Abstract
The research was designed to assess benefits of passive restoration, consisting of blocking from drainage ditches and in effect raise the groundwater level, its influence on properties of soil, water, species of plants and mesofauna in the fen Caltho-Alnetum community. The research was carried out in Outer Flysh Carpathians, the area of the Babiogórski National Park (Poland) in 2011–2014. It was established that the degraded communities showed positive reaction to an attempt to restore the original groundwater level. The increase in the groundwater level changed chemical properties of soil and water of the community such as: pH, sorption properties and the ionic composition of groundwater. Changes in habitat conditions resulted in different species composition of soil mesofauna and plants growing in the restorative community.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The mountain fens under Caltho-Alnetum community are priority biotopes listed in the annex to the 1st EU Habitats Directive (Council Directive 92/43/EEC1992), because they are rare, have high biodiversity, are imperiled and need the protection. They are some of the richest as regards flora and fauna ecosystems of the Babiogórski National Park (Parusel et al. 2004; Sterzyńska et al. 2014). They provide habitats for valuable and protected species of plants and animals (JoL 2012 No. 0 item 81; JoL 2011 No. 237 item 1419). In the 1960s and 1970s, the majority of those mountain fens suffered degradation due to drainage before they started to be protected. The main cause was the forestry management aimed at increasing timber production.
Drainage system of peat soil disturbs natural water relations (Dembek 2002; Grand-Clement et al. 2015), causes peat mass transformation to muck as well as results in enhanced greenhouse gases loss (Strack and Zuback 2013; Waddington et al. 2015). Changes in peat evolution under melioration processes are mostly characterized by mineralization and secondary humification. The above processes lead to changes in the morphological, chemical, biological, and physical properties of soils (Boguta and Sokołowska 2014). Similar relations in soils of mountain hydrogenic habitats have been described by: Nicia (2009), Nicia et al. (2009), Chimmer et al. (2010), Nicia and Bejger (2012) and Zhang et al. (2012).
A good indicator showing the degree of degradation of hydrogenic habitats is soil mesofauna which is sensitive to environmental change and could quickly respond to habitat disturbances, such as seasonal drought, air warming (Xu et al. 2012) or changes in hydrologic regime in a river floodplain (Sterzyńska et al. 2014). Soil mesofauna plays an important role in soil formation, transformation and decomposition of soil organic matter, distribution of soil particles, water infiltration rate, nutrient cycling, mineralization, immobilization, the availability of N and other nutrients, and the composition, abundance and dispersion and activity of bacteria and mycorrhizal fungi (Menta 2012; Briones 2014). In fact, changes in soil mesofauna communities may be detected prior to changes in physical or chemical soil properties making soil fauna useful indicators of successful or detrimental reclamation activities, including soil mesofauna as indicators of restoration of drainage ditches and water diversions in mountain fens.
Drained hydrogenic habitats require restoration. Restoration processes of plains and lowland areas involve: (1) removing the peaty–muck surface layer; (2) removing of dominant vegetation and (3) reintroduction of characteristic plant species as well as (4) increasing groundwater level (Grand-Clement et al. 2015). Such restoration processes are not possible in inaccessible Caltho-Alnetum habitats. It is mainly because of the following factors: (1) presence of valuable natural habitats, e.g. Dentarioglandulosae–Fagetum, which can be damaged while removing the peaty–muck surface layer; (2) land configuration—large inclinations and difficulties in transporting the peaty–muck surface layer and (3) risk of triggering erosion.
In the selected patches of degraded and later passive restored mountain fens changes in the groundwater level, water and soil physicochemical parameters, vegetation and soil mesofauna have been examined.
Materials and methods
Study area
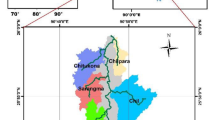
The study covered a three degraded habitats (as repeated measurements) of Caltho-Alnetum in the Babiogórski National Park (Fig. 1) in Outer Flysh Carpathians, Poland situated at the level of the lower montane zone on the slope of about 7°. The area was drained using ditches of 0.2–0.7 m in depth. The identification of typical species of Caltho-Alnetum for example: Caltha paluistris, Valeriana simplicifolia, Agropyron caninus, Alnus incana was carried out directly only at the least drained parts of the habitats.
Method of mountain fens restoration
In 2011, restoration processes started with preventing water drainage from existing ditches by applying valves made of dried spruce and ash and filling them with dry leaves (Fig. 2). Valves have different heights depending on the depth. Their height reached 0.6 m. The valves were made of hermetically sealed branches and trunks. The number of valves was varied depending on the slope of the restored habitats. In the area of low slope (1–3%), there were 2–3 valves per 10 running meters of drainage ditch, while in the area with higher slope (3%) even 5–7 valves were used. A total of about 220 valves in the area of three restored habitats were used. Water level was increased gradually to avoid water erosion that could result in sliding of the peaty–muck surface layer and finally lead to a complete degradation of the restored habitats.
Sampling of soils and groundwater levels
Representative soil samples in the central point of each studied habitats were collected from the following depths: (1) 0–18 cm, (2) 18–55 cm and (3) < 55 cm in 2011 and 2014. To control the increasing groundwater level and water samplings, near the each soil profile, three piezometers were installed.
Chemical analysis of soil and groundwater
The soil samples were air dried and milled for chemical analysis. The following parameters were determined by standard methods: (1) pH in a 1:2.5 (w/v) fresh soil: water suspension and 1 mol L−1 KCl solution using the potentiometric method; (2) the base cation (Ca2+, Mg2+, K+, and Na+) concentration, after extraction with 1 mol L−1 CH3COOHN4 solution using Perkin Elmer Optima 7300DV ICP-OES atomic emission spectrometer; (3) hydrolytic acidity (Hh) by the Kappen method; (4) organic carbon (Corg.) content by the Tiurin method;(5) total nitrogen (Ntot.) by the Kjeldahl method. Total exchangeable base (TEB) cation concentration, cation exchange capacity (CEC) and base saturation (BS) percentage were calculated as follows:
The following parameters were measured in water samples from piezometers PI–PII: temperature, electrical conductivity and ion composition according to Dojlido (1999). Mineralization was calculated as a sum total of ions (Tölgyessy 1993; Nikanorov and Brazhnikova 2009). Concentration of HCO3 − ions was measured by titrating. Concentration of SO4 2−, Cl−, NO3 −, NH4 + and PO4 3− ions was measured using Merck Spectroquant Pharo 100 spectrophotometer. Concentration of Ca+, Mg2+, Na+ and K+ ions were measured using Perkin Elmer Optima 7300DV ICP-OES atomic emission spectrometer.
Sampling of soil mesofauna
Soil mesofauna was sampled from the central point of each studied habitat in 2010–2012 and 2012–2014. At fen site soil mesofauna was sampled two times a year in spring and autumn. Collembola in degraded sites were sampled in years 2010–2012 and in restored sites in years 2012–2014; Acari, were sampled in years 2010–2012 in degraded sites and in restored fens in 2012–2013. For Collembola—three random litter + soil cores were taken from each site at each sampling occasion with a steel corer (diam. 5.5–10 cm depth). Collembola from soil and litter layers were extracted in a modified MacFadyen high gradient apparatus for a maximum of 10 days. All specimens are deposited at the Museum and Institute of Zoology PAS (Warsaw, Poland). For Acari three replicate samples randomly taken from each site were analyzed (diam. 5–5 cm depth). Acari were extracted using a modified high-gradient Tullgren funnels (Marshal 1972), fixed in 80% ethanol and determined to following groups: Acaridida, Actinedida, Gamasida, Oribatida. Further, the most abundant taxon Oribatida, was cleared in temporary slides with 80% lactic acid and identified at the species level. The material was transferred into glycerol and deposited in the comparative collection of the Institute of Soil Biology, AS CR, České Budějovice, Czech Republic.
To determine the influence of restoration processes of flora species in the habitat in 2011, 2012 and 2014, censuses of species were developed at 35 research areas, in the immediate vicinity of valves fitted in ditches. The composition of flora was compared with the characteristic combination of species at a Caltho-Alnetum community as described by Matuszkiewicz (2005).
Statistical analysis
The obtained results from the 4-year observations (2011–2014) were statistically analysed using the Statistica 10.0 software. Prior to analysis, data were tested for homogeneity of variance (Levene’s test) and for normal distribution (Kolmogorov–Smirnov test). Among the means, One-way analysis of variance (ANOVA) and Tukey’s test (P ≤ 0.05) were used to determine significant differences. The changes in community structure of soil mesofauna the two-tailed nonparametric Mann–Whitney U test (data failed the test for normality) was used to assess the differences in species density (D), species richness (S), Shannon’s diversity index (H′) and Pielou evenness (J′).
Results and discussion
While considering morphological and chemical properties of mountain fens under Caltho-Alnetum community, according to WRB (2015), the soils can be classified as Murshuric Drainic Sapric Histosols (Reic).
Before restoration processes started, the surface layers showed lower pH (Table 1) comparing to Caltho-Alnetum habitat soils of natural water relations as examined by Nicia et al. (2009). Lower pH of soil examined as compared to the soil of natural water relations was due to the reduced groundwater level and zero inflow of water with Ca2+ and Mg2+. pH values increased with depth of the soil profile. Higher pH values in deeper layers can be explained by the saturation of those layers with water rich in Ca2+ and Mg2+ which neutralize acidic products from organic matter decomposition (McCauley et al. 2009; Zeng et al. 2011).
Surface layers of the drained Caltho-Alnetum habitats also had lower values of such factors as: TEB, CEC, BS comparing to layers determined in deeper within the range of groundwater (Table 1). The content of Ca2+ and Mg2+ in the sorption complex in drained surface layer soils in was comparable with that in the drained Caltho-Alnetum habitat soils examined by Nicia et al. (2010) and was lower than in the case of organic soils in the phase of accumulation described by Nicia and Miechówka (2004).
Before restoration processes, according to the concentration of mineral substances, water in the studied habitats could be classified, based on criteria specified in the Regulation of the Minister of Health of 31st March 2011 on natural mineral water, natural spring water and table water, as low mineralized (mineralization from 50 to 500 mg L−1). While based on the ion concentration (Table 2), water before restoration processes could be classified as a calcium–sulfate (concentration of calcium cations and sulfate anions exceeds 20% mval) type of hydrogeochemical class 6 acc. Szczukariewa–Prikłońskiego (Macioszczyk 1987).
Blocking the flow of water in drainage ditches resulted in the increase in groundwater level within 4 years from 26.5 to 9.2 cm from the topsoil level, bringing it closer to the level considered to be natural in this type of habitats.
After the water level was raised, the studied habitat showed statistically significant increase of NO3 −, CO2(HCO3)−, mineralization and EC (Table 2). Although, the content of other ions (except NH4 − and PO4 3− which concentration decreased) increased, the increase was not statistically significant. The increase in the concentration of those ions after watering of the soil profile was due to the release of ions to the soil solution from decomposed organic matter (Holden et al. 2011).
After restoration processes, selected soil properties also changed (Table 1). In comparison to 2011, surface layers showed higher content of organic matter, organic carbon (Corg), C/N, pH, higher concentration of K+, Ca2+ and Mg2+ in the sorption complex, and higher BS. Decrease was recorded regarding soil ash content in surface layers, total nitrogen, Hh and CEC. The statistical analysis of results showed significant changes of ash content, Corg, and K+, Hh and BS.
The reduced of ash content in organic layers accompanied by increased organic carbon content resulted not only from restoration processes. Considering the rate of increase in organic matter content in mountain fen soils would not be possible in such a short time if it was not for the increase in organic carbon content in surface layers by about 5% comparing with the status before restoration processes. The above changes are attributable to two factors: (1) increased water level in the studied habitat and the changes flora structure. This stimulated the surface accumulation of organic matter and retention of biomass from leaves from neighboring habitants. The higher moisture soil content stimulated the retention of leaves which were wet and thus could not be shifted beyond the studied habitat by wind and runoffs; (2) surface erosion of little intensity leading to enrichment of surface layers with organic matter washed out from upper sections of the studied habitat. Similar relations applicable to the retention of biomass were described by other authors (Koreleski 2005; Dąbek et al. 2014).
The increase in the groundwater level in the mountain fen soils led to statistically significant increase of pH and the content of K+, Ca2+ and Mg2+ in the sorption complex, which contributed to higher BS (Table 1). The increase in those parameters is related to the supply of base components to surface layers due to a higher water level in the mountain fen soils. After the level of groundwater increased, Ca2+ and Mg2+ present in the inflowing water could gradually neutralized acidic products from organic matter decomposition, which led to higher pH and saturation of the sorption complex with base cations.
The higher groundwater level was also reflected in a different fauna and flora composition in the soil of the habitat (Table 3, Fig. 3). A total of 69 collembolan species and 86 oribatid mite species were identified across all six sites of degraded and regenerated mountain fens. The significant lowering in the mean values of total mesofauna, total Acari, Gamasida and Oribatida density occurring in restored mountain fens, and higher evenness of Oribatida communities, indicated on a significant change in oribatid mite species abundance distribution due to restoration. The soil measofana functional group, using the body width as the main classificatory criterion, includes soil microarthropods taxa below < 2 mm (Briones 2014). It is assumed that this functional group of soil biota exerts direct and indirect influence on soil biogeochemical processes, including litter decomposition and nutrient cycling, and contributes to top-down control of primary (bacteria, fungi) and secondary (nematodes Protoza) decomposers (Petersen and Luxton 1982; Wardle 2002; Coleman et al. 2004). Several studies indicated density-dependent relationship between soil mesofauna and microbial biomass (González et al. 2001; Lenoir et al. 2007; Crowther and A’Bear 2012). This fauna–microbe interactions are exhibited in the current soil biogeochemical models (Grandy et al. 2016). Nonetheless, the soil mesofauna represent coarse groupings that do not take into account the fact that different soil organisms within the same size group could exhibit different life history strategies or physiological capabilities (Coleman et al. 2004) and that their relative importance in performing a specific function could change across temporal and spatial scales. For example, Acari have a diversity of functions demonstrated by the range of feeding guilds to which they belong, including predators (Gamasida), fungal and bacterial feeders (Acaridida, Oribatida) or taxa demonstrating a variety of feeding habits (Actinedida), Further, Oribatida and Collembola exploit the same class of environmental resources in a similar way as detritivorous and fungivorous feeders; however, they differ in life-history traits. Collembola generally possess traits that are characteristic for r-selected animals in view of their relative small size, short generation time and high fecundity in contrast to k-selected oribatid mites with low metabolic rates, slow development and low fecundity (Behan-Pelletier 2002).
Our results also showed different reaction of soil mesofauna groups to mountain forested fen restoration, and indicated on a more sensitive response of Oribatida than Collembola communities.
Our results clearly demonstrated that restoration of natural hydrologic regime, and increasing level of soil saturation in mountain fens significantly reduce abundance of total soil mesofauna, total Acari and selected groups within Acari, such as Gamasida and Oribatida, however, have no effect on the abundance of collembolan communities. The increasing level of soil saturation also does not change significantly the diversity of soil fauna communities, with exception of increasing evenness of Oribatida communities. This finding is consistent with van Dijk et al. (2009) observation that reducing decomposition and mineralization of organic matter by increasing groundwater level could lead to differences in soil fauna communities. We can conclude that the observed changes in soil mesofauna communities support the thesis that restoration process was successful in mountain fens.
During restoration processes, the composition of flora in the studied habitat changed. In 2011–2014, it was established that 32 out of 35 patches near the valves developed flora typical for the habitat. The number of flora species near ditches increased from 11 to 16, respectively in 2011 and 2014. The marshland species of flora prevailed, chiefly hemicriptophite (12 species). Ten out of sixteen vascular plants belonged to a characteristic combination of species of the Caltho-Alnetum association (Matuszkiewicz 2005) (Fig. 2). Restoration processes led to establishing new microhabitats which initiated succession of plants towards the Caltho-Alnetum association.
In conclusion the increased groundwater level in the studied habitat concerned resulted in a statistically significant change of such chemical soil properties as: ash content, organic matter and organic carbon content, pH, Hh, and BS. Our results clearly demonstrated that restoration processes of natural hydrologic regime, and increasing level of soil saturation in mountain fens significantly reduce abundance of total soil mesofauna, total Acari and selected groups within Acari, such as Gamasida and Oribatida, however, have no effect on the abundance of collembolan communities. The increasing level of soil saturation also does not change significantly the diversity of soil fauna communities, with exception of increasing evenness of Oribatida communities. We can conclude that the observed changes in the soil mesofauna community allow for estimating wetland restoration efforts. The decrease in abundance of the soil mesofauna community indicates a slowing of organic matter decomposition and alteration of soil microbial communities, both parameters being of primary interest for estimating wetland restoration success (e.g. Bossio et al. 2006). The observed differences in response between Collembola and Oribatida, two taxa competing for the same food resources but differing in life history traits, in consistently submerged restored wetlands also support the claim that restoration processes have been successfully initiated in mountain fens. The change of habitat conditions resulted in the succession of plants typical for the Caltho-Alnetum habitats near ditches with valves that limited the outflow of water.
References
Behan-Pelletier VM (2002) Acari and Collembola biodiversity in Canadian agricultural soils. Can J Soil Sci 83:279–288
Boguta P, Sokołowska Z (2014) Statistical relationship between selected physicochemical properties of peaty-muck soils and their fraction of humic acids. Int Agrophys 28(3):269–278
Bossio DA, Fleck JA, Scow KM, Fujii R (2006) Alteration of soil microbial communities and water quality in restored wetlands. Soil Biol Biochem 38:1223–1233
Briones MJI (2014) Soil fauna and soil functions: a jigsaw puzzle. Front Environ Sci 2:7
Chimmer RA, Lemly JA, Coope DJ (2010) Mountain fen distribution, types and restoration, priorities, San Juan Mountain, Colorado, USA. Wetlands 30:763–771
Coleman DC, Crossley DA, Hendrix PF (2004) Fundamentals of soil ecology. Elsevier, Amsterdam
Council Directive 92/43/EEC, of 21 May 1992 on the conservation of natural habitats and of wild fauna and flora
Crowther TW, A’Bear AD (2012) Impacts of grazing soil fauna on decomposer fungi are species-specific and density-dependent. Fungal Ecol 5:277–281
Dąbek P, Żmuda R, Ćmielewski B, Szczepański J (2014) Analysis of water erosion processes using terrestrial laser scanning. Acta Geodyn Geomater 11(1 (173)):45–52
Dembek W (2002) Wetlands in Poland: present threats and perspective for protection. J Water Dev 6:3–17
Dojlido J (1999) Physico-chemical examination of water and wastewater. Arkady Publishers, Warsaw (in Polish)
González G, Ley RE, Schmidt SK, Zou X, Seasted TR (2001) Soil ecological interactions: comparisons between tropical and subalpine forests. Oecologia 128:549–556
Grand-Clement E, Anderson K, Smith D, Angus M, Luscombe DJ, Gatis N, Bray LS, Brazier RE (2015) New approaches to the restoration of shallow marginal peatlands. J Environ Manag 161:417–430
Grandy AS, Wieder WR, Wickings K, Kyker-Snowman E (2016) Beyond microbes: are fauna the next frontier in soil biogeochemical models? Soil Biol Biochem 102:40–44
Holden J, Wallage ZE, Lane SN, McDonald AT (2011) Water table dynamics in undisturbed, drained and restored blanket peat. J Hydrol 402(1–2):103–114
Koreleski K (2005) Selected problems of antierosion soil protection in the light of sustainable development. Acta Agrophys 5(1):49–55 (in Polish)
Lenoir L, Persson T, Bengtsson J, Wallander H, Wirén A (2007) Bottom-up or top-down control in forest microcosms? Effect of soil fauna on fungal biomass and C/N mineralization. Biol Fertil Soils 43:281–294
Macioszczyk A (1987) Hydrogeochemistry. Geological Publishers, Warsaw (in Polish)
Marshall VG (1972) Comparison of two methods of estimating efficiency of funnel extractors for soil microarthropods. Soil Biol Biochem 4:417–426
Matuszkiewicz W (2005) Guide for marking of Polish plant communities. National Scientific Publishers, Warsaw (in Polish)
McCauley A, Jones C, Jacobsen J (2009) Soil pH and organic matter. Nutr Manag 8:1–12
Menta C (2012) Soil fauna diversity—function, soil degradation, biological indices, soil restoration. In: Lameed GA (ed) Biodiversity conservation and utilization in a diverse world. InTech, New York
Nicia P (2009) Characteristics and problems of mountain and submontane fens protection. In: Łachacz A (ed) Contemporary problems of management and environmental protection. Wetlands—their functions and protection. Department of Land Reclamation and Environmental Management, University of Warmia and Mazury, Olsztyn
Nicia P, Bejger R (2012) Characteristics and problems of protection of mountain fen soils with diversified type of hydrogenic feeding. In: Szajdak LW, Gaca W, Meysner T, Styła K, Szczepański M (eds) Necessity of Peatland Protection. Institute for Agricultural and Forest Environment, Polish Academy of Sciences, Poznań
Nicia P, Miechówka A (2004) General characteristics of eutrophic fen soil. Pol J Soil Sci 37(1):39–47
Nicia P, Zadrożny P, Lamorski T (2009) General characteristic of selected soil profiles under the Caltho-Alnetum Association in the Babiogórski National Park. Ecol Chem Eng A 16(7):839–843
Nicia P, Zadrożny P, Lamorski T, Bejger R (2010) The properties of soils and waters in Krowiarki fen under Caltho-Alnetum community in the Babiogórski National Park. Woda-Środowisko-Obszary Wiejskie 1(29):123–132 (in Polish)
Nikanorov AM, Brazhnikova LV (2009) Types and properties of water, 2: Water chemical composition of rivers. Encyclopedia of life support systems. Hydrochemical Institute of the Federal Service of Russia, Rostov-on-Don
Parusel J, Kasprowicz M, Holeksa J (2004) Forest and brushwood communities in the Babiogórski National Park. In: Wołoszyn BW, Jaworski A, Szwagrzy J (eds) The nature of the Babiogórski National Park. Monograph. Babiogórski National Park, Kraków, pp 179–196 (in Polish)
Petersen H, Luxton M (1982) A comparative analysis of soil fauna populations and their role in decomposition processes. Oikos 39:287–388
Regulation of the Polish Minister of Health of 31 March 2011 on natural mineral waters, spring waters and table waters (Dz. U. Nr 136, poz. 914, Nr 182, poz. 1228 and Nr 230, poz. 1511) (in Polish)
Regulation of the Polish Minister of the Environment of 12 October 2011 on the protection of animal species (Dz.U. 2011 Nr 237 poz. 1419) (in Polish)
Regulation of the Polish Minister of the Environment of 5 January 2012 on the protection of plant species (Dz.U. 2012 Nr 0 poz. 81) (in Polish)
Sterzyńska M, Shrubovych J, Kaprus I (2014) Effect of hydrologic regime and forest age on Collembola in riparian forests. Appl Soil Ecol 75:199–209
Strack M, Zuback YCA (2013) Annual carbon balance of a peatland 10 yr following restoration. Biogeosciences 10(5):2885–2896
Tölgyessy J (ed) (1993) Chemistry and biology of water, air and soil: environmental aspects. Studies in environmental science, vol 53. Elsevier, Amsterdam
Van Dijk J, Didden WAM, Kuenen F, van Bodegon PM, Verhoeff HA, Aerts R (2009) Can differences in soil community composition after peat meadow restoration lead to different decomposition and mineralization rates? Soil Biol Biochem 41:1717–1725
Waddington JM, Morris PJ, Kettridge N, Granath G, Thompson DK, Moore PA (2015) Hydrological feedbacks in northern peatlands. Ecohydrology 8(1):113–127
Wardle DA (2002) Communities and ecosystems: linking the aboveground and belowground components. Princeton University Press, Princeton
WRB (2015) World reference base for soil resources 2014, update 2015. International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106, FAO, Rome
Xu G-L, Kuster TM, Gűnthardt-Goerg MS, Dobbertin M, Li M-H (2012) Seasonal exposure to drought and air warming affects soil Collembola and mites. PLoS ONE 7(8):e43102
Zeng F, Ali S, Zhang H, Ouyang Y, Qiu B, Wu F (2011) The influence of pH and organic matter content in paddy soil on heavy metal availability and their uptake by rice plants. Environ Pollut 159(1):84–91
Zhang X, Liu H, Baker Ch, Graham S (2012) Restoration approaches used for degraded peatlands in Ruoeragi (Zoige), Tibetan, Plateau, China for sustainable land management. Ecol Eng 38:86–92
Acknowledgements
This research was supported by National Centre of Sciences (NCS), Poland, Projects Nos. N N305 107540, N N304 156240 and N N305 396838.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Paweł, N., Romualda, B., Maria, S. et al. Restoration of hydro-ecological conditions in Carpathian forested mountain fens. Wetlands Ecol Manage 26, 537–546 (2018). https://doi.org/10.1007/s11273-017-9590-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-017-9590-7