H., Kawamura, A., Kihara, T., Hara, T., Takita, E., & Shibata, D. (2000). Overexpression of mitochondrial citrate synthase in Arabidopsis thaliana improved growth on a phosphorus-limited soil. Plant & Cell Physiology, 41, 1030–1037.
Kumar, R., & Shastri, B. (2017). Role of phosphate-solubilising microorganisms in sustainable agricultural development. In J. Singh & G. Seneviratne (Eds.), Agro-environmental sustainability. Springer.
Li, Z., Bai, T., Dai, L., Wang, F., Tao, J., Meng, S., Hu, Y., Wang, S., & Hu, S. (2016). A study of organic acid production in contrasts between two phosphate solubilizing fungi: Penicillium oxalicum and Aspergillus niger. Scientific Reports, 6, 25313.
Liu, H., Zheng, Z., Wang, P., Gong, G., Wang, L., & Zhao, G. (2013). Morphological changes induced by class III chitin synthase gene silencing could enhance penicillin production of Penicillium chrysogenum. Applied Microbiology and Biotechnology, 97, 3363–3372.
López-Bucio, J., de La Vega, O. M., Guevara-García, A., & Herrera-Estrella, L. (2000). Enhanced phosphorus uptake in transgenic tobacco plants that overproduce citrate. Nature Biotechnology, 18, 450–453.
Mäkelä, M. R., Hildén, K., & Lundell, T. K. (2010). Oxalate decarboxylase: Biotechnological update and prevalence of the enzyme in filamentous fungi. Applied Microbiology and Biotechnology, 87, 801–814.
Marx, N., Croguennec, L., Carlier, D., Bourgeois, L., Kubiak, P., Cras, F. L., & Delmas, C. (2010). Structural and electrochemical study of a new crystalline hydrated iron(III) phosphate FePO4·H2O obtained from LiFePO4(OH) by ion exchange. Chemistry of Materials, 22, 1854–1861.
Mattevi, A., Obmolova, G., Schulze, E., Kalk, K. H., Westphal, A. H., de Kok, A., & Hol, W. G. (1992). Atomic structure of the cubic core of the pyruvate dehydrogenase multienzyme complex. Science, 255, 1544–1550.
McIntyre, M., Müller, C., Dynesen, J., & Nielsen, J. (2001). Metabolic engineering of the morphology of Aspergillus. Advances in Biochemical Engineering/biotechnology, 73, 103–128.
Menezes-Blackburn, D., Paredes, C., Zhang, H., Giles, C. D., Darch, T., Stutter, M., George, T. S., Shand, C., Lumsdon, D., Cooper, P., et al. (2016). Organic acids regulation of chemical microbial phosphorus transformations in soils. Environmental Science & Technology, 50, 11521–11531.
Merzendorfer, H. (2011). The cellular basis of chitin synthesis in fungi and insects: Common principles and differences. European Journal of Cell Biology, 90, 759–769.
Palmieri, F., Estoppey, A., House, G. L., Lohberger, A., Bindschedler, S., Chain, P. S. G., & Junier, P. (2019). Chapter two-oxalic acid, a molecule at the crossroads of bacterial-fungal interactions. In G. M. Gadd & S. Sariaslani (Eds.), Advances in applied microbiology (Vol. 106, pp. 49–77). Academic Press.
Papagianni, M., & Mattey, M. (2006). Morphological development of Aspergillus niger in submerged citric acid fermentation as a function of the spore inoculum level. Application of neural network and cluster analysis for characterization of mycelial morphology. Microbial Cell Factories, 5, 3.
Papagianni, M., & Moo-Young, M. (2002). Protease secretion in glucoamylase producer Aspergillus niger cultures: Fungal morphology and inoculum effects. Process Biochemistry, 37, 1271–1278.
Qaswar, M., Dongchu, L., Jing, H., Tianfu, H., Ahmed, W., Abbas, M., Lu, Z., Jiangxue, D., Khan, Z. H., Ullah, S., et al. (2020). Interaction of liming and long-term fertilization increased crop yield and phosphorus use efficiency (PUE) through mediating exchangeable cations in acidic soil under wheat-maize cropping system. Scientific Reports, 10, 19828.
Rawat, P., Das, S., Shankhdhar, D., & Shankhdhar, S. C. (2021). Phosphate-solubilizing microorganisms: Mechanism and their role in phosphate solubilization and uptake. Journal of Soil Science and Plant Nutrition, 21, 49–68.
Ryan, P., Delhaize, E., & Jones, D. (2001). Function and mechanism of organic anion exudation from plant roots. Annual Review of Plant Physiology and Plant Molecular Biology, 52, 527–560.
Saxena, J., Basu, P., Jaligam, V., & Chandra, S. (2013). Phosphate solubilization by a few fungal strains belonging to the genera Aspergillus and Penicillium. African Journal of Microbiology Research, 7, 4862–4869.
Schmalenberger, A., Duran, A. L., Bray, A. W., Bridge, J., Bonneville, S., Benning, L. G., Romero-Gonzalez, M. E., Leake, J. R., & Banwart, S. A. (2015). Oxalate secretion by ectomycorrhizal Paxillus involutus is mineral-specific and controls calcium weathering from minerals. Scientific Reports, 5, 12187.
Spohr, A., Carlsen, M., Nielsen, J., & Villadsen, J. (1997). Morphological characterization of recombinant strains of Aspergillus oryzae producing alpha-amylase during batch cultivations. Biotechnology Letters, 19, 257–262.
Su, M., Meng, L., Zhao, L., Tang, Y., Qiu, J., Tian, D., & Li, Z. (2021). Phosphorus deficiency in soils with red color: Insights from the interactions between minerals and microorganisms. Geoderma, 404, 115311.
Tian, D., Wang, W., Su, M., Zheng, J., Wu, Y., Wang, S., Li, Z., & Hu, S. (2018). Remediation of lead-contaminated water by geological fluorapatite and fungus Penicillium oxalicum. Environmental Science and Pollution Research International, 25, 21118–21126.
Tian, D., Jiang, Z., Jiang, L., Su, M., Feng, Z., Zhang, L., Wang, S., Li, Z., & Hu, S. (2019). A new insight into lead (II) tolerance of environmental fungi based on a study of Aspergillus niger and Penicillium oxalicum. Environmental Microbiology, 21, 471–479.
Tian, D., Su, M., Zou, X., Zhang, L., Tang, L., Geng, Y., Qiu, J., Wang, S., Gao, H., & Li, Z. (2021a). Influences of phosphate addition on fungal weathering of carbonate in the red soil from karst region. Science of the Total Environment, 755, 142570.
Tian, D., Wang, L., Hu, J., Zhang, L., Zhou, N., Xia, J., Xu, M., Yusef, K. K., Wang, S., Li, Z., et al. (2021b). A study of P release from Fe–P and Ca–P via the organic acids secreted by Aspergillus niger. Journal of Microbiology, 59, 819–826.
Tomer, S., Suyal, D. C., & Goel, R. (2016). Biofertilizers: a timely approach for sustainable agriculture. In D. Choudhary, A. Varma, & N. Tuteja (Eds.), Plant-microbe interaction: An approach to sustainable agriculture. Springer.
Tsuizaki, M., Takeshita, N., Ohta, A., & Horiuchi, H. (2009). Myosin motor-like domain of the class VI chitin synthase CsmB is essential to its functions in Aspergillus nidulans. Bioscience, Biotechnology, and Biochemistry, 73, 1163–1167.
Van Suijdam, J. C., Kossen, N. W. F., & Paul, P. G. (1980). An inoculum technique for the production of fungal pellets. European Journal of Applied Microbiology and Biotechnology, 10, 211–221.
Varadachari, C., Barman, A. K., & Ghosh, K. (1994). Weathering of silicate minerals by organic acids II. Nature of Residual Products. Geoderma, 61, 251–268.
Veneklaas, E. J., Lambers, H., Bragg, J., Finnegan, P. M., Lovelock, C. E., Plaxton, W. C., Price, C. A., Scheible, W. R., Shane, M. W., et al. (2012). Opportunities for improving phosphorus-use efficiency in crop plants. New Phytologist, 195, 306–320.
Wang, L., Guan, H., Hu, J., Feng, Y., Li, X., Yusef, K. K., Gao, H., & Tian, D. (2022). Aspergillus niger enhances organic and onorganic phosphorus release from wheat straw by secretion of degrading enzymes and oxalic acid. Journal of Agricultural and Food Chemistry, 70, 10738–10746.
Wongwicharn, A., McNeil, B., & Harvey, L. M. (1999). Effect of oxygen enrichment on morphology, growth, and heterologous protein production in chemostat cultures of Aspergillus niger B1-D. Biotechnology and Bioengineering, 65, 416–424.
Zhou, R., Wang, Y., Zhang, M., Yu, P. X., & Li, J. (2019). Adsorptive removal of phosphate from aqueous solutions by thermally modified copper tailings. Environmental Monitoring and Assessment, 191, 198.
Žnidaršić, P., & Pavko, A. (2001). The morphology of filamentous fungi in submerged cultivations as a bioprocess parameter. Food Technology and Biotechnology, 39, 237–252.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (NO. 42007030 and 41877099), the Science and Technology Major Project of Anhui Province (202103a06020012), the program at the department of natural resources of Anhui Province (NO. 2021-K-4 and 2021-K-11), and the program at Anhui Agricultural University (NO. yj2019-20). The authors would like to thank BGI Genomics Co., Ltd. (Shenzhen, China) for assistance with sequencing services, and Yang Xu at Anhui Agricultural University for the assistance in SEM analysis.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declared that they have no conflicts of interest to this work. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, L., Tian, D., Zhang, X. et al. The Regulation of Phosphorus Release by Penicillium chrysogenum in Different Phosphate via the TCA Cycle and Mycelial Morphology. J Microbiol. 61, 765–775 (2023). https://doi.org/10.1007/s12275-023-00072-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-023-00072-2