Summary
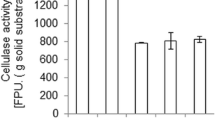
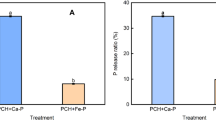
Celite is a unique variety of 2:1 clay mineral and is referred to as hydrous mica because it is believed to be the␣weathering product of mica. The present study describe the effect of celite on microbiological conversion of l-tyrosine to 3,4-dihydroxyphenyl-l-alanine (l-DOPA) by Aspergillus oryzae ME2. At optimum pH (pHo=6), and temperature (t=30 °C), 100% sugars were utilized for cell mass formation. Mould mycelium was used for biochemical conversion of l-tyrosine to l-DOPA because tyrosinases, β-carboxylases and tyrosine hydroxylases are intracellular enzymes. The maximum conversion of l-tyrosine to l-DOPA (0.428 mg/ml) was achieved after 60 min of reaction. To further enhance the production of l-DOPA, different concentration of celite were added to the reaction mixture. Best results were observed when the concentration of celite was 3.5 μM (1.686 mg l-DOPA produced/ml with 1.525 mg l-tyrosine consumed/ml). There was up to 3-fold enhancement in the product formation rate. This enhancement is the highest so far reported. Celite not only increased enzyme activity but also enhanced the permeability of the cell membrane to facilitate the secretion of enzymes in to the reaction broth. The comparison of kinetic parameters showed the ability of the mutant to yield l-DOPA (i.e., Y p/x 7.360±0.04 mg/mg). When the culture grown on various celite levels was monitored for Q p, Q s and q p, there was significant enhancement (P<0.025) in these variables over the control, which indicates that the study can be commercially applicable (HS, LSD 0.456).
Similar content being viewed by others
References
Andrews G., 2002. Estimating cell and product yields Biotechnology and Bioengineering 33: 256–265
Archer D.B., 2001. Genetic engineering: yeasts and filamentous fungi In Ratledge C., Kristiansen B., eds Basic Biotechnology 2nd edn. Cambridge University Press Cambridge pp. 95–126ISBN 0521779170
Arnow L.E., 1937. Colorimetric determination of the components of 3,4-dihydroxy phenyl l-alanine in tyrosine mixtures Journal of Biochemistry 88: 531
Bubbico R., DiMichele A., Verdone N., 2001. Selectivity in metabolic products of Yarrowia lipolyticaEnzyme and Microbial Technology 28: 189–195
Fling G., 1998. Radiation induced effects on l−3,4 dihydroxyphenylalanine by Aspergillus oryzae Journal of Analytical Chemistry 67: 185–187
Haneda K., Watanabe S., Takeda P., 1973. l-DOPA by microorganisms Journal of Fermentation Technology 51: 398–406
Haq I., Ali S., Manzoor S., Qadeer M.A., 2000. Optimization of cultural conditions for the production of l-DOPA from l-tyrosine by Aspergillus oryzaePakistan Journal of Botany 32: 255–258
Haq I., Iqbal F., Qadeer M.A., 1998. Production of l-DOPA from l-tyrosine by Aspergillus oryzaeBiologia 44: 31–39
James P., Fling G., 2001. Tyrosinase activity of different fungal strains Biochemistry and Biophysics 112: 98–103
Kumagai L.S., Fling M., Sarin A.N., 1969. Mushroom tyrosinases Journal of Biological Chemistry 68: 178–185
Nofre C., Cier A., Saucet C.M., Parent J., 1960. Action of free hydroxyl radicals on amino acidsLyon Medical 251: 81–82
Paul S.P., 1974. Amino acids and 3,4 dihydroxy phenyl l-alanine idiopathic actions Science and Medicines 43: 73–76
Pirt S.J., 1975. Principles of Cell Cultivation 2nd Edition. Blackwell Scientific Oxford, UK p. 116 ISBN 06322081503
Pontecorvo J., Roper J.A., Hemmous L.M., Buftan W., 1969. Basic techniques of UV-irradiation and chemical treatments Advances in Genetics 5: 141–183
Raju B.G., Rao G.H., Ayyanna C., 1993. Bioconversion of Tyrosine to Melanin using Mycelia of Aspergillus sppCBS Publishers Visakhapatnam India pp. 37–58 ISBN 0712916820
Rarveen J.K., Michael L.C. 1992, Clinical medicines. ELBS 2nd edn. CBS Publishers, London, UK. pp. 141, 210 ISBN 0723926134
Rosazza K.C., Paul G.M., Wolf S., 1995. The microbial sources of tyrosinases Biotechnology Letters 16: 210–215
Roy D., Das R.M., 1978. Mutation of microorganisms to complete change in physiological sequences Amniotic and Technological Chemistry 159: 172–183
Sehgal D., Gopinathan K.P., 2004. Molecular biology and biotechnological applications for large-scale synthesis of recombinant proteins Current Science 83: 455–465
Sih C.J., Foss P., Rosazza J., Lemberger M., (1969). Biosynthesis of l−3, 4-dihydroxy phenylalanine from mealworms Journal of the American Chemical Society 91: 6204
Singh H., 1999. Mechanism of oxidation of l-tyrosine by fungal tyrosinases Journal of Chemistry 172: 83
Snedecor G., Cochran W.G., 1980. Statistical Methods 7th Edition Iowa State University USA pp. 80–86 ISBN 0510003121
Toyama H., Yamada M., Shinagawa E., Matsushita K., 2003 New developments in oxidative fermentation Applied Microbiology and Biotechnology 60: 643–653
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ali, S., Ikram-ul-Haq Kinetic basis of celite (CM 2:1) addition on the biosynthesis of 3,4-dihydroxyphenyl-l-alanine (l-DOPA) by Aspergillus oryzae ME2 using l-tyrosine as a basal substrate. World J Microbiol Biotechnol 22, 347–353 (2006). https://doi.org/10.1007/s11274-005-9040-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-005-9040-1