Abstract
Metal enrichment in aquatic ecosystems has compromised the potential of fish to enhance food security. The uMgeni River drains urban and industrialized catchment and flows through economically disadvantaged rural communities that opt for fish to supplement their protein needs. However, there are uncertainties on the safety of consuming fish from the uMgeni River. The present study aims to explore metal distribution between the water, sediment, and tissues of the preferred Oreochromis mossambicus, and evaluate whether concentrations in the muscle are safe for human consumption. The water, sediment, and fish samples were collected from Inanda and Nagle dams, and metal concentrations were analysed using inductively coupled plasma mass spectrometry. Alkaline pH was observed at both dams and metals were below detectable level in the water column. Even though metal concentrations in the water column were below detectable levels, significant concentrations were found in the sediment. No consistent trend in metal concentrations was observed across tissues, but higher levels were generally found in the liver, followed by the gill, and then the muscle. Nickel (Ni) and lead (Pb) exhibited concentrations exceeding the permissible limit for human consumption. Similarly, the target hazard quotient exceeded the threshold of 1 for Pb, chromium (Cr), and antimony (Sb) at both dams with Inanda Dam showing higher indices for Sb and Pb. The carcinogenic risk for the three metals was also found to be higher than 10–6. These findings suggest that consumption of O. mossambicus from the uMgeni River should be limited to no more than 150 g per week.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Fish are regarded as inexpensive protein sources for communities residing in the vicinity of rivers and dams (Esilaba et al., 2020). However, fish consumption is reported to be one of the routes of metal exposure to humans due to their capacity to accumulate metals when inhabiting contaminated environments (Kumar et al., 2022; Pal & Maiti, 2018). The accumulation of metals may be influenced by numerous factors i.e. pH, conductivity, temperature, salinity, fish age and size, sex, and the presence of other metals (Bawuro et al., 2018). According to Jezierska and Witeska (2006), fish length may exhibit an inverse relationship with metals due to dilution whereas the metabolic capacity of smaller fish makes them susceptible to metal accumulation. Moreover, different fish organs accumulate metals differently depending on their affinity, location relative to the medium, and the duration of exposure. The liver, kidney, gills, and spleen have shown to accumulate higher concentrations compared to muscle (Debipersadh et al., 2023; Gilbert et al., 2017; Njinga et al., 2023). However, muscle is included in metal accumulation studies as it is the part consumed by humans (Şirin et al., 2024).
Metals such as Sb, cadmium (Cd), Cr, molybdenum (Mo), Ni, and Pb are potential threats to human health and they are also documented by the United States Environmental Protection Agency (USEPA) as metals of concern during the Anthropocene era (Heath et al., 2004; US-EPA, 2000). Studies reported increased concentrations of these metals in water bodies impacted by agricultural activities (Han & Gu, 2023), wastewater effluents (Soleimani et al., 2023), mining (Lakra et al., 2019; Mishra et al., 2008), acid mine drainage (Lebepe et al., 2020), and industrial activities (Adegbola et al., 2021). It is, therefore, imperative to assess the behaviour and concentrations of these metals in aquatic biota, particularly those receiving water from impacted catchments.
The uMgeni River system is one of the important freshwater systems supporting over 10 million people in South Africa. Its middle stretch which includes the Nagle and Inanda dams is experiencing extensive artisanal fishery by local communities. Studies reported high metal concentrations of Sb, Cd, Cr, Mo, Ni, and Pb in the surface water and sediment in Inanda Dam which were associated with the inflow from a highly polluted uMsunduzi River (Mdluli et al., 2023; Olaniran et al., 2014). These metals were found to be problematic in most river systems draining industrialized catchment (Adegbola et al., 2021; Hao et al., 2022), and they are among the metals that resulted in fish advisories in other parts of the world (Heath et al., 2004). However, no study was conducted in the uMgeni River to evaluate the safety upon consuming the inhabitant fish. Therefore, the present study aims to evaluate concentrations of these selected metals in one of the preferred fish species, Oreochromis mossambicus (Peters, 1852). and check whether they are safe for human consumption. The first hypothesis was that fish from both dams would exhibit a common descending trend of liver > gill > muscle for all metals. Based on the location of the Inanda and Nagle dams relative to the uMsunduzi River, we also hypothesized that metal concentrations would be higher in fish from Inanda Dam compared to Nagle Dam with fish from the former exhibiting concentrations exceeding acceptable levels for human consumption.
2 Materials and Methods
2.1 Sampling Localities
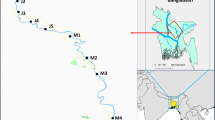
Fish were sampled at Inanda and Nagle dams of the uMgeni River system in KwaZulu-Natal, South Africa. The Inanda Dam is bordered by rural communities that practice subsistence fishery to supplement their protein needs. Moreover, the dam receives water from a heavily polluted uMsunduzi River and the two smaller streams (Fig. 1) (a: uMngcweni River and b: Sikelekehleni River). The water quality in the Inanda Dam has been deteriorating over the past few decades (Gumbi et al., 2017). Olaniran et al. (2014) recorded elevated metal concentrations in Inanda Dam. In contrast, Nagle Dam is located approximately 49 km upstream, before the uMsunduzi-uMgeni rivers confluence. Moreover, the dam is known for its better water quality compared to Inanda Dam due to negligible anthropogenic activities along the river (Adeyinka, 2014; Oosthuizen & Ehrlich, 2001).
2.2 Water and Sediment Quality Assessment
The physical parameters of water were measured using the HANNA multi-parameter instrument during wet and dry seasons in 2020 and 2021. Water samples were then collected using polyethylene sampling bottles and refrigerated until processing and analysis. During analysis, water was acidified using hydrochloric acid and filtered through a 0.45 µm membrane. Filtered water was analysed for metals using Inductively coupled plasma mass spectrometry (ICP-MS, Agilent 7900). The guidelines by the Canadian Council of Ministers of the Environment (CCME, 2001, 2012) and the Department of Water Affairs and Forestry (DWAF, 1996) were used for evaluating water quality.
Similarly, sediment samples were collected using AMS Shallow Water Bottom Dredge sampler. Three grabs were made at each site and mixed to form a composite and stored in a 1 L polyethylene sampling bottles. Three composites were collected from each site. Samples were kept in the fridge for metal analyses. Sediment processing was done following the protocol described in Misra et al. (2024). Approximately 0.2 g of sediment samples were digested using aqua regia, 3-hydrochloric:1-nitric acid (3HCL:1HNO3) supplemented with 30% hydrogen peroxide (H2O2) for digestion of organic matter. The digested solution was double filtered with 0.45 µm pored membranes and syringe filters. The filtered sample was topped to 250 ml mark using deionized water. Metal analysis was done using ICP-MS (Agilent 7900).
2.3 Fish Sample Collection
Fish were caught using gillnets with different mesh sizes (45 to 90 mm stretch) and electro-shocker. Fifteen fish were caught at Inanda Dam over two seasons whereas 12 were caught at Nagle Dam. Fish were euthanized by cutting through the spinal cord and weighed using a digital spring scale. The lengths were measured using a measuring tape. Fish were then opened ventrally for harvesting of other organs such as liver and gills. Muscle tissue was also harvested as it is the primary organ consumed by humans.
2.4 Fish Sample Digestion and Metal Analysis
Approximately 0.2 g of fish tissues were sectioned and digested following the method described in Misra et al. (2024) using aqua-regia. Samples were heated until the brown smoke faded and then allowed to cool at room temperature. After cooling, the samples were double filtered using 0.45 μm pore membranes and syringe filters, then topped off to the 250 ml mark with deionized water. Solutions were kept in the fridge until analysis using ICP-MS.
2.5 Quality Control
All samples were digested with blanks and multi-element standards were also used during analyses. The standards ranged from 0.01 – 5 mg L−1 for Sb, Cd, and Pb, and 0.1 – 10 mg L−1 for Cr, Mo, and Ni. The accuracy of the analytical technique was determined by analysing certified reference materials (CRMs) with every 5 samples analysed. For fish tissues, the certified reference materials for fish (DORM-4) were used. The recovery in all these analyses ranged from 93—103% (Table 1) and the detection limit of the instrument was 0.001 mg L−1.
2.6 Bioaccumulation Factor
The bioaccumulation factor (BAF) is the ratio of metal concentration in the organ to the medium, either water or sediment (Arnot & Gobas, 2006). It may be used to determine whether bioaccumulation has occurred (Melake et al., 2023). It was calculated using the Eq. 1 as per Arnot and Gobas (2006).
2.7 Non-Carcinogenic and Carcinogenic Risk Assessment
The non-carcinogenic risk assessment was carried out by calculating the target hazard quotient (THQ) following US-EPA (2000) protocols as described in Gilbert et al. (2017) and Lebepe et al. (2020). Parameters used for calculating the THQs are presented in Table 2. The calculation of THQs were done using Eq. 2 whereas the carcinogenic risk index (CRI) was calculated out using Eq. 3.
where C is the metal concentration in mg/kg wet weight, EF: exposure frequency (days per year), ED: exposure duration (years), IRF: ingestion rate (kg per week), RfD: reference dose (mg/kg), BW: body weight (kg), AT: average time (days) and OSF: oral slope factor. The non-carcinogenic health risks are probable if the THQ is > 1 whereas no health risk are expected for THQ of < 1 (US-EPA, 2000). The slope factor available in the literature is only for Cd, Cr, and Pb with the maximum acceptable index being 10–4 (US-EPA, 2013).
2.8 Data Analysis
Statistical analysis was done using various packages of R-4.3.1 (R Development Core Team, 2014). Homogeneity of variance and normality were determined using Levene’s and Shapiro–Wilk tests, respectively. Depending on the satisfaction of the assumption, the Mann–Whitney U Test or independent sample t-test was used to compare the difference between means of concentrations in sediment and fish tissues. The non-metric multidimensional scaling (NMDS) was performed using a vegan package (Oksanen et al., 2013). The correlation was determined using Pearson or Spearman’s tests using the PerformanceAnalytics package (Peterson et al., 2020). Statistical results were considered significant at p < 0.05.
3 Results
3.1 Metals in the Water and Sediment
Antimony and Cd showed mean concentrations of 1.48 mg L−1 and 0.001 mg L−1 in the water column at Inanda Dam whereas other metals were below detection level at both dams. However, sediment exhibited notable metal concentrations at both dams (Fig. 2). Antimony was significantly higher at Inanda Dam compared to Nagle Dam (w = 20, p < 0.05). Similarly, Cd exhibited a significantly higher concentration at Inanda Dam compared to Nagle Dam (w = 25, p < 0.05). No significant differences were observed for Cr (w = 25.5, p > 0.05) and Ni (w = 6, p > 0.05) between the two dams. In contrast, a significantly higher concentration was observed for Mo at Inanda Dam (w = 25, p < 0.05). Coinciding Ni trend, Pb has also showed a significantly higher concentration at Inanda Dam (w = 31, p > 0.05).
4 Metal Concentrations in Fish Tissues
4.1 Comparison of Metals in Organs Between Dams
Metal concentrations observed in the tissues of O. mossambicus are shown in Fig. 3. The gill showed significant differences for Sb (t = 2.31, p < 0.05), Cr (t = 3.32, p < 0.05), Mo (t = 3.87, p < 0.05), Ni (t = 4.59, p < 0.05) and Pb (t = 2.35, p < 0.05) with Sb and Pb exhibiting relatively higher concentrations at Inanda Dam and Cr, Mo and Ni showing higher concentrations at Nagle Dam (Fig. 3). In contrast, no significant difference was observed for Cd (t = 1.52, p > 0.05) in the gills between the two populations. In the muscle, significant differences were observed for Sb (t = 2.34, p < 0.05), Cr (t = 2.32, p < 0.05), Mo (t = 2.39, p < 0.05), Ni (t = 1.95, p < 0.05) and Pb (t = 2.97, p < 0.05). A trend similar to that of the gills was observed for muscle where Sb and Pb showed relatively higher concentrations in the Inanda Dam population with Cr, Mo and Ni showing higher concentrations at Nagle Dam. Moreover, Cd concentration in the muscle showed no significant difference between the two populations (t = 1.66, p > 0.05). A different trend was observed for the liver with only Cr (t = 2.28, p < 0.05) and Mo (t = 2.57, p < 0.05) showing significant differences between the two populations. Higher concentrations for both metals were observed in the Inanda Dam population compared to Nagle Dam. No significant differences were observed for Sb (t = 1.77, p > 0.05), Cd (t = 1.74, p > 0.05), Ni (t = 0.72, p > 0.05) and Pb (t = 0.16, p > 0.05) between the two dams.
Coinciding with the trends described, NMDS showed no clear separation for metal concentrations in the liver (p > 0.05) between the two dams and the dispersion was also not significant (p > 0.05) with average distances to median being 0.35 for Inanda Dam and 0.37 for Nagle Dam. In contrast, gills and muscle exhibited clear separations between the two populations (p < 0.05) with non-significant dispersion within each group (Fig. 4). The gill average distance to median was 0.29 for Inanda Dam and 0.24 for Nagle Dam whereas the average distance to median in the muscle was 0.25 in Inanda Dam and 0.28 at Nagle Dam.
4.2 Comparison of Metals Between Tissues in Each Dam
Descending trends liver > gill > muscle for Sb, Ni and Pb, muscle > liver > gill for Cd, liver > muscle > gill for Cr and muscle > gill > liver for Mo were observed at Inanda Dam (Fig. 3). At Nagle Dam, descending trends liver > gill > muscle for Sb, Cd, Ni and Pb, and muscle > gill > liver for Cr and Mo were observed at Nagle Dam (Fig. 3). An NMDS showed liver being separated from the gill and muscle at Inanda Dam (p < 0.05) (Fig. 4) with average distances to median being 0.35, 0.29 and 0.31 for the liver, gills and muscle, respectively (p > 0.005). In contrast, dispersion at Nagle Dam showed a significant difference between organs (p < 0.05) (Fig. 4) with average distances to median being 0.37, 0.22 and 0.44 for the liver, gill and muscle, respectively. Nevertheless, MANOVA showed a significant difference between groups (p < 0.05) (Fig. 4).
4.3 Bioaccumulation Factor
Bioaccumulation factors were not calculated for tissue-water concentrations as metals were below detection levels in the water. Sediment showed notable concentrations, hence, bioaccumulation factors were calculated. The bioaccumulation factor for tissue-sediment concentrations were generally higher at Nagle Dam compared to Inanda Dam (Table 3). Only Cr in the liver and muscle exhibited a higher bioaccumulation factor at Inanda Dam (Table 3).
4.4 Inter-Metal and Metal-Fish Length Correlation
The gills and muscle showed strong to moderate significant relationships for Cr–Mo, Cr-Ni, Mo-Ni and Sb-Pb whereas fish weight showed significant moderate relationships with Cr, Mo and Ni (Figs. 5 & 6). In the liver, a moderate significant relationship was observed for fish weight and Sb (Fig. 7).
4.5 Non-Carcinogenic and Carcinogenic Risk Assessment
The non-carcinogenic THQs observed for O. mossambicus from Inanda and Nagle dams are presented in Fig. 8. Over 50% of the population at Inanda Dam and over 25% of the population at Nagle Dam exhibited THQ > 1 for Sb. Chromium is also THQ > 1 for approximately 25% of the Inanda Dam population and over 75% of the Nagle Dam population. Molybdenum was within the threshold value of 1, however, approximately 10% of the Nagle Dam population was > 0.5 (Fig. 8). The Ni THQ was less than 0.5 at both dams whereas Pb showed THQs exceeding the threshold value of 1 at both dams. Over 75% of the population at Inanda Dam exhibited THQs > 1 whereas just over 25% of Nagle Dam population exhibited THQs > 1 (Fig. 8). The CRIs were generally higher at Inanda Dam compared to Nagle Dam. The Inanda Dam populations exhibited CRIs of 1.67 × 10–1, 3.13 × 10–2, 2.45 × 10–4 for Cd, Cr and Pb, respectively whereas the Nagle Dam population showed CRIs of 3.53 × 10–2, 6.36 × 10–2 and 1.10 × 10–4 for Cd, Cr and Pb, respectively.
5 Discussion
5.1 Metals in the Water and Sediment
The uMgeni River is known for its poor water quality, particularly in the lower stretch that receives water from a tributary that drains a highly urbanized and industrialized catchment, the uMsunduzi River (Misra et al., 2024; Olaniran et al., 2014). In the present study, the neutral to alkaline pH was observed at both dams. The observed pH was complemented by metals showing undetectable concentrations in the water column and significant concentrations in sediment. The pH plays a crucial role in metal solubility and mobility in aquatic ecosystems (Arnous & Hassan, 2015). At non-acidic pH, metals precipitate and sink to the bottom sediment (Saalidong et al., 2022), which could be the explanation for the observed metal trend between the water column and bottom sediment at both dams. The Inanda Dam exhibited relatively higher metal concentrations in sediment compared to the Nagle Dam. The Inanda Dam is located downstream of the uMgeni-uMsunduzi rivers confluence (Misra et al., 2024). Given that the uMsunduzi Rivers drains a highly industrialized catchment, it is evident that its impact on the water quality is reflected in the Inanda Dam. This trend of metal concentrations between the two dams is comparable to that reported by Mdluli et al. (2023) in these dams. Moreover, these metal concentrations were comparable to those observed by Egbe et al. (2023) in wastewater-impacted River Meme in Cameroon. Moreover, concentrations reported in the sediment at both dams are substantially lower than those reported by Sojka and Jaskuła (2022) in numerous rivers impacted by industrial and agricultural activities in Poland and those reported by Lebepe et al. (2020) in an acid mine drainage impacted Olifants River in South Africa. Nevertheless, the Cd concentration observed in the present study was higher than that reported by Algül and Beyhan (2020) in wastewater and agriculture-impacted Lake Bafa in Türkiye. However, all metal concentrations in sediment were within the CCME (2001) guidelines recommended for freshwater ecosystems. Nevertheless, the anthropogenic activities impacting metal concentrations in the Inanda Dam are a cause for concern given that the dam is used for subsistence fishery by local communities.
5.2 Metal Concentrations in Fish Tissues Between Dams
Fish can accumulate metals with various organs being target sites for metal taken up through different routes (Njinga et al., 2023). Gills are in direct contact with water and were found to be a target site for waterborne metals (Zaghloul et al., 2024). Despite the water column showing concentrations below the detection limit for most metals, the gills of the Inanda Dam population showed relatively higher concentrations for Sb and Pb with Cr, Mo, and Ni being higher at Nagle Dam. Lead and Cr concentration ranges observed at both dams were lower than those observed by Pan et al. (2022) in numerous fish species from the metal-polluted Yellow River in China with Sb, Cd, and Ni being comparable. In contrast, Cd was lower than those reported by Zaghloul et al. (2024) in numerous fish species with Pb being comparable. Moreover, Cd, Cr, Ni, and Pb concentrations in the gills of the Inanda Dam population were comparable to those observed by Jayaprakash et al. (2015) in rivers impacted by wastewater works and industrial activities and exhibiting significantly low metal concentrations in the water column and substantial concentrations in sediment. The Cd concentrations were lower, with Cr and Pb being higher than those reported by Njinga et al. (2023) in the gills of Tilapia brevimanus from waters exhibiting insignificant concentrations of these metals. Gills are susceptible to waterborne metals and are among the components that sequestrate metals from the water column in polluted aquatic ecosystems, which could be the explanation for higher concentrations in the gill even when metals are significantly low in the water column.
The liver is a primary organ for detoxification, hence, a target site for metals that have already entered the body (Pan et al., 2022). This was also evident in the present study with most metals showing elevated concentration at both dams. Chromium and Mo exhibited higher concentrations in Inanda Dam than in Nagle Dam whereas other metals showed no difference between the two populations. Despite the liver being a site for detoxification, its potential to reflect concentration in the water body is poor as it does not respond rapidly to metal increase in the outside environment but stores accumulated metals (Rajeshkumar & Li, 2018). These dynamics of metal accumulation in the liver could explain the non-significant difference observed in the liver for most metals between the two dams. Nevertheless, the observed concentrations were lower than those observed in rivers impacted by wastewater works and industrial activities (Arantes et al., 2016; Blankson et al., 2024; Jayaprakash et al., 2015) and comparable to those observed by Zaghloul et al. (2024) in a non-polluted Bardawil Lake in Egypt. In contrast, Cr, Cd and Ni were higher than those reported by Kotacho et al. (2024) in Lates niloticus and Oreochromis niloticus from a waterbody impacted by industrial and agricultural activities. Moreover, Cd and Pb concentrations were relatively higher than those reported by Hasan et al. (2023) in numerous fish species from wastewater effluents-impacted Turag River in Bangladesh, with Cr being lower. Metal concentrations accumulating in the liver showed no definite trend in relation to concentrations in the water or sediment (Hasan et al., 2023; Jayaprakash et al., 2015; Yang et al., 2023). The metal concentration in the liver did not reflect the trend observed in sediment between the two dams. This could be expected as metals reaching the liver could be through both dietary and external environmental exposure.
Despite the muscle location relative to the external water environment, it does not respond rapidly to metal increase in the water column as it does not play any metabolic role (Blankson et al., 2024; Pan et al., 2022). However, a trend similar to that observed for the gill was observed in the muscle with Sb and Pb showing relatively higher concentrations in the Inanda Dam population and Cr, Mo, and Ni showing higher concentrations at Nagle Dam. Metal concentration ranges observed in the present study are comparable to those observed in other related studies on Labeo umbratus (Gilbert et al., 2017), Labeo rosae, and O. mossambicus (Jooste et al., 2015; Lebepe et al., 2020), Tilapia brevimanus and Euthynnus alletteratus (Njinga et al., 2023), and Oreochromis niloticus and Coptodon kottae (Egbe et al., 2023) from metal polluted waters. Moreover, Arantes et al. (2016) reported relatively higher Pb concentrations in a polluted inter-state São Francisco River compared to that observed at Inanda Dam with comparable concentrations observed for Cr and Cd for both populations. Despite metal concentrations in muscle being not conclusive in segregating the two populations, it is evident that the concentration in the muscle does not rapidly respond to the increase in the external water environment. However, analysis of metal concentrations in muscle tissues remains a crucial exercise as muscle is the part consumed by humans.
Despite metal concentrations showing differences between organs, it is evident that metal pollution could be detrimental to aquatic environments. Metals are found in minute concentrations in the water column with much sinking to the bottom sediment. This was also observed in the present study which could be problematic since changes in physico-chemical properties such as pH, water hardness, salinity, temperature etc. could result in metal bioavailability through resuspension back into the water column (Ali et al., 2019; Olayinka-Olagunju et al., 2021). This metal bioavailability through resuspension phenomenon has been a cause for concern in water bodies exhibiting elevated concentrations of metals, as it has implications on the well-being of the environment.
5.3 Metal Concentration Between Organs
The liver and gills are usually the target organs for contaminants due to their role in fish physiology whereas muscle does not have any metabolic role (Ali et al., 2019). In the present study, a descending trend, liver > gill > muscle was observed for Sb, Ni, and Pb at both dams with the inclusive of Cd at Nagle Dam. On the contrary, Cd and Mo at Inanda Dam and Cr and Mo at Nagle Dam exhibited higher concentrations in the muscle compared to other tissues. The liver is a primary site for detoxification whereas gills are in direct contact with the external water environments (Shahjahan et al., 2022), hence, the elevated concentration of metals in both organs is not uncommon. As mentioned earlier, fish muscle usually exhibits low concentrations compared to gills and liver. The liver > gill > muscle trend is comparable to those observed in other related studies in polluted waters (Afandi et al., 2018; Mahboob et al., 2016; Marr et al., 2017). However, other trends such as muscle > liver > gill for Cd, liver > muscle > gill for Cr, and muscle > gill > liver for Mo were observed at Inanda Dam with liver > gill > muscle for Sb, Cd, Ni, and Pb, and muscle > gill > liver for Cr and Mo being observed at Nagle Dam. Shovon et al. (2017) reported an uncommon trend for Cd, Cr, and Mo where the muscle of Labeo rohita and Gibelion catla exhibited higher concentrations compared to the gills. Moreover, Bawuro et al. (2018) reported muscle > gill > liver for Pb and Cd on C. gariepinus and Tilapia zilli.
Despite Inanda and Nagle dam populations exhibiting relatively higher concentrations in the muscle than other tissues for some metals, Sb, Cd, and Cr were within the recommended limits of 1.5 mg/kg (FAO, 1983), 0.3 mg/kg (OJEU, 2006), and 2 mg/kg (MOHSAC, 2006), respectively. On the contrary, Pb concentration exceeded the FAO (2003) and OJEU (2006) limit of 0.02 mg/kg at both dams with the Inanda Dam population exhibiting higher concentration. Moreover, the Ni concentration exceeded the limit of 0.5 mg/kg (UNEP/WHO, 1991) at both dams. Generally, the concentrations of Pb and Ni are concerning and their edibility should be monitored closely.
5.4 Bioaccumulation Factor
The bioaccumulation factor (BAF) may be used to tell whether metals have bioaccumulated or not. In the present study metal concentrations in the water column were below the detection level, hence, the BAF for the water (BAFw) could not be calculated. On the contrary, the bioaccumulation factor for sediment (BAFs) exhibited values ranging from < 1 to > 1. Molybdenum and Ni exhibited BAFs of > 1 in all tissues at Nagle Dam whereas the Inanda Dam population exhibited the BAFs of > 1 for Ni. According to Melake et al. (2023) a BAF of > 1 indicates that metal accumulated more in a tissue whereas a BAF of < 1 suggests that metals accumulated more in sediment and they were not biologically available for fish. The BAFs observed in the present study suggests that Mo and Ni at Nagle Dam may have been biologically available to be taken up by fish. Melake et al. (2023) reported the BAFs of < 1 for Cd, Cr, Ni, and Pb in the muscle of C. gariepinus. In contrast, Olayinka-Olagunju et al. (2021) observed the BAFs > 1 for Pb in the muscle of Parachanna obscura from the Ogbese River. Nevertheless, BAF can be classified into three categories, namely: no probability of accumulation (BAF < 1000), bioaccumulative (1000 < BAF < 5000), and extremely bioaccumulative (BAF > 5000) (Arnot & Gobas, 2006). Based on Arnot and Gobas (2006) classification, there is no probability of accumulation for all metals at both dams. However, this interpretation may be misleading particularly in multi-compartment subjects such as fish where different organs accumulate metals differently depending on their roles, location relative to the medium, and affinity to these metals.
5.5 Metal-Fish Length and Inter-Metal Correlation
Metal accumulation may be influenced by factors such as pH, salinity, temperature, fish size, life history, feeding habits, and fish metabolic rate (Bawuro et al., 2018; Echevarría et al., 2024). According to Jezierska and Witeska (2006), the presence of other metals may also influence bioaccumulation in fish. As a result, metals may display antagonistic or synergistic associations during accumulation. In the present study, inter-metal relationships were observed for Cr–Mo, Cr-Ni, Mo-Ni, and Sb-Pb with fish length showing positive relationships with Cr, Mo, and Ni in the gill and muscle. None of the inter-metal relationships observed in the gill and muscle were observed in the liver with only Sb showing a moderate positive relationship with fish length. Inter-metal relationships observed in the gill and muscle differ from those observed in other related studies (Ahmed et al., 2019; Hashim et al., 2014; Jayaprakash et al., 2015). However, Khan et al. (2023) reported a strong positive relationship for Cr-Ni, Cr-Pb, Ni-Pb in the muscle. Despite liver showing a positive relationship for fish length and Sb, Jezierska and Witeska (2006) and Echevarría et al. (2024) reported that metal concentration may decrease with increasing fish size due to a reduction in feeding as the fish grows, a high metabolic rate in smaller fish and/or dilution factor. The present study has shown not definite trend with regard to inter-metal relationship for all three tissues which affirms that metal affinity, and the physiology, age and sex of fish are among the drivers of metal concentration increase in fish tissues.
5.6 Non-Carcinogenic and Carcinogenic Risk Assessment
Consumption of fish has recently become a cause for concern due to increasing pollution levels in aquatic ecosystems. Despite fish muscle not being the target organ for metals and accumulating lower concentrations compared to other organs such as the liver and gills, these low concentrations are reported to have the probability to induce adverse threats to human health. In the present study, THQs > 1 were observed for Sb, Cr and Pb at both dams with Mo showing a THQ > 1 at Nagle Dam. The Cr and Pb THQs observed in the present study were comparable to those reported further downstream of the river, in the estuarine area of the Indian Ocean for Trachurus trachurus and Chrysoblephus puniceus (Debipersadh et al., 2023), and in the Delta River impacted by industrial, urbanization, and intensive agricultural activities in Ethiopia for Lates niloticus and Oreochromis niloticus (Kotacho et al., 2024). Moreover, Esilaba et al. (2020) observed the Pb THQ of > 1 in Oreochromis niloticus from urban Lake Nakuru in Kenya whereas Lebepe et al. (2020) observed THQ exceeding the acceptable threshold on 1 for Sb on O. mossambicus and L. rosae from water bodies impacted by acid mine drainage. It is evident that anthropogenic activities in the uMgeni River catchment are causing an increase in metal concentration in the system. Other metals such as As, Se, and Hg were also found to exceed acceptable levels for human consumption in numerous fish species at the Inanda and Nagle dams (Misra et al., 2024). On the contrary, fish exhibiting THQs < 1 were was observed in water bodies used for fisheries (Huang et al., 2019; Noman et al., 2022), in protected wetlands (Hossain et al., 2023) and in an urban lake (Han et al., 2021). Nevertheless, low metal concentrations may have a carcinogenic effect if consumed over a long period of time.
In the present study, the CRIs were higher than 10–6 for Cd, Cr and Pb at both dams with the Inanda Dam population exhibiting a relatively higher index compared to Nagle Dam. Similar indices were observed for Trachurus trachurus and Chrysoblephus puniceus in the lower uMgeni River (Debipersadh et al., 2023). However, the CRIs observed at Inanda and Nagle dams were higher than those observed by Kotacho et al. (2024) at the Delta River impacted by industrial and agricultural activities. Moreover, Adegbola et al. (2021) reported CRIs higher than those observed in the present study for Pb and Ni in C. gariepinus at the industrially polluted Ogun and Aleyele rivers. Metals such as Cr, Ni and Pb are showing to be a threat to human health in water bodies impacted by industrial, agricultural and urban activities which suggest that the industrial and urban activities, wastewater works and a landfill could be the explanation for the increased metal concentrations at Inanda Dam. According to (Briffa et al., 2020), metal concentration increases in rivers draining catchments with negligible anthropogenic activities may also be influenced by the geological characteristics of the area. With the Nagle Dam bordered by mountains, the geological characteristics of the area as the driver for the metal increase could not be dismissed.
6 Conclusion
The study explored metal levels at Inanda and Nagle dams to determine the effect of tributaries joining the uMgeni River downstream of the Nagle Dam on the metal increase in the Inanda Dam. The water column exhibited concentrations below detection limits for metals with sediment showing significant concentrations. Moreover, no definite trend was observed for metal concentrations between the two dams, as some metals were higher at Inanda with some being higher at Nagle Dam. Similarly, metal accumulation in fish tissues also showed no definite trend between the two dams and a common trend of liver > gill > muscle was observed for some metals. Nevertheless, it is evident that metal pollution could be a potential threat to the well-being of aquatic life and it should be viewed with concern. Additionally, the muscle of fish exhibited THQs of > 1 for Sb, Cr and Pb with the Nagle Dam population showing a higher THQ for Cr and the Inanda Dam population showing relatively higher THQs for Sb and Pb. Moreover, Sb, Cr and Pb have also shown the probability of carcinogenic effects when consumed over a long period for both populations. Despite polluted tributaries joining the uMgeni River downstream of the Inanda Dam, notable metal concentrations were also observed at Nagle Dam. However, it is evident that the impact of these tributaries manifests in the Inanda Dam. Given the episodic discharge of acidic effluents into the system, it is recommended that the behaviour of metals should be constantly monitored for the sustainability of the well-being of aquatic biota and the safety of fish consumers.
Data Availability
Data is available upon request.
References
Adegbola, I. P., Aborisade, B. A. & Adetutu, A. (2021). Health risk assessment and heavy metal accumulation in fish species (Clarias gariepinus and Sarotherodon melanotheron) from industrially polluted Ogun and Eleyele Rivers, Nigeria. Toxicology Reports 81445–1460. https://doi.org/10.1016/j.toxrep.2021.07.007
Adeyinka, G. (2014). 'The determination of organochlorinated pesticides and polychlorinated biphenyls in sediment, soil and water of the Msunduzi River, KwaZulu-Natal, South Africa', MSc Dissertation, University of Kwa-Zulu Natal, Durban.
Afandi, I., Talba, S., Benhra, A., Benbrahim, S., Chfiri, R., Labonne, M., Masski, H., Laë, R., Tito De Morais, L., Bekkali, M., & Bouthir, F. Z. (2018). Trace metal distribution in pelagic fish species from the north-west African coast (Morocco). International Aquatic Research, 10(2), 191–205. https://doi.org/10.1007/s40071-018-0192-7
Ahmed, A. S., Sultana, S., Habib, A., Ullah, H., Musa, N., Hossain, M. B., Rahman, M. M., & Sarker, M. S. I. (2019). Bioaccumulation of heavy metals in some commercially important fishes from a tropical river estuary suggests higher potential health risk in children than adults. PLoS ONE, 14(10), e0219336. https://doi.org/10.1371/journal.pone.0219336
Algül, F., & Beyhan, M. (2020). Concentrations and sources of heavy metals in shallow sediments in Lake Bafa. Turkey. Scientific Reports, 10(1), 11782. https://doi.org/10.1038/s41598-020-68833-2
Ali, H., Khan, E., & Ilahi, I. (2019). Environmental Chemistry and Ecotoxicology of Hazardous Heavy Metals: Environmental Persistence, Toxicity, and Bioaccumulation. Journal of Chemistry, 2019, 6730305. https://doi.org/10.1155/2019/6730305
Arantes, F. P., Savassi, L. A., Santos, H. B., Gomes, M. V., & Bazzoli, N. (2016). Bioaccumulation of mercury, cadmium, zinc, chromium, and lead in muscle, liver, and spleen tissues of a large commercially valuable catfish species from Brazil. Anais Da Academia Brasileira De Ciências, 88(1), 137–147. https://doi.org/10.1590/0001-3765201620140434
Arnot, J. A., & Gobas, F. A. (2006). A review of bioconcentration factor (BCF) and bioaccumulation factor (BAF) assessments for organic chemicals in aquatic organisms. Environmental Reviews, 14(4), 257–297. https://doi.org/10.1139/a06-005
Arnous, M. O., & Hassan, M. A. A. (2015). Heavy metals risk assessment in water and bottom sediments of the eastern part of Lake Manzala, Egypt, based on remote sensing and GIS. Arabian Journal of Geosciences, 8(10), 7899–7918. https://doi.org/10.1007/s12517-014-1763-6
Bawuro, A. A., Voegborlo, R. B. & Adimado, A. A. (2018). Bioaccumulation of Heavy Metals in Some Tissues of Fish in Lake Geriyo, Adamawa State, Nigeria. Journal of Environmental and Public Health, 1854892. 1854810.1851155/1852018/1854892
Blankson, E. R., Ohene-Obeng, N. K. A., Awuah, B. A., Oduro, D., Ewool, J., & Gbogbo, F. (2024). Heavy Metal Bioaccumulation in Highly Consumed Pelagic and Benthic Fish and Associated Health Risk. Biological Trace Element Research, 202(8), 3781–3788. https://doi.org/10.1007/s12011-023-03943-2
Briffa, J., Sinagra, E., & Blundell, R. (2020). Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon, 6(9), e04691. https://doi.org/10.1016/j.heliyon.2020.e04691
CCME (2001). Canadian sediment quality guidelines for the protection of aquatic life. Retrieved January 25, 2018, from https://www.elaw.org/system/files/sediment_summary_table.pdf
CCME (2012). Canadian water quality guidelines for the protection of aquatic life. Introduction, Canadian environmental quality guidelines, Canada. Retrieved September 9, 2019, from http://st-ts.ccme.ca/en/index.html
Debipersadh, S., Ogola, H. J. O., Mearns, K., & Selvarajan, R. (2023). Bioaccumulation, Bioindication and Health Risk Assessment of Heavy Metals in Cape Horse Mackerel (Trachurus trachurus) and Slinger Seabream (Chrysoblephus puniceus) in the Durban Basin and Cape Vidal, South Africa. Archives of Environmental Contamination and Toxicology, 85(2), 165–180. https://doi.org/10.1007/s00244-023-01028-8
DWAF: 1996, South African Water Quality Guideline, Volume 7, Aquatic Ecosystem, 2nd Edition, Pretoria. Retrieved January 23, 2016, from https://www.dws.gov.za/iwqs/wq_guide/edited/Pol_saWQguideFRESH_vol7_Aquaticecosystems.pdf
Echevarría, G., Lujan, N. K., Montoya, J., Granda-Albuja, M. G., Valdiviezo-Rivera, J., Sánchez, F., Cuesta, F., & Ríos-Touma, B. (2024). Abiotic and biotic factors influencing heavy metals pollution in fisheries of the Western Amazon. Science of The Total Environment, 908, 168506. https://doi.org/10.1016/j.scitotenv.2023.168506
Egbe, A. M., Tabot, P. T., Fonge, B. A., & Ngole-Jeme, V. M. (2023). Heavy metal exposure risk associated with ingestion of Oreochromis niloticus and Coptodon kottae harvested from a lacustrine ecosystem. Environmental Monitoring and Assessment, 195(3), 427. https://doi.org/10.1007/s10661-023-10936-0
Esilaba, F., Moturi, W. N., Mokua, M., & Mwanyika, T. (2020). Human health risk assessment of trace metals in the commonly consumed fish species in Nakuru Town. Kenya. Environmental Health Insights, 14, 1178630220917128. https://doi.org/10.1177/1178630220917128
FAO: 1983, 'Complation of legal limits for hazardous substances in fish and fishery products', FAO Fisheries Circular No. 764. Rome. Retrieved May 23, 2024, from https://www.fao.org/inland-fisheries/topic/detail/ar/c/1150083/
FAO: 2003, 'Heavy metals regulations ', Legal Notice No. 66/2003. Retrived June 8, 2018, from http://extwprlegs1.fao.org/docs/pdf/eri42405.pdf
Gilbert, B. M., Hussain, E., Jirsa, F., & Avenant-Oldewage, A. (2017). Evaluation of Trace Element and Metal Accumulation and Edibility Risk Associated with Consumption of Labeo umbratus from the Vaal Dam, South Africa. International Journal of Environmental Research and Public Health, 14(7), 678–693. https://doi.org/10.3390/ijerph14070678
Gumbi, B. P., Moodley, B., Birungi, G., & Ndungu, P. G. (2017). Detection and quantification of acidic drug residues in South African surface water using gas chromatography-mass spectrometry. Chemosphere, 168, 1042–1050. https://doi.org/10.1016/j.chemosphere.2016.10.105
Han, Y., & Gu, X. (2023). Enrichment, contamination, ecological and health risks of toxic metals in agricultural soils of an industrial city, northwestern China. Journal of Trace Elements and Minerals, 3, 100043. https://doi.org/10.1016/j.jtemin.2022.100043
Han, J.-L., Pan, X.-D., Chen, Q., & Huang, B.-F. (2021). Health risk assessment of heavy metals in marine fish to the population in Zhejiang. China. Scientific Reports, 11(1), 11079. https://doi.org/10.1038/s41598-021-90665-x
Hao, Y., Miao, X., Song, M., & Zhang, H. (2022). The Bioaccumulation and Health Risk Assessment of Metals among Two Most Consumed Species of Angling Fish (Cyprinus carpio and Pseudohemiculter dispar) in Liuzhou (China): Winter Should Be Treated as a Suitable Season for Fish Angling. International Journal of Environmental Research and Public Health, 19(3), 1519. https://doi.org/10.3390/ijerph19031519
Hasan, G. M. M. A., Das, A. K., Satter, M. A., & Asif, M. (2023). Distribution of Cr, Cd, Cu, Pb and Zn in organs of three selected local fish species of Turag river, Bangladesh and impact assessment on human health. Emerging Contaminants, 9(1), 100197. https://doi.org/10.1016/j.emcon.2022.11.002
Hashim, R., Song, T. H., Muslim, N. Z., & Yen, T. P. (2014). Determination of Heavy Metal Levels in Fishes from the Lower Reach of the Kelantan River, Kelantan. Malaysia. Tropical Life Sciences Research, 25(2), 21–39.
Heath, R., Du Preez, H., Genthe, B., & Avenant-Oldewage, A. (2004). Freshwater Fish and Human Health Reference Guide, WRC Report No TT213/04. Pretoria: Water Research Commission.
Hossain, M. B., Ahmed, M. M., Jolly, Y. N., Nur, A. U., Sultana, S., Akter, S., Yu, J., Paray, B. A. & Arai, T. (2023). Potential Toxic Elements and Their Carcinogenic and Non-Carcinogenic Risk Assessment in Some Commercially Important Fish Species from a Ramsar Site. Biology (Basel) 12(8). https://doi.org/10.3390/biology12081072
Huang, X., Qin, D., Gao, L., Hao, Q., Chen, Z., Wang, P., Tang, S., Wu, S., Jiang, H., & Qiu, W. (2019). Distribution, contents and health risk assessment of heavy metal(loid)s in fish from different water bodies in Northeast China. RSC Advances, 9(57), 33130–33139. https://doi.org/10.1039/C9RA05227E
Jayaprakash, M., Kumar, R. S., Giridharan, L., Sujitha, S. B., Sarkar, S. K., & Jonathan, M. P. (2015). Bioaccumulation of metals in fish species from water and sediments in macrotidal Ennore creek, Chennai, SE coast of India: A metropolitan city effect. Ecotoxicology and Environmental Safety, 120, 243–255. https://doi.org/10.1016/j.ecoenv.2015.05.042
Jezierska, B., & Witeska, M. (2006). The metal uptake and accumulation in fish living in polluted waters. In I. Twardowska, H. E. Allen, M. M. Häggblom, M. M. Häggblom, S. Stefaniak, & S. Stefaniak (Eds.), Soil and water pollution monitoring, protection and remediation (pp. 107–114). Poland.
Jooste, A., Marr, S., Addo-Bediako, A., & Luus-Powell, W. (2015). Sharptooth catfish shows its metal: A case study of metal contamination at two impoundments in the Olifants River, Limpopo River system, South Africa. Ecotoxicology and Environmental Safety, 112, 96–112104. https://doi.org/10.1016/j.ecoenv.2014.10.033
Khan, K., Zeb, M., Younas, M., Sharif, H. M. A., Yaseen, M., Al-Sehemi, A. G., Kavil, Y. N., Shah, N. S., Cao, X., Maryam, A., & Qasim, M. (2023). Heavy metals in five commonly consumed fish species from River Swat, Pakistan, and their implications for human health using multiple risk assessment approaches. Marine Pollution Bulletin, 195, 115460. https://doi.org/10.1016/j.marpolbul.2023.115460
Kotacho, A. A., Yimer, G. T., Sota, S. S., & Berego, Y. S. (2024). Level of Heavy Metals in Fish and Associated Human Health Risk From the Omo Delta in Southern Ethiopia: A First-Hand Report. Environmental Health Insights, 18, 11786302241238180. https://doi.org/10.1177/11786302241238180
Kumar, S., Raman, R. K., Talukder, A., Mahanty, A., Sarkar, D. J., Das, B. K., Bhowmick, S., Samanta, S., Manna, S. K., & Mohanty, B. P. (2022). Arsenic Bioaccumulation and Identification of Low-Arsenic-Accumulating Food Fishes for Aquaculture in Arsenic-Contaminated Ponds and Associated Aquatic Ecosystems. Biological Trace Element Research, 200(6), 2923–2936. https://doi.org/10.1007/s12011-021-02858-0
Lakra, K. C., Lal, B., & Banerjee, T. K. (2019). Coal mine effluent-led bioaccumulation of heavy metals and histopathological changes in some tissues of the catfish Clarias batrachus. Environmental Monitoring and Assessment, 191(3), 136. https://doi.org/10.1007/s10661-019-7229-2
Lebepe, J., Marr, S., & Luus-Powell, W. (2016). Metal contamination and human health risk associated with the consumption of Labeo rosae from the Olifants River system, South Africa. African Journal of Aquatic Science, 41(2), 161–170. https://doi.org/10.2989/16085914.2016.1138100
Lebepe, J., Oberholster, P. J., Ncube, I., Smit, W., & Luus-Powell, W. J. (2020). Metal levels in two fish species from a waterbody impacted by metallurgic industries and acid mine drainage from coal mining in South Africa. Journal of Environmental Science and Health, Part A, 55(4), 421–432. https://doi.org/10.1080/10934529.2019.1704604
Mahboob, S., Kausar, S., Jabeen, F., Sultana, S., Sultana, T., Al-Ghanim, K., Hussain, B., Al-Misned, F., & Ahmed, Z. (2016). Effect of heavy metals on liver, kidney, gills and muscles of Cyprinus carpio and Wallago attu inhabited in the Indus. Brazilian Archives of Biology and Technology, 59, e16150275. https://doi.org/10.1590/1678-4324-2016150275
Marr, S. M., Lebepe, J., Steyl, J. C. A., Smit, W. J., & Luus-Powell, W. J. (2017). Bioaccumulation of selected metals in the gill, liver and muscle tissue of rednose labeo Labeo rosae from two impoundments on the Olifants River, Limpopo river system, South Africa. African Journal of Aquatic Science, 42(2), 123–130. https://doi.org/10.2989/16085914.2017.1351915
Mdluli, S., Vosloo, D., & Lebepe, J. (2023). Biochemical and Histopathologic Biomarkers of Pollution in the uMgeni River System in KwaZulu-Natal, South Africa. Polish Journal of Environmental Studies, 32(5), 4739–4752. https://doi.org/10.15244/pjoes/168136
Melake, B. A., Endalew, S. M., Alamirew, T. S., & Temesegen, L. M. (2023). Bioaccumulation and Biota-Sediment Accumulation Factor of Metals and Metalloids in Edible Fish: A Systematic Review in Ethiopian Surface Waters. Environ Health Insights, 17, 11786302231159348. https://doi.org/10.1177/11786302231159349
Mishra, V. K., Upadhyaya, A. R., Pandey, S. K., & Tripathi, B. D. (2008). Heavy metal pollution induced due to coal mining effluent on surrounding aquatic ecosystem and its management through naturally occurring aquatic macrophytes. Bioresource Technology, 99(5), 930–936. https://doi.org/10.1016/j.biortech.2007.03.010
Misra, A., Bissessur, A., Selala, M. C., & Lebepe, J. (2024). Accumulation and health implications of arsenic, mercury, and selenium in selected freshwater fish species in the uMgeni River. South Africa. Environmental Pollutants and Bioavailability, 36(1), 2296973. https://doi.org/10.1080/26395940.2023.2296973
MOHSAC: 2006, 'China, peoples Republic of China's maximum levels of contaminants in foods', GAIN Report No. CH6064. USDA Foreign Agricultural Service: Beijing, China. Retrieved January 18, 2024, from https://apps.fas.usda.gov/newgainapi/api/report/downloadreportbyfilename?filename=Maximum%20Levels%20of%20Contaminants%20in%20Foods%20_Beijing_China%20-%20Peoples%20Republic%20of_12-11-2014.pdf
Njinga, R. L., Adebayo, A. S., & Olufemi, A. P. (2023). Bioaccumulation of heavy metals in water and organs of Tilapia brevimanus and Euthynnus alletteratus from a coaster water in southwestern Nigeria. Environmental Sciences Europe, 35(1), 85. https://doi.org/10.1186/s12302-023-00794-5
Noman, M. A., Feng, W., Zhu, G., Hossain, M. B., Chen, Y., Zhang, H., & Sun, J. (2022). Bioaccumulation and potential human health risks of metals in commercially important fishes and shellfishes from Hangzhou Bay. China. Scientific Reports, 12(1), 4634. https://doi.org/10.1038/s41598-022-08471-y
OJEU (2006). Commission Regulation No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Official Journal of the European Union. Retrieved June 03, 2024, from https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:364:0005:0024:EN:PDF
Oksanen, J., Blanchet, F., Kindt, R., Legendre, P., O’Hara, R., Simpson, G., Solymos, P., Stevens, M. & Wagner, H.: 2013, VEGAN: Community Ecology Package version 2.0–9. Retrieved July 18, 2022, from http://RForgeR-project.org/projects/vegan/
Olaniran, A. O., Naicker, K., & Pillay, B. (2014). Assessment of physico-chemical qualities and heavy metal concentrations of uMgeni and Umdloti Rivers in Durban. South Africa. Environmental Monitoring and Assessment, 186(4), 2629–2639. https://doi.org/10.1007/s10661-013-3566-8
Olayinka-Olagunju, J. O., Dosumu, A. A., & Olatunji-Ojo, A. M. (2021). Bioaccumulation of Heavy Metals in Pelagic and Benthic Fishes of Ogbese River, Ondo State, South-Western Nigeria. Water, Air, & Soil Pollution, 232(2), 44. https://doi.org/10.1007/s11270-021-04987-7
Oosthuizen, J., & Ehrlich, R. (2001). The impact of pollution from a mercury processing plant in KwaZulu-Natal, South Africa, on the health of fish-eating communities in the area: An environmental health risk assessment. International Journal of Environmental Health Research, 11(1), 41–50. https://doi.org/10.1080/09603120020019638
Pal, D., & Maiti, S. K. (2018). Seasonal variation of heavy metals in water, sediment, and highly consumed cultured fish (Labeo rohita and Labeo bata) and potential health risk assessment in aquaculture pond of the coal city, Dhanbad (India). Environmental Science Pollution Research, 25(13), 12464–12480. https://doi.org/10.1007/s11356-018-1424-5
Pan, B., Wang, Y., Li, D., Wang, T., & Du, L. (2022). Tissue-specific distribution and bioaccumulation pattern of trace metals in fish species from the heavily sediment-laden Yellow River. China. Journal of Hazardous Materials, 425, 128050. https://doi.org/10.1016/j.jhazmat.2021.128050
Peterson, B. G., Carl, P., Boudt, K., Bennett, R., Ulrich, J., Zivot, E., Cornilly, D., Hung, E., Lestel, M., Balkissoon, K., Wuertz, D., Christidis, A. A., Martin, R. D., Zhou, Z. Z. & Shea, J. M. (2020). Econometric Tools for Performance and Risk Analysis: Package ‘PerformanceAnalytics’. Retrieved July 18, 2022, from https://github.com/braverock/PerformanceAnalytics
R Development Core Team: 2014, R: A Language and Environment for Statistical Computing., R Foundation for Statistical Computing, Vienna, Austria. Retrieved July 18, 2022, from http://www.Rproject.org
Rajeshkumar, S., & Li, X. (2018). Bioaccumulation of heavy metals in fish species from the Meiliang Bay, Taihu Lake, China. Toxicology Reports, 5, 288–295. https://doi.org/10.1016/j.toxrep.2018.01.007
Saalidong, B. M., Aram, S. A., Otu, S., & Lartey, P. O. (2022). Examining the dynamics of the relationship between water pH and other water quality parameters in ground and surface water systems. PLoS ONE, 17(1), e0262117. https://doi.org/10.1371/journal.pone.0262117
Shahjahan, M., Taslima, K., Rahman, M. S., Al-Emran, M., Alam, S. I., & Faggio, C. (2022). Effects of heavy metals on fish physiology – A review. Chemosphere, 300, 134519. https://doi.org/10.1016/j.chemosphere.2022.134519
Shovon, M., Majumdar, B., & Rahman, Z. (2017). Heavy metals (lead, cadmium and nickel) concentration in different organs of three commonly consumed fishes in Bangladesh. Fisheries and Aquaculture Journal, 8(3), 1–6. https://doi.org/10.4172/2150-3508.1000207
Şirin, M., Bayrak, E. Y., & Baltaş, H. (2024). Human health risk assessment of heavy metals accumulation in different genders and tissues of whiting fish (Merlangius merlangus euxinus Nordmann, 1840) from Rize, Turkey. Journal of Food Composition and Analysis, 127, 105971. https://doi.org/10.1016/j.jfca.2024.105971
Sojka, M. & Jaskuła, J. (2022). Heavy Metals in River Sediments: Contamination, Toxicity, and Source Identification-A Case Study from Poland. International Journal of Environmental Research and Public Health 19(17). https://doi.org/10.3390/ijerph191710502
Soleimani, H., Mansouri, B., Kiani, A., Omer, A. K., Tazik, M., Ebrahimzadeh, G., & Sharafi, K. (2023). Ecological risk assessment and heavy metals accumulation in agriculture soils irrigated with treated wastewater effluent, river water, and well water combined with chemical fertilizers. Heliyon, 9(3), e14580. https://doi.org/10.1016/j.heliyon.2023.e14580
UNEP/WHO (1991). Nickel, Nickel Carbonyl and Some Nickel Compounds: Health and Safety Guide No 62. Geneva: World Health Organization. Retrieved June 3, 2024, from https://www.inchem.org/documents/hsg/hsg/hsg062.htm
US-EPA (2000). 'Guidance for assessing chemical contaminant data for use in fish advisories. Volume 1, Fish Sampling and Analysis', Report No. EPA 823-B-00–007. Washington DC. Retrived January 23, 2018, from https://www.epa.gov/sites/default/files/2015-06/documents/volume2.pdf
US-EPA (2013). ‘Integrated Risk Information System (IRIS).’ United States Environmental Protection Agency, Environmental Criteria and Assessment Office. Retrieved July 5, 2021, from http://www.epa.gov/IRIS/
Yang, F., Zhang, H., Xie, S., Wei, C., & Yang, X. (2023). Concentrations of heavy metals in water, sediments and aquatic organisms from a closed realgar mine. Environmental Science and Pollution Research, 30(2), 4959–4971. https://doi.org/10.1007/s11356-022-22563-2
Zaghloul, G. Y., Eissa, H. A., Zaghloul, A. Y., Kelany, M. S., Hamed, M. A., & Moselhy, K. M. E. (2024). Impact of some heavy metal accumulation in different organs on fish quality from Bardawil Lake and human health risks assessment. Geochemical Transactions, 25(1), 1. https://doi.org/10.1186/s12932-023-00084-2
Funding
Open access funding provided by Sefako Makgatho Health Sciences University. This work was supported by the National Research Foundation (NRF) of South Africa (Grant numbers [121848].
Author information
Authors and Affiliations
Contributions
Jeffrey Lebepe: Conceptualisation, sample collection and processing, sample analyses, manuscript drafting, and funding acquisition.
Mapurunyane Callies Selala: Sample analyses, data generation, and drafting of the manuscript.
Yuki Takai: Conceptualisation, data analyses, and interpretation and manuscript editing.
Maleka Neo: Data analysis, manuscript drafting, sample analyses, manuscript editing.
Sanelisiwe Siphumelele Brightness Hlatshwayo: Conceptualisation, collection, processing, and analyses of samples, and interpretation, and manuscript editing.
Corresponding author
Ethics declarations
Consent to publish
All authors have read, understood, and complied as applicable with the statement on “Ethical responsibilities of Authors” as found in the Instructions for Authors.
Ethical Approval
The study was approved by the Animal Research Ethics Committee of the University of KwaZulu-Natal (Ref: AREC/019/018).
Conflict of interest
No conflict of interest to be declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lebepe, J., Selala, M.C., Takai, Y. et al. Accumulation of Heavy Metals in Tissues of Oreochromis Mossambicus from the uMgeni River and Human Health Risk Assessment. Water Air Soil Pollut 235, 726 (2024). https://doi.org/10.1007/s11270-024-07553-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-024-07553-z