Abstract
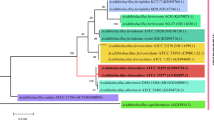
Sulphate-reducing bacteria (SRB) in the thermal springs of Vajreshwari were investigated with combined microbiological and molecular approaches. A sulphate-reducing bacteria medium containing lactate was used for enrichment and isolation, which yielded Gram negative, rod shaped, anaerobic, non-spore forming and motile bacteria capable of reducing sulphate to sulphide. These grew at temperatures ranging from 25 to 55 °C and could use pyruvate, lactate and ethanol as electron donors. Desulfoviridin was detected in all the isolates. The partial 16S rRNA and dissimilatory sulphite reductase (DSR) gene sequences of five representative isolates revealed that the strains belonged to the sulphur reducing bacterial species Desulfovibrio vulgaris.


Similar content being viewed by others
References
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
APHA(1998) Standard methods for the examination of water and waste water. In: Clesceri LS, Greenberg AE, Eaton DA (eds) American Public Health Association, 20th edn. Washington DC, p 1220
Bak F, Pfennig NM (1991) Sulfate reducing bacteria in littoral sediment of Lake Constance. FEMS Microbiol Ecol 85:43–52
Barton LL, Tomei FA (1995) Characteristics and activities of sulphate-reducing bacteria. In: Barton LL (ed) Sulphate-reducing bacteria, Plenum Press, New York, pp 1–32
Castro HF, Williams NH, Ogram A (2000) Phylogeny of sulfate-reducing bacteria. FEMS Microbiol Ecol 31:1–9
Caumette P (1993) Ecology and physiology of phototrophic bacteria and sulphate-reducing bacteria in marine salterns. Experientia 49:473–481
Cottrell MT, Cary SC (1999) Diversity of dissimilatory sulfate reductase genes of bacteria associated with the deep-sea hydrothermal vent polychaete annelid Alvinella pompejana. Appl Environ Microbiol 65:1127–1132
Devereux R, Stahl DA (1993) Phylogeny of sulphate-reducing bacteria and a perspective for analyzing their natural communities. In: Odom JM, Singleton R Jr (eds) The sulphate-reducing bacteria: contemporary perspectives. Springer-Verlag, New York, pp 131–160
Fauque GD (1995) Ecology of sulphate-reducing bacteria. In: Barton LL (ed) Sulphate-reducing bacteria. Plenum Press, New York, pp 217–241
Joulian C, Ramsing NB, Ingvorsen K (2001) Congruent phylogenies of most common small-subunit rRNA and dissimilatory sulphite reductase gene sequences retrieved from estuarine sediments. Appl Environ Microbiol 67:3314–3318
Keith SM, Herbert RA, Harfoot CG (1982) Isolation of new types of sulphate-reducing bacteria from estuarine and marine sediments using chemostat enrichments. J Appl Bacteriol 53:29–33
Klein M, Friedrich M, Rogers AJ, Hugenholtz P, Fishbain S, Abicht H, Blackall LL, Stahl DA, Wagner M (2001) Multiple lateral transfers of dissimilatory sulfite-reductase genes between major lineages of sulfate reducing prokaryotes. J Bacteriol 183:6028–6035
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. John Wiley and Sons, New York, pp 115–175
McIntyre PE, Edenborne HM, Hammock RW (1990) Incorporation of bacterial sulphate-reducton into constructed wetlands for the treatment of acid and metal mine drainage. In: Skousen J, Scencindiver J, Samuel D (eds) Proceedings of the 1990 mining and reclamation conference, Morgantown, WV. West Virginia University Publications, pp 409–415
Minz D, Flax JL, Green SJ, Muyzer G, Cohen Y, Wagner M, Rittmann BE, Stahl DA (1999) Diversity of sulphate-reducing bacteria in oxic and anoxic regions of a microbial mat characterized by comparative analysis of dissimilatory sulphite reductase genes. Appl Environ Microbiol 65:4666–4671
Parkes RJ, Gibson GR, Mueller-Harvey I, Buckingham WJ, Herbert R (1989) Determination of the substrates for sulphate-reducing bacteria within marine and estuarine sediments with different rates of sulphate reduction. J Gen Microbiol 135:175–187
Postgate JR (1984) The sulphate-reducing bacteria, 2nd edn. Cambridge University Press, Cambridge, p 208
Scala DJ, Kerkhof LJ (1998) Nitrous oxide reductase (nosZ) gene-specific PCR primers for detection of denitrifiers and three nosZ genes from marine sediments. FEMS Microbiol Lett 162:61–68
Scala DJ, Kerkhof LJ (1999) Diversity of nitrous oxide reductase (nosZ) genes in continental shelf sediments. Appl Environ Microbiol 65:1681–1687
Thomsen TR, Finster K, Ramsing NB (2001) Biogeochemical and molecular signatures of anaerobic methane oxidation in a marine sediment. Appl Environ Microbiol 67:1646–1656
Wagner M, Roger AJ, Flax JL, Brusseau GA, Stahl DA (1998) Phylogeny of dissimilatory sulfite reductases supports an early origin of sulfate respiration. J Bacteriol 180:2975–2982
White C, Gadd GM (1996) Mixed sulphate-reducing bacterial cultures for bioprecipitation of toxic metals: factorial and response-surface analysis of the effects of dilution rate, sulphate and substrate concentration. Microbiology 142:2197–2205
Widdel F, Pfennig N (1984) Dissimilatory sulfate—or sulfur-reducing bacteria. In: Krieg NR (ed) Bergey’s manual of systematic bacteriology, 2nd edn. vol I. The Williams & Wilkins Co., Baltimore, pp 663–679
Woese CR (1987) Bacterial evolution. Microbiol Rev 51:221–271
Zehr JP, Capone DG (1996) Problems and promises of assaying the genetic potential for nitrogen fixation in the marine environment. Microb Ecol 32:263–281
Zumft WG (1997) Cell biology and molecular basis of denitrification. Microbiol Mol Biol Rev 61:533–616
Acknowledgements
The authors are grateful to Dr. S. C. Mukherjee, Director, Central Institute of Fisheries Education, for providing all the facilities for carrying out this study. J.G. is thankful to the Indian Council of Agricultural Research, New Delhi, for the Junior Research Fellowship awarded to him.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
George, J., Purushothaman, C.S. & Shouche, Y.S. Isolation and characterization of sulphate-reducing bacteria Desulfovibrio vulgaris from Vajreshwari thermal springs in Maharashtra, India. World J Microbiol Biotechnol 24, 681–685 (2008). https://doi.org/10.1007/s11274-007-9524-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-007-9524-2